Keywords
antimicrobial action
antitumor activity
hybrids
pyrrolecarboxamides
silatranes
Abstract
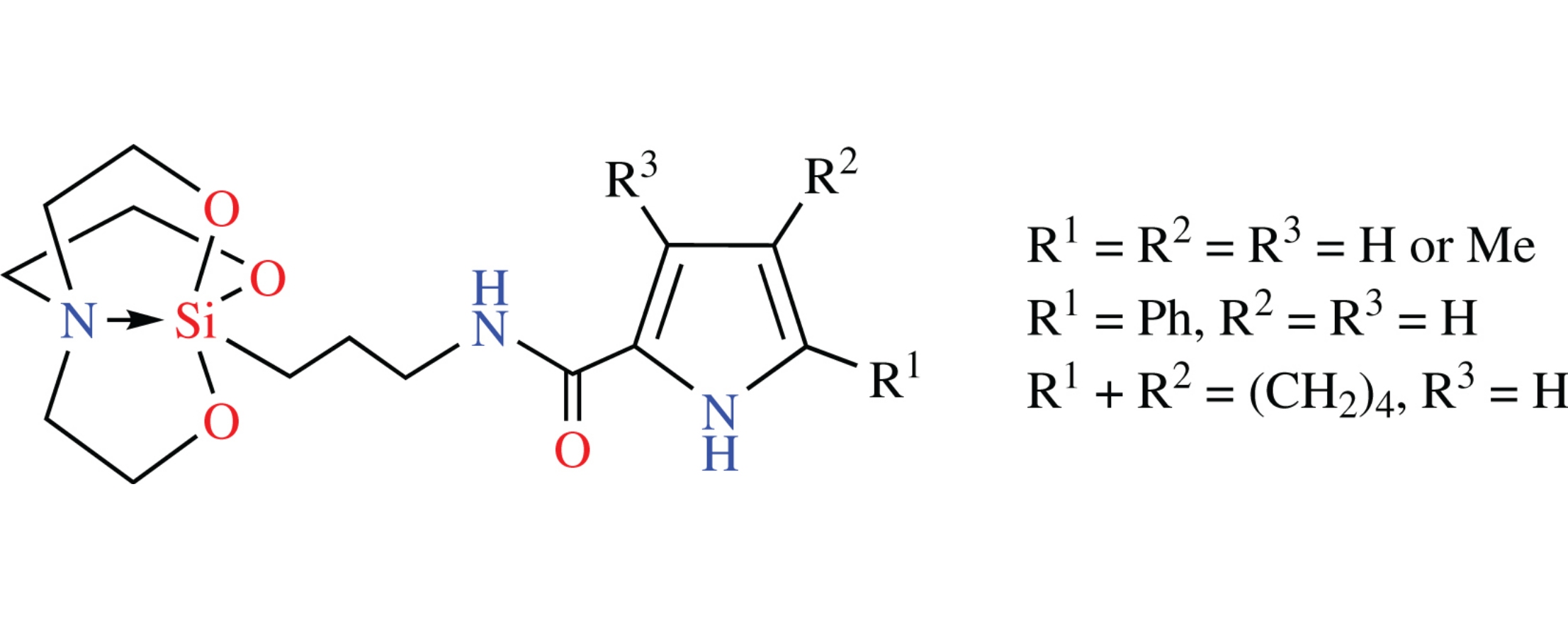
Silatrane-1H-pyrrole-2-carboxamide hybrids conjugated via propane-1,3-diyl linker have been synthesized by the reaction of 1-(3-aminopropyl)silatrane with 2-(trichloroacetyl)pyrroles. Their in silico screening would predict their antitumor activity. Their in vitro screening for antimicrobial activity revealed the representative with R1=R2=R3=H to be the most potent against Gram-positive microorganisms such as E. durans, B. subtilis and E. coli.
References
1.

Voronkov M.G., Dyakov V.M., Kirpichenko S.V.
Journal of Organometallic Chemistry,
1982
2.

Verkade J.G.
Coordination Chemistry Reviews,
1994
3.

Puri J.K., Singh R., Chahal V.K.
Chemical Society Reviews,
2011
4.
Belyaeva V.V., Belogolova E.F., Bolgova Y.I., Trofimova O.M.
Mendeleev Communications,
2015
5.
Alentiev D.A., Chapala P.P., Filatova M.P., Finkelshtein E.S., Bermeshev M.V.
Mendeleev Communications,
2016
6.

