Keywords
clusters
group 12 elements
molecular precursors
N-heterocyclic carbenes
silyl sulfides
zinc complexes
Abstract
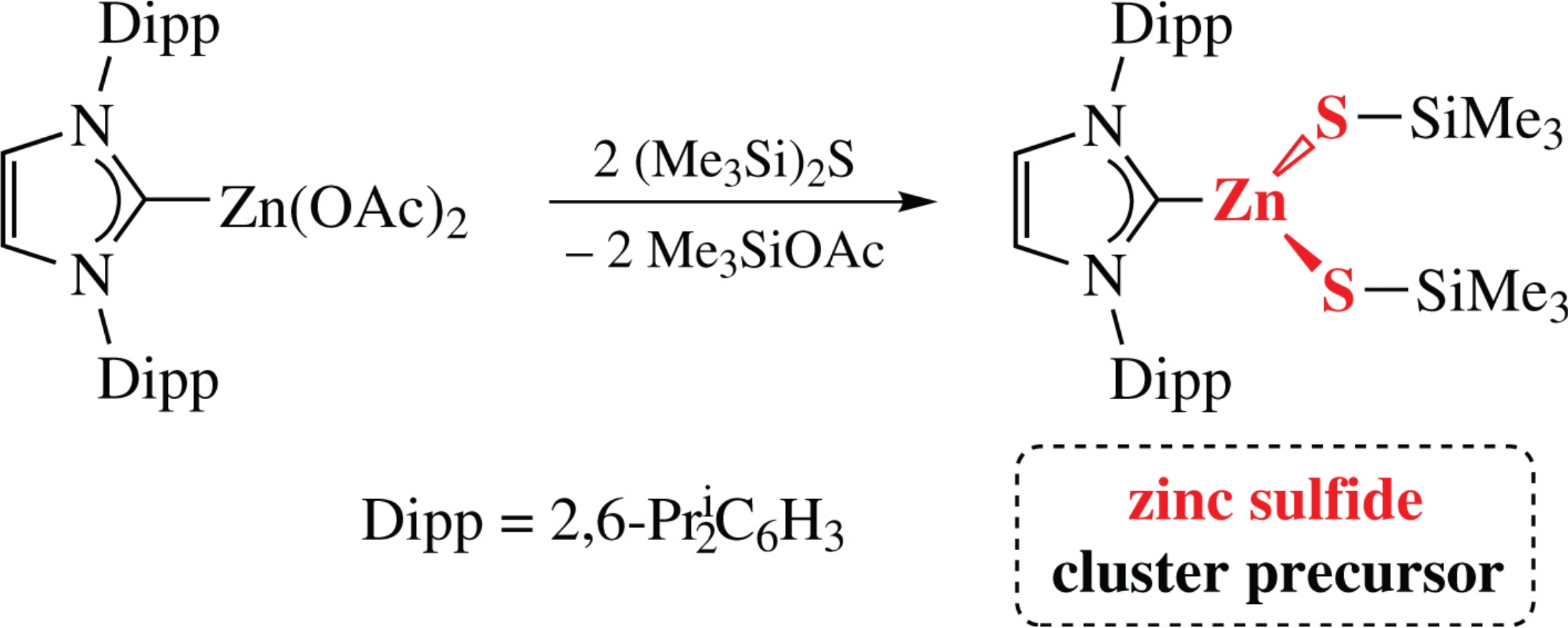
A series of N-heterocyclic carbene (NHC) supported group 12 acetate complexes were prepared and their use as building blocks toward soluble group 12 sulfide precursors was explored. The highly moisture-sensitive NHC-ligated zinc dithiolato complex [(IPr)Zn(SSiMe3)2] was also synthesized [IPr=1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene], and its partial hydrolysis gave the dianionic zinc sulfide cluster [Zn3(μ3-S)(μ2-SSiMe3)3(SSiMe3)3]2−, which contained a rare example of a Zn3S structural motif. This preliminary observation indicates that the future growth of related metallosulfide clusters from [(IPr)Zn(SSiMe3)2] should be possible.
References
1.
10.1016/j.mencom.2021.03.009_bib0005
Cotton
Advanced Inorganic Chemistry,
1988
2.

Dabbousi B.O., Rodriguez-Viejo J., Mikulec F.V., Heine J.R., Mattoussi H., Ober R., Jensen K.F., Bawendi M.G.
Journal of Physical Chemistry B,
1997
3.

