Keywords
closo-decaborate
alanine
amidines
amino acid esters
glycine
nitrilium derivatives
phenylalanine
Abstract
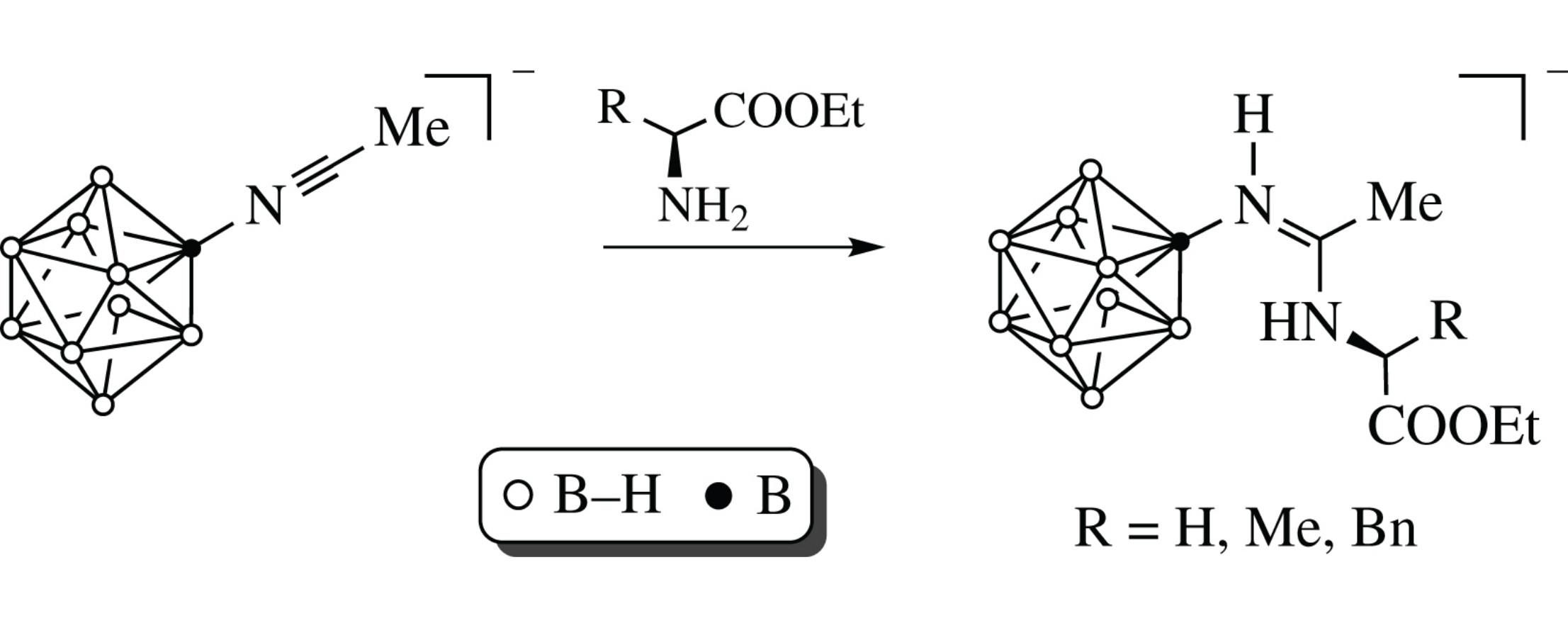
The addition of amino acid esters to [2-B10H9–N≡CMe]− Bu4N+ salt under mild conditions affords amidine closo-decaborates in high yields. The structure of compound (PPh4)[2-B10H9NH=C(Me)NHCH2COOEt] was elucidated by single-crystal XRD analysis.
References
1.

Snyder H.R., Reedy A.J., Lennarz W.J.
Journal of the American Chemical Society,
1958
2.

Li J., Shi Y., Zhang Z., Liu H., Lang L., Liu T., Chen X., Liu Z.
Bioconjugate Chemistry,
2019
3.

Lin Y., Chou F., Yang B., Chang C., Chen Y., Hwang J.
Annals of Nuclear Medicine,
2019
4.

Hung Y., Lin Y., Lin Y., Shih G., Liao J., Chen K., Liu H., Chen Y., Chuang Y., Yang C., Peir J., Yang C., Chou F.
Applied Radiation and Isotopes,
2020
5.

6.

Nakagawa F., Kawashima H., Morita T., Nakamura H.
Cells,
2020
7.

Lee J., Ueno M., Miyajima Y., Nakamura H.
Organic Letters,
2006
8.

Perry C.C., Ramos-Méndez J., Milligan J.R.
Radiation Physics and Chemistry,
2020
9.

Hu K., Yang Z., Zhang L., Xie L., Wang L., Xu H., Josephson L., Liang S.H., Zhang M.
Coordination Chemistry Reviews,
2020
10.

Nomoto T., Inoue Y., Yao Y., Suzuki M., Kanamori K., Takemoto H., Matsui M., Tomoda K., Nishiyama N.
Science advances,
2020
11.

Ali F., S Hosmane N., Zhu Y.
Molecules,
2020
12.
Sivaev I.B., Prikaznov A.V., Naoufal D.
Collection of Czechoslovak Chemical Communications,
2010
13.
10.1016/j.mencom.2021.03.018_bib0065
Boron-Based Compounds: Potential and Emerging Applications in Medicine,
2018
14.

