Keywords
3-amino-1,2,4-triazole
catalysis
N-heterocyclic carbenes
Palladium complexes
postmodification
Abstract
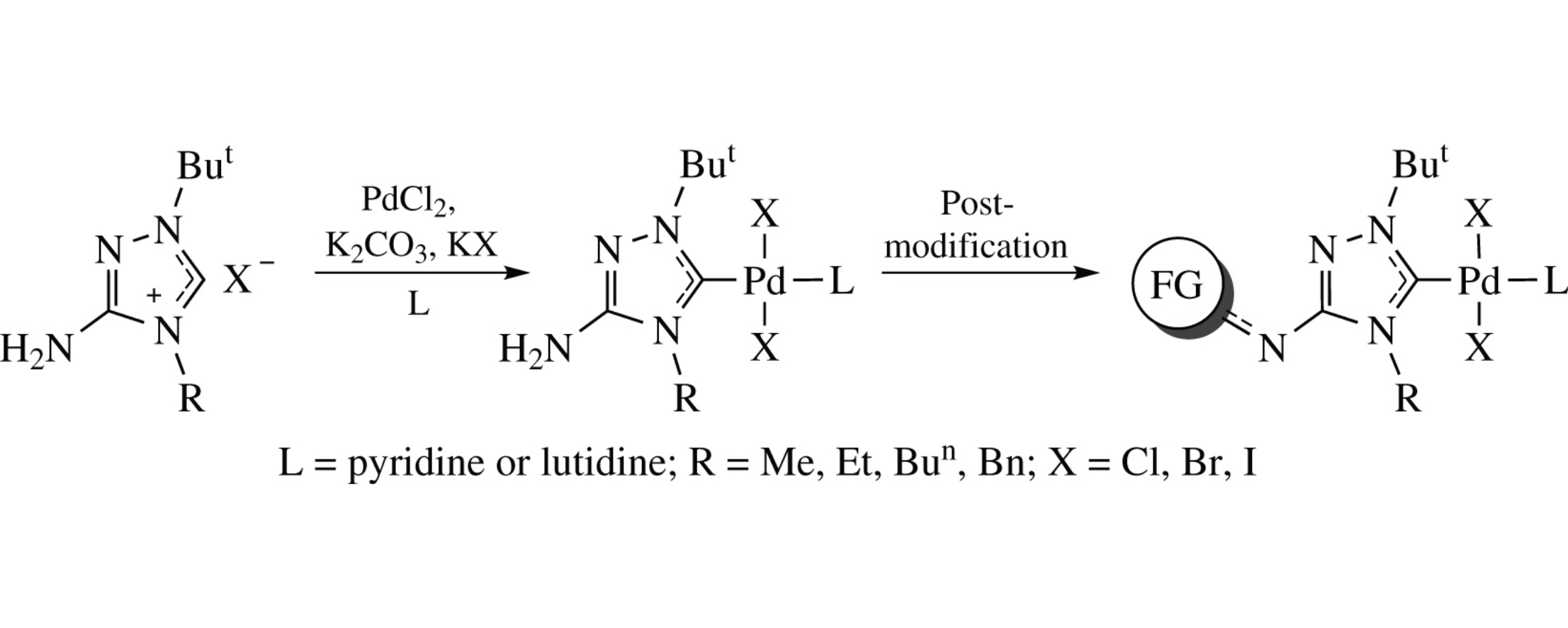
Palladium complexes with N-heterocyclic carbene ligands of a new type containing free NH2 group were obtained by direct palladation of 3-amino-1,4-dialkyl-1,2,4-triazolium salts. These amino-functionalized complexes were found to be a versatile platform for postmodification via reactions of the NH2 group with acyl and sulfonyl chlorides.
References
1.

Froese R.D., Lombardi C., Pompeo M., Rucker R.P., Organ M.G.
Accounts of Chemical Research,
2017
2.

Zhao Q., Meng G., Nolan S.P., Szostak M.
Chemical Reviews,
2020
3.

Hopkinson M.N., Richter C., Schedler M., Glorius F.
Nature,
2014
4.

Chernyshev V.M., Denisova E.A., Eremin D.B., Ananikov V.P.
Chemical Science,
2020
5.

Zou T., Lok C., Wan P., Zhang Z., Fung S., Che C.
Current Opinion in Chemical Biology,
2018
6.

Hussaini S.Y., Haque R.A., Razali M.R.
Journal of Organometallic Chemistry,
2019
7.

Khazipov O.V., Shevchenko M.A., Pasyukov D.V., Chernenko A.Y., Astakhov A.V., Tafeenko V.A., Chernyshev V.M., Ananikov V.P.
Catalysis Science and Technology,
2020
8.

