Keywords
azo dyes
crystal engineering
DFT
halogen bonding
non-covalent interactions
QTAIM
Abstract
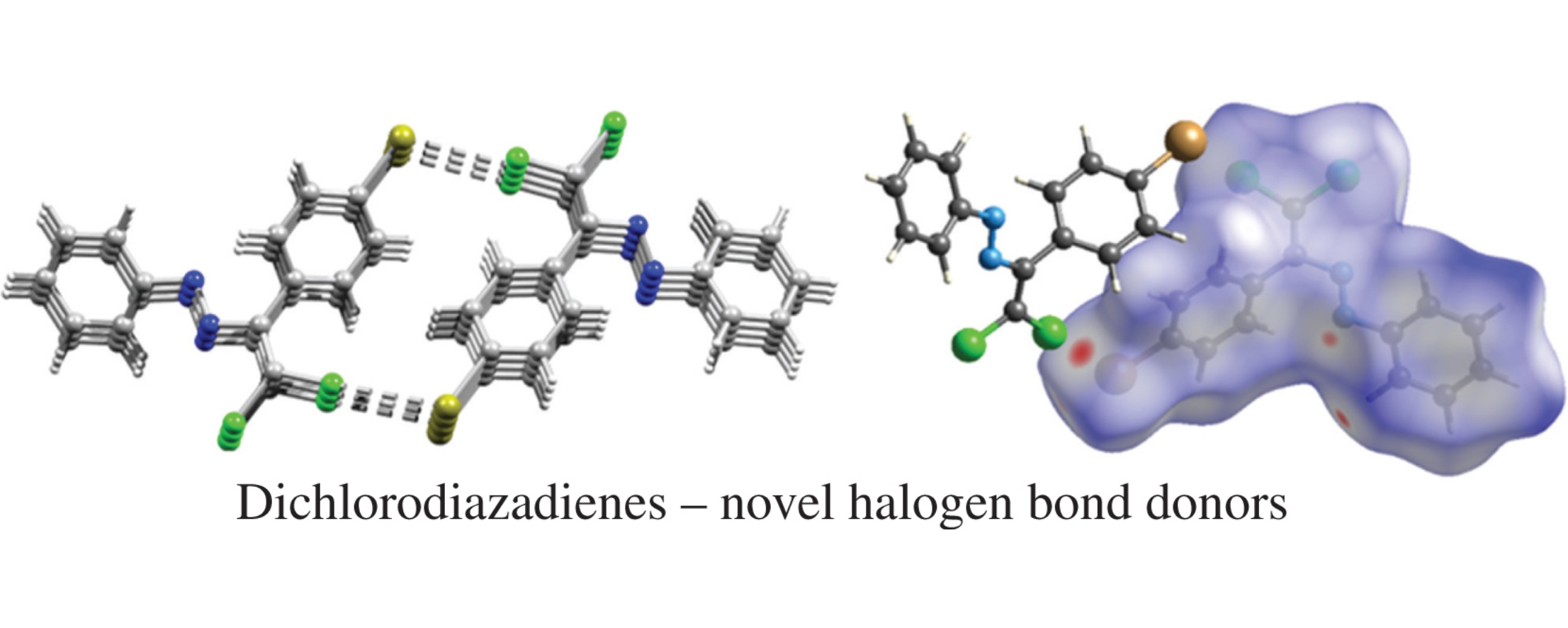
1-Aryl-3-(4-bromophenyl)-4,4-dichloro-1,2-diazabuta-1,3- dienes were synthesized via copper-catalyzed reaction of 4-bromobenzaldehyde arylhydrazones with CCl4. X-ray diffraction analysis revealed non-covalent Cl···Br interactions in the crystalline state and formation of 3D supramolecular framework; DFT calculations and topological analysis of the electron density distribution within the framework of Bader's theory (QTAIM method) confirmed these observations. Theoretical calculations showed that highly polarizable dichlorodiazadiene unit is capable of acting as a relatively strong halogen bond donor.
References
1.

Bertani R., Sgarbossa P., Venzo A., Lelj F., Amati M., Resnati G., Pilati T., Metrangolo P., Terraneo G.
Coordination Chemistry Reviews,
2010
2.

Cook T.R., Stang P.J.
Chemical Reviews,
2015
3.

Tskhovrebov A.G., Vasileva A.A., Goddard R., Riedel T., Dyson P.J., Mikhaylov V.N., Serebryanskaya T.V., Sorokoumov V.N., Haukka M.
Inorganic Chemistry,
2018
4.
Torubaev Y.V., Skabitsky I.V., Lyssenko K.A.
Mendeleev Communications,
2020
5.
Nelyubina Y.V., Antipin M.Y., Lyssenko K.A.
Mendeleev Communications,
2011
6.