Voronkov M.G., Baryshok V.P.
Pharmaceutical Chemistry Journal,
2004
7.
M. G. Voronkov and V. P. Baryshok, Silatrany v meditsine i sel'skom khozyaistve (Silatranes in Medicine and Agriculture), SO RAN, Novosibirsk, 2005.(in Russian).
8.

Adamovich S.N.
Applied Organometallic Chemistry,
2019
9.

Adamovich S.N., Kondrashov E.V., Ushakov I.A., Shatokhina N.S., Oborina E.N., Vashchenko A.V., Belovezhets L.A., Rozentsveig I.B., Verpoort F.
Applied Organometallic Chemistry,
2020
10.

Boon J.M., Lambert T.N., Smith B.D., Beatty A.M., Ugrinova V., Brown S.N.
Journal of Organic Chemistry,
2002
11.

Materna K.L., Brennan B.J., Brudvig G.W.
Dalton Transactions,
2015
12.

Huang K., Hsieh C., Kan H., Hsieh M., Hsieh S., Chau L., Cheng T., Lin W.
Sensors and Actuators, B: Chemical,
2012
13.

Hsieh S., Chao W., Hsieh C.
Journal of Nanoscience and Nanotechnology,
2009
14.

15.

Singh G., Rani S., Arora A., Aulakh D., Wriedt M.
New Journal of Chemistry,
2016
16.

Aoyagi N., Endo T.
Tetrahedron,
2017
17.

Singh G., Saroa A., Girdhar S., Rani S., Sahoo S., Choquesillo-Lazarte D.
Inorganica Chimica Acta,
2015
18.

Han A., Li L., Qing K., Qi X., Hou L., Luo X., Shi S., Ye F.
Bioorganic and Medicinal Chemistry Letters,
2013
19.

Ye F., Song X., Liu J., Xu X., Wang Y., Hu L., Wang Y., Liang G., Guo P., Xie Z.
Chemical Biology and Drug Design,
2015
20.

Lyubchenko Y.L., Shlyakhtenko L.S.
Methods,
2009
21.
Adamovich S.N., Oborina E.N., Ushakov I.A.
Mendeleev Communications,
2019
22.

Huang C., Zheng Y.
Langmuir,
2018
23.

Gubala V., Giovannini G., Kunc F., Monopoli M.P., Moore C.J.
Cancer Nanotechnology,
2020
24.

Istratov V.V., Vasnev V.A.
Russian Chemical Bulletin,
2020
25.
10.1016/j.mencom.2021.03.019_sbrefe0035a
Trofimov
Chemistry of Pyrroles,
2014
26.

Bhardwaj V., Gumber D., Abbot V., Dhiman S., Sharma P.
RSC Advances,
2015
27.

Curreli F., Belov D.S., Kwon Y.D., Ramesh R., Furimsky A.M., O'Loughlin K., Byrge P.C., Iyer L.V., Mirsalis J.C., Kurkin A.V., Altieri A., Debnath A.K.
European Journal of Medicinal Chemistry,
2018
28.

Brasca M.G., Gnocchi P., Nesi M., Amboldi N., Avanzi N., Bertrand J., Bindi S., Canevari G., Casero D., Ciomei M., Colombo N., Cribioli S., Fachin G., Felder E.R., Galvani A., et. al.
Bioorganic and Medicinal Chemistry,
2015
29.

Rasal N.K., Sonawane R.B., Jagtap S.V.
Bioorganic Chemistry,
2020
30.
10.1016/j.mencom.2021.03.019_sbrefe0045c
Zhou
New J. Chem.,
2020
31.

Yao T., Xiao D., Li Z., Cheng J., Fang S., Du Y., Zhao J., Dong X., Zhu G.
Journal of Agricultural and Food Chemistry,
2017
32.

Bhosale J., Dabur R., Jadhav G., Bendre R.
Molecules,
2018
33.
Palomba M., Pompei S., Roscini L., Bagnoli L.
Arkivoc,
2020
34.

Li Y., Zhang P., Ma Q., Song H., Liu Y., Wang Q.
Bioorganic and Medicinal Chemistry Letters,
2012
35.

