Keywords
1,2,4-oxadiazoles
aryl azides
aryl nitroso oxides
nitrile oxides
photooxidation
Abstract
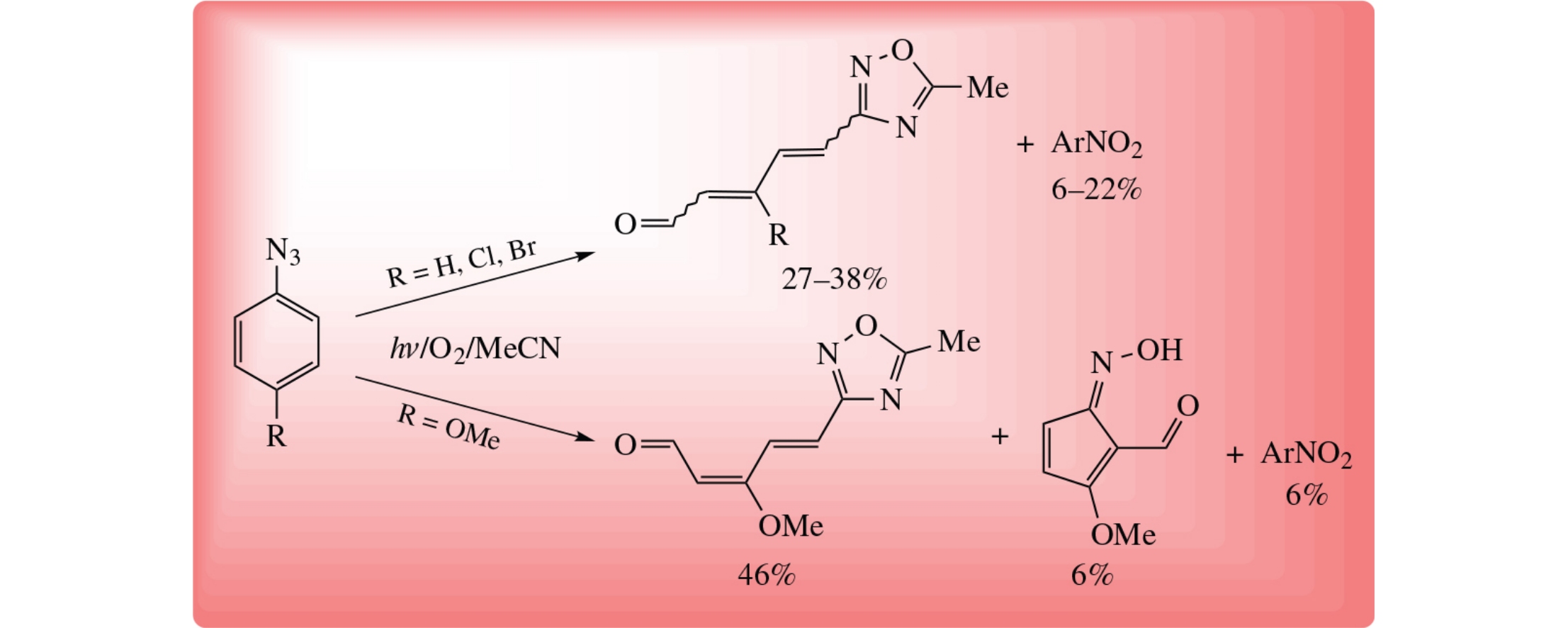
Photolysis of aryl azides at room temperature in acetonitrile in the presence of oxygen proceeds as arene ring opening and acetonitrile trapping to afford 5-methyl-3-(5-oxopenta-1,3-dien-1-yl)-1,2,4-oxadiazoles. In the case of 4-methoxyphenyl azide, a product depriving of acetonitrile reactant, 1-hydroxyimino-3-methylcyclopentadiene-2-carbaldehyde, is also formed.
References
1.

Ishiguro K., Sawaki Y.
Bulletin of the Chemical Society of Japan,
2000
2.

Gritsan N.P.
Russian Chemical Reviews,
2007
3.

Sawwan N., Greer A.
Chemical Reviews,
2007
4.
10.1016/j.mencom.2021.03.029_bib0020
Chainikova
2014
5.

Dequirez G., Pons V., Dauban P.
Angewandte Chemie,
2012
6.

Wentrup C.
Angewandte Chemie,
2018
7.
10.1016/j.mencom.2021.03.029_bib0035
Gritsan
J. Inf. Rec. Mater.,
1989
8.

Hiberty P.C., Ohanessian G.
Journal of the American Chemical Society,
1982
9.

Pritchina E.A., Gritsan N.P., Bally T.
Physical Chemistry Chemical Physics,
2006
10.

Pritchina E.A., Gritsan N.P.
Journal of Photochemistry and Photobiology A: Chemistry,
1988
11.

Chainikova E.M., Safiullin R.L., Spirikhin L.V., Abdullin M.F.
Journal of Physical Chemistry A,
2012
12.

Abramovitch R.A., Challand S.R.
Journal of the Chemical Society Chemical Communications,
1972
13.

Go C.L., Waddell W.H.
Journal of Organic Chemistry,
1983
14.

Safiullin R.L., Khursan S.L., Chainikova E.M., Danilov V.T.
Kinetics and Catalysis,
2004
15.

Harder T., Wessig P., Bendig J., Stösser R.
Journal of the American Chemical Society,
1999
16.

Chainikova E.M., Yusupova A.R., Khursan S.L., Teregulova A.N., Lobov A.N., Abdullin M.F., Enikeeva L.V., Gubaydullin I.M., Safiullin R.L.
Journal of Organic Chemistry,
2017
17.
10.1016/j.mencom.2021.03.029_bib0085
Jager
2002
18.

Annunziata R., Cinquini M., Cozzi F., Gennari C., Raimondi L.
Journal of Organic Chemistry,
1987
19.

Hassner A., Amarasekara A.S., Padwa A., Bullock W.H.
Tetrahedron Letters,
1988
20.

Chainikova E., Khursan S., Lobov A., Erastov A., Khalilov L., Mescheryakova E., Safiullin R.
Tetrahedron Letters,
2015
21.

Albini A., Bettinetti G., Minoli G.
Journal of Organic Chemistry,
1987
22.
![A simple one-pot preparation of 3,3a-dihydro-5H-pyrano[3,3a-c]isoxazol-5-ylideneethanal from 4-vinyloxyphenyl azide: an example of aromatic azide photooxidation for the synthesis of nitrogen-containing heterocyclic compounds](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chainikova E., Teregulova A., Lobov A., Erastov A., Safiullin R.
Tetrahedron Letters,
2015
23.

Chainikova E., Khursan S., Yusupova A., Lobov A., Abdullin M., Safiullin R.
Tetrahedron Letters,
2018
24.
10.1016/j.mencom.2021.03.029_bib0120
Hemming
Sci. Synth.,
2004
25.

Kuznetsov M.L., Kukushkin V.Y., Pombeiro A.J.
Dalton Transactions,
2008
26.
![The substituent effects on the [3+2] cycloaddition of nitrile oxides generated by photooxidation of arylazides to acetonitrile](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Yarullin A.R., Ovchinnikov M.Y., Khursan S.L., Chainikova E.M., Safiullin R.L.
Journal of Molecular Graphics and Modelling,
2020
27.

Stephenson L.M., Gemmer R.V., Brauman J.I.
Journal of the American Chemical Society,
1972
28.

Marley W.M., Jeffers P.M.
The Journal of Physical Chemistry,
1975