Saha B.K., Rather S.A., Saha A.
Crystal Growth and Design,
2016
7.

Nenajdenko V.G., Shastin A.V., Gorbachev V.M., Shorunov S.V., Muzalevskiy V.M., Lukianova A.I., Dorovatovskii P.V., Khrustalev V.N.
ACS Catalysis,
2016
8.
Tsyrenova B.D., Tarasevich B.N., Khrustalev V.N., Gloriozov I.P., Nenajdenko V.G.
Mendeleev Communications,
2020
9.

Repina O.V., Novikov A.S., Khoroshilova O.V., Kritchenkov A.S., Vasin A.A., Tskhovrebov A.G.
Inorganica Chimica Acta,
2020
10.

Tskhovrebov A.G., Novikov A.S., Odintsova O.V., Mikhaylov V.N., Sorokoumov V.N., Serebryanskaya T.V., Starova G.L.
Journal of Organometallic Chemistry,
2019
11.

Mikhaylov V., Sorokoumov V., Liakhov D., Tskhovrebov A., Balova I.
Catalysts,
2018
12.

Tskhovrebov A.G., Luzyanin K.V., Kuznetsov M.L., Sorokoumov V.N., Balova I.A., Haukka M., Kukushkin V.Y.
Organometallics,
2011
13.

Mikhaylov V.N., Sorokoumov V.N., Novikov A.S., Melnik M.V., Tskhovrebov A.G., Balova I.A.
Journal of Organometallic Chemistry,
2020
14.

Tskhovrebov A.G., Luzyanin K.V., Haukka M., Kukushkin V.Y.
Journal of Chemical Crystallography,
2012
15.

Tskhovrebov A.G., Novikov A.S., Kritchenkov A.S., Khrustalev V.N., Haukka M.
Zeitschrift fur Kristallographie - Crystalline Materials,
2020
16.

Nenajdenko V.G., Shikhaliyev N.G., Maharramov A.M., Bagirova K.N., Suleymanova G.T., Novikov A.S., Khrustalev V.N., Tskhovrebov A.G.
Molecules,
2020
17.

Tskhovrebov A.G., Lingnau J.B., Fürstner A.
Angewandte Chemie - International Edition,
2019
18.

Tskhovrebov A.G., Solari E., Scopelliti R., Severin K.
Organometallics,
2014
19.

Liu Y., Varava P., Fabrizio A., Eymann L.Y., Tskhovrebov A.G., Planes O.M., Solari E., Fadaei-Tirani F., Scopelliti R., Sienkiewicz A., Corminboeuf C., Severin K.
Chemical Science,
2019
20.

Shikhaliyev N.Q., Kuznetsov M.L., Maharramov A.M., Gurbanov A.V., Ahmadova N.E., Nenajdenko V.G., Mahmudov K.T., Pombeiro A.J.
CrystEngComm,
2019
21.

Shikhaliyev N.Q., Ahmadova N.E., Gurbanov A.V., Maharramov A.M., Mammadova G.Z., Nenajdenko V.G., Zubkov F.I., Mahmudov K.T., Pombeiro A.J.
Dyes and Pigments,
2018
22.

Maharramov A.M., Shikhaliyev N.Q., Suleymanova G.T., Gurbanov A.V., Babayeva G.V., Mammadova G.Z., Zubkov F.I., Nenajdenko V.G., Mahmudov K.T., Pombeiro A.J.
Dyes and Pigments,
2018
23.
T. V. Serebryanskaya, A. S. Lyakhov, L. S. Ivashkevich, Y. V. Grigoriev, A. S. Kritchenkov, V. N. Khrustalev, A. G. Tskhovrebov and O. A. Ivashkevich, Z. Kristallogr. Cryst. Mater., 000010151520200082.
24.

Bader R.F.
Chemical Reviews,
1991
25.

Usoltsev A.N., Adonin S.A., Novikov A.S., Samsonenko D.G., Sokolov M.N., Fedin V.P.
CrystEngComm,
2017
26.

Espinosa E., Alkorta I., Elguero J., Molins E.
Journal of Chemical Physics,
2002
27.

Johnson E.R., Keinan S., Mori-Sánchez P., Contreras-García J., Cohen A.J., Yang W.
Journal of the American Chemical Society,
2010
28.

Contreras-García J., Johnson E.R., Keinan S., Chaudret R., Piquemal J., Beratan D.N., Yang W.
Journal of Chemical Theory and Computation,
2011
29.
10.1016/j.mencom.2021.03.015_bib0145
Sheldrick
Acta Crystallogr.,
2015
30.
10.1016/j.mencom.2021.03.015_bib0150
Sheldrick
Acta Crystallogr.,
2015