Mane Y.D., Surwase S.M., Biradar D.O., Sarnikar Y.P., Jawle B.H., Shinde V.S., Khade B.C.
Journal of Heterocyclic Chemistry,
2017
36.

Tomašič T., Mirt M., Barančoková M., Ilaš J., Zidar N., Tammela P., Kikelj D.
Bioorganic and Medicinal Chemistry,
2017
37.

Mane Y.D., Patil S.S., Biradar D.O., Khade B.C.
Heterocyclic Communications,
2018
38.

Barker D., Lee S., Varnava K.G., Sparrow K., van Rensburg M., Deed R.C., Cadelis M.M., Li S.A., Copp B.R., Sarojini V., Pilkington L.I.
Molecules,
2020
39.

Claudio Viegas-Junior, Eliezer J. Barreiro, Carlos Alberto Manssour Fraga
Current Medicinal Chemistry,
2007
40.

Corson T.W., Aberle N., Crews C.M.
ACS Chemical Biology,
2008
41.

Oliveira Pedrosa M., Duarte da Cruz R., Oliveira Viana J., de Moura R., Ishiki H., Barbosa Filho J., Diniz M., Scotti M., Scotti L., Bezerra Mendonca F.
Current Topics in Medicinal Chemistry,
2016
42.

Gontijo V.S., Viegas F.P., Ortiz C.J., de Freitas Silva M., Damasio C.M., Rosa M.C., Campos T.G., Couto D.S., Tranches Dias K.S., Viegas C.
Current Neuropharmacology,
2019
43.

Ivasiv V., Albertini C., Gonçalves A.E., Rossi M., Bolognesi M.L.
Current Topics in Medicinal Chemistry,
2019
44.

Shang H., Li L., Ma L., Tian Y., Jia H., Zhang T., Yu M., Zou Z.
Molecules,
2020
45.

Singh G., Saroa A., Girdhar S., Rani S., Choquesillo-Lazarte D., Sahoo S.C.
Applied Organometallic Chemistry,
2015
46.

Singh G., Arora A., Rani S., Kalra P., Aulakh D., Wriedt M.
Applied Organometallic Chemistry,
2017
47.

Singh G., Satija P., Singh A., Sanchita, Aulakh D., Wriedt M., Ruiz C.E., Esteban M.A., Sinha S., Sehgal R.
Applied Organometallic Chemistry,
2019
48.

Gao S., Bethel T.K., Kakeshpour T., Hubbell G.E., Jackson J.E., Tepe J.J.
Journal of Organic Chemistry,
2018
49.

Thoi V.S., Stork J.R., Niles E.T., Depperman E.C., Tierney D.L., Cohen S.M.
Inorganic Chemistry,
2008
50.

La Regina G., Silvestri R., Artico M., Lavecchia A., Novellino E., Befani O., Turini P., Agostinelli E.
Journal of Medicinal Chemistry,
2007
51.

Daneshrad A., Eaborn C., Walton D.R.
Journal of Organometallic Chemistry,
1975
52.
10.1016/j.mencom.2021.03.019_sbrefe0080b
Voronkov
Zh. Obshch. Khim.,
1986
53.

Lazareva N.F., Vakul'skaya T.I., Lazarev I.M.
Journal of Physical Organic Chemistry,
2009
54.

Maddila S., Gorle S., Sampath C., Lavanya P.
Journal of Saudi Chemical Society,
2016
55.
SwissADME, http://www.swissadme.ch.
56.

Lipinski C.A., Lombardo F., Dominy B.W., Feeney P.J.
Advanced Drug Delivery Reviews,
2001
57.

Veber D.F., Johnson S.R., Cheng H., Smith B.R., Ward K.W., Kopple K.D.
Journal of Medicinal Chemistry,
2002
58.
Prediction of Activity Spectra for Substances, http://www.pharmaexpert.ru/PASSOnline/.
59.

Poroikov V.V., Filimonov D.A., Gloriozova T.A., Lagunin A.A., Druzhilovskiy D.S., Rudik A.V., Stolbov L.A., Dmitriev A.V., Tarasova O.A., Ivanov S.M., Pogodin P.V.
Russian Chemical Bulletin,
2019
60.

Wiegand I., Hilpert K., Hancock R.E.
Nature Protocols,
2008