Keywords
atom–atom potentials method
benzene
benzotrifuroxan
bimolecular complex
cocrystal
complexation energy
crystal structure
DFT
global optimization
potential energy surface
Abstract
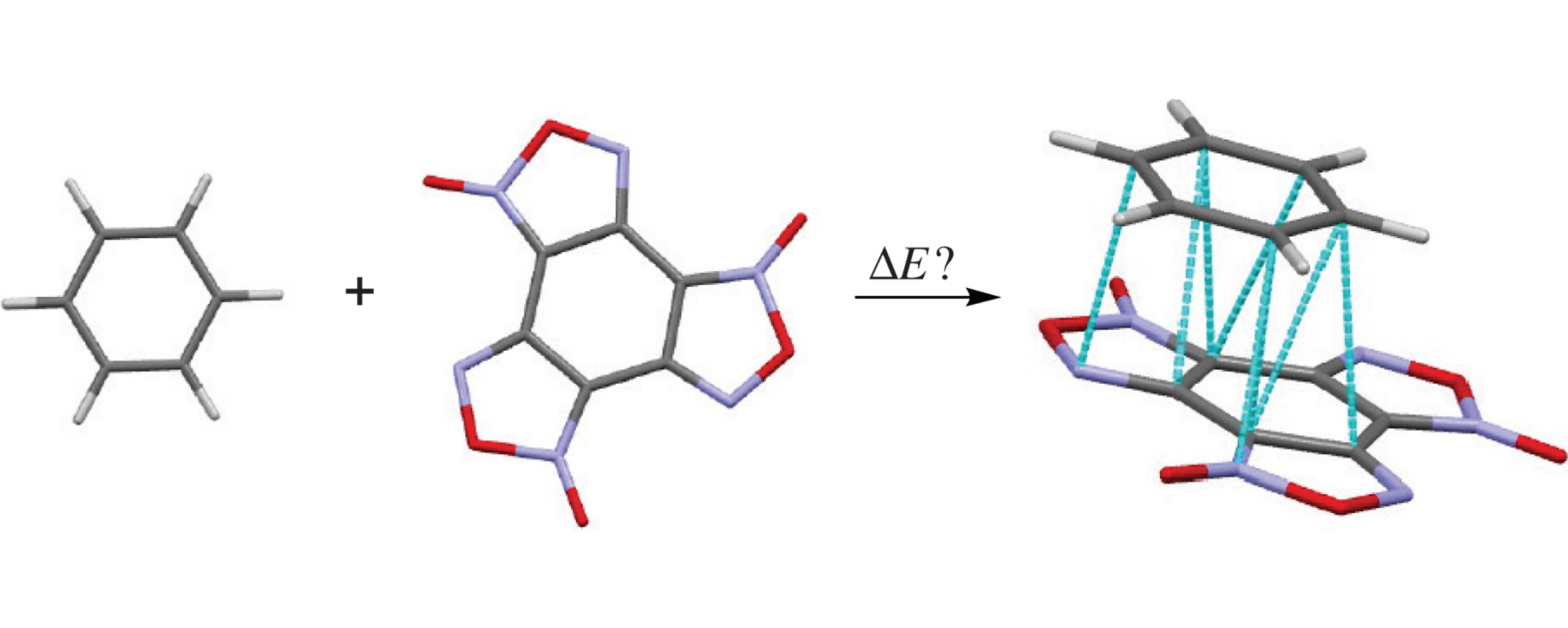
Based on a combination of quantum chemistry and atom–atom potentials methods, we developed a technique for modeling the structure and estimating the complexation energy of a binary organic complex in gas and crystal phases. The efficiency of this technique was illustrated by an example of the benzotrifuroxan–benzene molecular complex. For both phases, the same molecular binary structure (π-stacking) was obtained with a parallel arrangement of the planes of BTF and benzene molecules and complexation energies of −11.9 and −11.7kcalmol−1 for a gas phase and a crystal cluster, respectively.
References
1.

Newman A., Wenslow R.
AAPS Open,
2016
2.

TRASK A., MOTHERWELL W., JONES W.
International Journal of Pharmaceutics,
2006
3.

Zhang H., Guo C., Wang X., Xu J., He X., Liu Y., Liu X., Huang H., Sun J.
Crystal Growth and Design,
2013
4.

Tan Y., Yang Z., Wang H., Li H., Nie F., Liu Y., Yu Y.
Crystal Growth and Design,
2019
5.

Bennion J.C., McBain A., Son S.F., Matzger A.J.
Crystal Growth and Design,
2015
6.

Zhang J., Shreeve J.M.
CrystEngComm,
2016
7.

Hartke B.
Wiley Interdisciplinary Reviews: Computational Molecular Science,
2011
8.

Wales D.J., Hodges M.P.
Chemical Physics Letters,
1998
9.
10.1016/j.mencom.2021.03.017_bib0045
Marques
Philos. Trans. R. Soc., A,
2017
10.

Modeling Coronene Nanostructures: Analytical Potential, Stable Configurations and Ab Initio Energies
Bartolomei M., Pirani F., Marques J.M.
Journal of Physical Chemistry C,
2017
11.

Takeuchi H.
Journal of Chemical Information and Modeling,
2006
12.

Cheung A., Adjiman C.S., Kolar P., Ishikawa T.
Fluid Phase Equilibria,
2002
13.
10.1016/j.mencom.2021.03.017_bib0065
Pertsin
J. Comput. Chem.,
1987
14.

Boeyens J.C., Herbstein F.H.
The Journal of Physical Chemistry,
1965
15.

Dzyabchenko A.V.
Russian Journal of Physical Chemistry A,
2008
16.
10.1016/j.mencom.2021.03.017_bib0080
Frisch
Revision D. 01,
2009
17.
CPOSS Website, The FIT and WILL01 Empirical ‘Repulsion-Dispersion’ Potentials, 2020, http://www.chem.ucl.ac.uk/cposs/dmacrys/fit_and_will01_empirical_potentials.htm. (accessed October 2020).
18.

Dzyabchenko A.V.
Russian Journal of Physical Chemistry A,
2008
19.

Dzyabchenko A.V.
Acta Crystallographica Section A Foundations of Crystallography,
1983
20.
10.1016/j.mencom.2021.03.017_bib0100
Fletcher
FORTRAN Subroutines for Minimization by Quasi-Newton Methods,
1972
21.

Grimme S.
Wiley Interdisciplinary Reviews: Computational Molecular Science,
2011
22.

Bludský O., Rubeš M., Soldán P., Nachtigall P.
Journal of Chemical Physics,
2008
23.

Ewald P.P.
Annalen der Physik,
1921
24.

Belsky V.K., Zorkaya O.N., Zorky P.M.
Acta Crystallographica Section A Foundations of Crystallography,
1995
25.

Cruz Cabeza A.J., Pidcock E., Day G.M., Motherwell W.D., Jones W.
CrystEngComm,
2007
26.

Khakimov D.V., Dzyabchenko A.V., Pivina T.S.
Russian Chemical Bulletin,
2020
27.
![Crystal Structure Prediction of Bifurazano[3,4‐b:3′,4′‐f][3′′,4′′‐d](BFFO) in the Experimentally Known Monohydrated and Proposed Anhydrous Forms](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Khakimov D.V., Dzyabchenko A.V., Pivina T.S.
Propellants, Explosives, Pyrotechnics,
2019