Keywords
(S)-methionine S-oxide
diastereomers
kinetic resolution
methionine-γ-lyase
stereoselectivity
Abstract
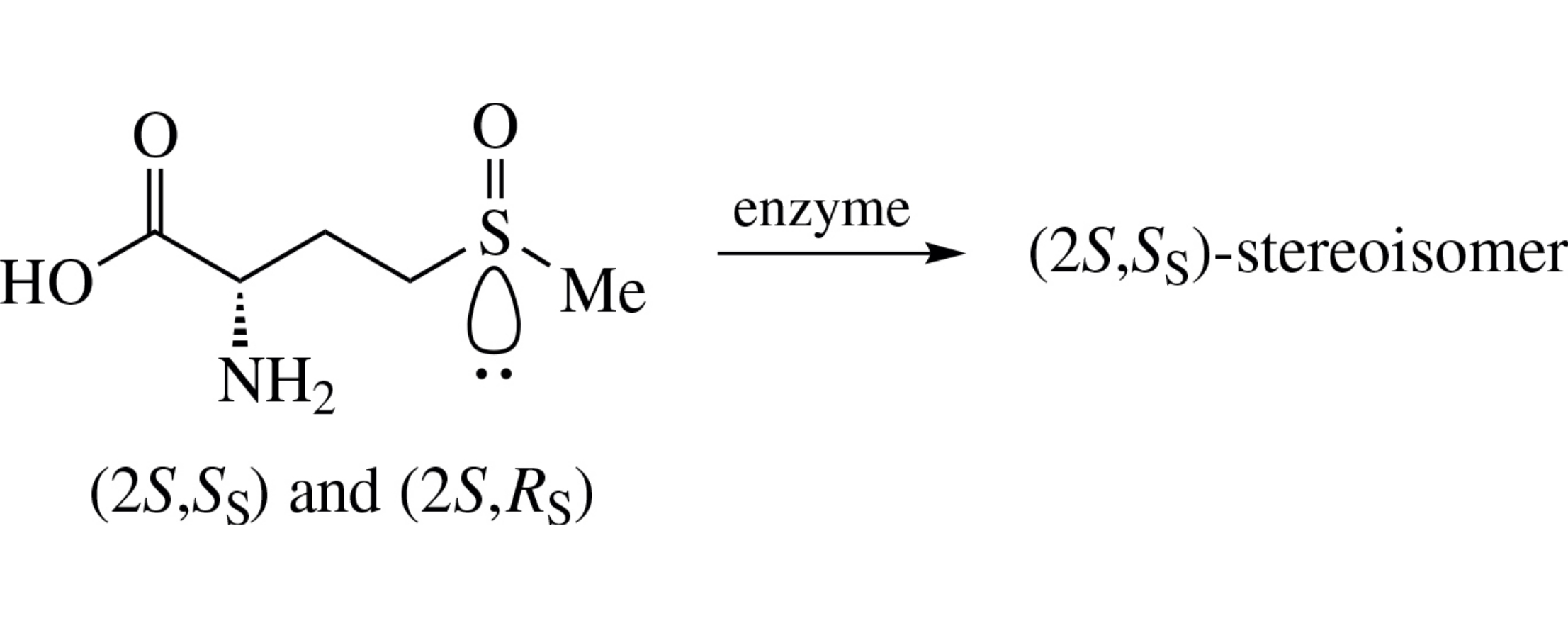
Using a diastereomeric mixture of (S)-methionine S-oxide as an example, kinetic preference of methionine-γ-lyase toward a stereogenic center at the γ-sulfur atom of the (2S, RS) diastereomer was discovered for the first time.
References
1.
10.1016/j.mencom.2021.03.030_bib0005
Bornscheuer
Hydrolases in Organic Synthesis,
2006
2.

Stergiou P., Foukis A., Filippou M., Koukouritaki M., Parapouli M., Theodorou L.G., Hatziloukas E., Afendra A., Pandey A., Papamichael E.M.
Biotechnology Advances,
2013
3.