Fortman G.C., Nolan S.P.
Chemical Society Reviews,
2011
9.

Cisnetti F., Gibard C., Gautier A.
Journal of Organometallic Chemistry,
2015
10.

Peris E.
Chemical Reviews,
2017
11.

Bonnet L.G., Douthwaite R.E., Hodgson R., Houghton J., Kariuki B.M., Simonovic S.
Dalton Transactions,
2004
12.

O W.W., Lough A.J., Morris R.H.
Chemical Communications,
2010
13.

Cross W.B., Daly C.G., Boutadla Y., Singh K.
Dalton Transactions,
2011
14.

15.
![Primary amino-functionalized N-heterocyclic carbene ligands as support for Au(i)⋯Au(i) interactions: structural, electrochemical, spectroscopic and computational studies of the dinuclear [Au2(NH2(CH2)2imMe)2][NO3]2](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Ballarin B., Busetto L., Cassani M.C., Femoni C., Ferrari A.M., Miletto I., Caputo G.
Dalton Transactions,
2012
16.

O W.W., Lough A.J., Morris R.H.
Organometallics,
2012
17.

Ohara H., O W.W., Lough A.J., Morris R.H.
Dalton Transactions,
2012
18.

Jansen E., Jongbloed L.S., Tromp D.S., Lutz M., de Bruin B., Elsevier C.J.
ChemSusChem,
2013
19.

O W.W., Lough A.J., Morris R.H.
Organometallics,
2013
20.

Zhuang R., Lin W., Zhuang R.R., Hwang W.
Polyhedron,
2013
21.

Ramsay W.J., Foster J.A., Moore K.L., Ronson T.K., Mirgalet R.J., Jefferson D.A., Nitschke J.R.
Chemical Science,
2015
22.

Wan K.Y., Lough A.J., Morris R.H.
Organometallics,
2016
23.

Mahanti B., González Miera G., Martínez-Castro E., Bedin M., Martín-Matute B., Ott S., Thapper A.
ChemSusChem,
2017
24.

Takahashi H., Watanabe T., Tobita H.
Chemistry Letters,
2018
25.

Wan K.Y., Sung M.M., Lough A.J., Morris R.H.
ACS Catalysis,
2017
26.
27.

DeJesus J.F., Sherman L.M., Yohannan D.J., Becca J.C., Strausser S.L., Karger L.F., Jensen L., Jenkins D.M., Camden J.P.
Angewandte Chemie - International Edition,
2020
28.

Chernyshev V.M., Vlasova A.G., Astakhov A.V., Shishkina S.V., Shishkin O.V.
Journal of Organic Chemistry,
2014
29.

Voitekhovich S.V., Lyakhov A.S., Ivashkevich L.S., Matulis V.E., Grigoriev Y.V., Gaponik P.N., Ivashkevich O.A.
Tetrahedron,
2012
30.

Astakhov A.V., Khazipov O.V., Chernenko A.Y., Pasyukov D.V., Kashin A.S., Gordeev E.G., Khrustalev V.N., Chernyshev V.M., Ananikov V.P.
Organometallics,
2017
31.

Chernenko A.Y., Astakhov A.V., Pasyukov D.V., Dorovatovskii P.V., Zubavichus Y.V., Khrustalev V.N., Chernyshev V.M.
Russian Chemical Bulletin,
2018
32.

Kumar A., Gangwar M.K., Prakasham A.P., Mhatre D., Kalita A.C., Ghosh P.
Inorganic Chemistry,
2016
33.

Du Y., Liang B., Yang F., Shi Y., Li X., Pang G., Cao C.
Transition Metal Chemistry,
2017
34.

Dash C., Shaikh M.M., Ghosh P.
European Journal of Inorganic Chemistry,
2009
35.

Chardon E., Dahm G., Guichard G., Bellemin-Laponnaz S.
Inorganica Chimica Acta,
2017
36.

Mondal M., Choudhury J.
Journal of Molecular Catalysis A Chemical,
2017
37.

Erdemir F., Barut Celepci D., Aktaş A., Gök Y.
ChemistrySelect,
2019
38.
![Restricting shuttling in bis(imidazolium)…pillar[5]arene rotaxanes using metal coordination](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Langer P., Yang L., Pfeiffer C.R., Lewis W., Champness N.R.
Dalton Transactions,
2019
39.
10.1016/j.mencom.2021.03.010_bib0195
Evans
Acta Crystallogr.,
2006
40.
10.1016/j.mencom.2021.03.010_bib0200
Battye
Acta Crystallogr.,
2011
41.
10.1016/j.mencom.2021.03.010_bib0205
Sheldrick
Acta Crystallogr.,
2015