Avdeeva V.V., Malinina E.A., Zhizhin K.Y., Kuznetsov N.T.
Russian Journal of Inorganic Chemistry,
2020
15.
10.1016/j.mencom.2021.03.018_bib0075
Abi-Ghaida
J. Nanomater.,
2015
16.

Wilbur D.S., Chyan M., Nakamae H., Chen Y., Hamlin D.K., Santos E.B., Kornblit B.T., Sandmaier B.M.
Bioconjugate Chemistry,
2012
17.

Li Y., Hamlin D.K., Chyan M., Wong R., Dorman E.F., Emery R.C., Woodle D.R., Manger R.L., Nartea M., Kenoyer A.L., Orozco J.J., Green D.J., Press O.W., Storb R., Sandmaier B.M., et. al.
PLoS ONE,
2018
18.
![Nucleophilic addition of alcohols to anionic [2-B10H9NCR]− (R = Et, t-Bu): An approach to producing new borylated imidates](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Zhdanov A.P., Klyukin I.N., Bykov A.Y., Grigoriev M.S., Zhizhin K.Y., Kuznetsov N.T.
Polyhedron,
2017
19.

Mindich A.L., Bokach N.A., Kuznetsov M.L., Starova G.L., Zhdanov A.P., Zhizhin K.Y., Miltsov S.A., Kuznetsov N.T., Kukushkin V.Y.
Organometallics,
2013
20.
![N-Borylated Hydroxylamines [B12H11NH2OH]– as a Novel Type of Substituted Derivative of the closo-Dodecaborate Anion](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Nelyubin A.V., Selivanov N.A., Bykov A.Y., Klyukin I.N., Novikov A.S., Zhdanov A.P., Zhizhin K.Y., Kuznetsov N.T.
Russian Journal of Inorganic Chemistry,
2020
21.

Losytskyy M.Y., Kovalska V.B., Varzatskii O.A., Kuperman M.V., Potocki S., Gumienna-Kontecka E., Zhdanov A.P., Yarmoluk S.M., Voloshin Y.Z., Zhizhin K.Y., Kuznetsov N.T., Elskaya A.V.
Journal of Luminescence,
2016
22.

Burianova V.K., Bolotin D.S., Novikov A.S., Kolesnikov I.E., Suslonov V.V., Zhdanov A.P., Zhizhin K.Y., Kuznetsov N.T.
Inorganica Chimica Acta,
2018
23.

Stogniy M.Y., Erokhina S.A., Sivaev I.B., Bregadze V.I.
Phosphorus, Sulfur and Silicon and the Related Elements,
2019
24.

Chlupatý T., Růžička A.
Coordination Chemistry Reviews,
2016
25.

Kretschmer R.
Chemistry - A European Journal,
2019
26.
![Reactions of nucleophilic addition of primary amines to the nitrilium derivative of the closo-decaborate anion [2-B10H9(N≡CCH3)]−](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Zhdanov A.P., Polyakova I.N., Razgonyaeva G.A., Zhizhin K.Y., Kuznetsov N.T.
Russian Journal of Inorganic Chemistry,
2011
27.
10.1016/j.mencom.2021.03.018_bib0135
Rattle
1981
28.
![Crystal Structure of Bis(triethylammonium)closo-decahydrodecaborate, [(C2H5)3NH]2[B10H10]](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Hofmann K., Albert B.
Zeitschrift fur Naturforschung - Section B Journal of Chemical Sciences,
2000
29.
![Complex [Ag(PPh3)4][2-B10H9NH3 · 2DMF]: Synthesis and Structure](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Zhdanov A.P., Voinova V.V., Klyukin I.N., Buzanov G.A., Grigor’ev M.S., Zhizhin K.Y., Kuznetsov N.T.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2019
30.

Allen F.H., Kennard O., Watson D.G., Brammer L., Orpen A.G., Taylor R.
Journal of the Chemical Society Perkin Transactions 2,
1987
31.
10.1016/j.mencom.2021.03.018_bib0160
Bakhmutov
Dihydrogen Bonds: Principles, Experiments, and Applications,
2008
32.

Belkova N.V., Epstein L.M., Filippov O.A., Shubina E.S.
Chemical Reviews,
2016
33.

Bondi A.
The Journal of Physical Chemistry,
1964
34.

Schulz F., Sumerin V., Heikkinen S., Pedersen B., Wang C., Atsumi M., Leskelä M., Repo T., Pyykkö P., Petry W., Rieger B.
Journal of the American Chemical Society,
2011
35.

Biallas P., Kirsch S.F.
Tetrahedron Letters,
2017
36.
Khrenova M.G., Nemukhin A.V., Tsirelson V.G.
Mendeleev Communications,
2020
37.
Vatsadze S.Z., Medved’ko A.V., Bodunov A.A., Lyssenko K.A.
Mendeleev Communications,
2020
38.
Klyukin I.N., Novikov A.S., Zhdanov A.P., Zhizhin K.Y., Kuznetsov N.T.
Mendeleev Communications,
2020
39.
APEX2, version 2.1-0, Bruker AXS, Madison, WI, 2006.
40.
SADABS, version 2004/1, Bruker AXS, Madison, WI, 2004.
41.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
42.
10.1016/j.mencom.2021.03.018_bib0215
Sheldrick
Acta Crystallogr.,
2015
43.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009