Kamal A., Khanna G.B., Ramu R., Krishnaji T.
Tetrahedron Letters,
2003
4.

Luna A., Alfonso I., Gotor V.
Organic Letters,
2002
5.

Sakai T., Miki Y., Tsuboi M., Takeuchi H., Ema T., Uneyama K., Utaka M.
Journal of Organic Chemistry,
2000
6.

Liu R., Berglund P., Högberg H.
Tetrahedron Asymmetry,
2005
7.

8.

Souza R.O., Matos L.M., Gonçalves K.M., Costa I.C., Babics I., Leite S.G., Oestreicher E.G., Antunes O.A.
Tetrahedron Letters,
2009
9.

Plieva F.M., Kochetkov K.A., Singh I., Parmar V.S., Belokon' Y.N., Lozinsky V.I.
Biotechnology Letters,
2000
10.

Markvicheva E.A., Kuptsova S.V., Mareeva T.Y., Vikhrov A.A., Dugina T.N., Strukova S.M., Belokon Y.N., Kochetkov K.A., Baranova E.N., Zubov V.P., Poncelet D., Parmar V.S., Kumar R., Rumsh L.D.
Applied Biochemistry and Biotechnology,
2000
11.
Kochetkov K.A., Galkina M.A., Galkin O.M.
Mendeleev Communications,
2010
12.
Alferov K.V., Faleev N.G., Khurs E.N., Zhukov Y.N., Khomutov R.M.
Mendeleev Communications,
2002
13.
Alferov K.V., Zhukov Y.N., Faleev N.G., Khurs E.N., Khomutov R.M.
Mendeleev Communications,
2003
14.

Kolodiazhna O.O., Kolodiazhna A.O., Kolodiazhnyi O.I.
Tetrahedron Asymmetry,
2013
15.

Gogoi S., Argade N.P.
Tetrahedron Asymmetry,
2006
16.

Saravanan T., Selvakumar R., Doble M., Chadha A.
Tetrahedron Asymmetry,
2012
17.

Tanaka H., Esaki N., Soda K.
Enzyme and Microbial Technology,
1985
18.

Engineered Citrobacter freundii methionine γ-lyase effectively produces antimicrobial thiosulfinates
Morozova E.A., Kulikova V.V., Rodionov A.N., Revtovich S.V., Anufrieva N.V., Demidkina T.V.
Biochimie,
2016
19.

Kulikova V.V., Anufrieva N.V., Revtovich S.V., Chernov A.S., Telegin G.B., Morozova E.A., Demidkina T.V.
IUBMB Life,
2016
20.

Kulikova V., Morozova E., Rodionov A., Koval V., Anufrieva N., Revtovich S., Demidkina T.
Biochimie,
2018
21.

Mecham J.O., Rowitch D., Wallace C.D., Stern P.H., Hoffman R.M.
Biochemical and Biophysical Research Communications,
1983
22.

Hoffman R.M.
Expert Opinion on Biological Therapy,
2014
23.

Kawaguchi K., Han Q., Li S., Tan Y., Igarashi K., Murakami T., Unno M., Hoffman R.M.
Cells,
2019
24.

Messerschmidt A., Worbs M., Steegborn C., Wahl M.C., Huber R., Laber B., Clausen T.
Biological Chemistry,
2003
25.

Nosek V., Míšek J.
Chemical Communications,
2019
26.

Yang J., Wen Y., Peng L., Chen Y., Cheng X., Chen Y.
Organic and Biomolecular Chemistry,
2019
27.

Dunina V.V., Razmyslova E.D., Gorunova O.N., Livantsov M.V., Yuri K.Grishin
Tetrahedron Asymmetry,
2005
28.
![First enantiopure imine CN-palladacycle of non-metallocenic planar chirality with the [2.2]paracyclophane backbone](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Dunina V.V., Turubanova E.I., Livantsov M.V., Lyssenko K.A., Vorontsova N.V., Antonov D.Y., Grishin Y.K.
Tetrahedron Asymmetry,
2009