Ghosh Chaudhuri R., Paria S.
Chemical Reviews,
2011
4.

Crawford N.F., Leblanc R.M.
Coordination Chemistry Reviews,
2014
5.

Tiwari A., Dhoble S.J.
RSC Advances,
2016
6.

Mongin C., Garakyaraghi S., Razgoniaeva N., Zamkov M., Castellano F.N.
Science,
2016
7.
10.1016/j.mencom.2021.03.009_sbref0015a
Sidot
Compt. Rend.,
1866
8.

Getz W.A., Wentzel D.A., Palmer M.J., Campbell D.J.
Journal of Chemical Education,
2017
9.

Thimer K.C., Al-Rafia S.M., Ferguson M.J., McDonald R., Rivard E.
Chemical Communications,
2009
10.

Purkait T.K., Swarnakar A.K., De Los Reyes G.B., Hegmann F.A., Rivard E., Veinot J.G.
Nanoscale,
2015
11.

Sinclair J., Dai G., McDonald R., Ferguson M.J., Brown A., Rivard E.
Inorganic Chemistry,
2020
12.

Omaña A.A., Green R.K., Kobayashi R., He Y., Antoniuk E.R., Ferguson M.J., Zhou Y., Veinot J.G., Iwamoto T., Brown A., Rivard E.
Angewandte Chemie - International Edition,
2020
13.
![Three-coordinate thiolato complexes of zinc: solution and solid-state structures and EHMO analysis of the bonding pattern of [Zn(S-tert-Bu3C6H2-2,4,6)2]2](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Bochmann M., Bwembya G., Grinter R., Lu J., Webb K.J., Williamson D.J., Hursthouse M.B., Mazid M.
Inorganic Chemistry,
1993
14.
![Mercury(II) and methylmercury(II) complexes of novel sterically hindered thiolates: carbon-13 and mercury-199 NMR studies and the crystal and molecular structures of [MeHg(SC6H2-2,4,6-Pr-iso3)], [Hg(SC6H4-2-SiMe3)2], [Hg(2-SC5H3N-3-SiMe3)2], and [Hg{(2-SC6H4)2SiMe2}]2](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Block E., Brito M., Gernon M., McGowty D., Kang H., Zubieta J.
Inorganic Chemistry,
1990
15.

Ellison J.J., Power P.P.
Inorganic Chemistry,
1994
16.
Power P.P., Shoner S.C.
1990
17.
![Solid-state mercury-199 and cadmium-113 NMR studies of mercury- and cadmium-thiolate complexes. Spectroscopic models for [Hg(SCys)n] centers in the bacterial mercury resistance proteins](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Santos R.A., Gruff E.S., Koch S.A., Harbison G.S.
Journal of the American Chemical Society,
1991
18.
![Sterically hindered chalcogenolato complexes. Mono- and di-meric thiolates and selenolates of zinc and cadmium; structure of [{Cd(SeC6H2But3-2,4,6)2}2], the first three-co-ordinate cadmium–selenium complex](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Bochmann M., Webb K.J., Hursthouse M.B., Mazid M.
Journal of the Chemical Society Dalton Transactions,
1991
19.

Bochmann M., Webb K.J.
Journal of the Chemical Society Dalton Transactions,
1991
20.
![Trigonal-planar [M(SR)3]1- complexes of cadmium and mercury. Structural similarities between mercury-cysteine and cadmium-cysteine coordination centers](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Gruff E.S., Koch S.A.
Journal of the American Chemical Society,
1990
21.

Varonka M.S., Warren T.H.
Inorganica Chimica Acta,
2007
22.

Grützmacher H., Steiner M., Pritzkow H., Zsolnai L., Huttner G., Sebald A.
Chemische Berichte,
1992
23.

Polgar A.M., Weigend F., Zhang A., Stillman M.J., Corrigan J.F.
Journal of the American Chemical Society,
2017
24.

Rit A., Zanardi A., Spaniol T.P., Maron L., Okuda J.
Angewandte Chemie - International Edition,
2014
25.

Wang D., Wurst K., Buchmeiser M.R.
Journal of Organometallic Chemistry,
2004
26.

Ibrahim Al-Rafia S.M., Lummis P.A., Swarnakar A.K., Deutsch K.C., Ferguson M.J., McDonald R., Rivard E.
Australian Journal of Chemistry,
2013
27.

Taher D., Wallbank A.I., Turner E.A., Cuthbert H.L., Corrigan J.F.
European Journal of Inorganic Chemistry,
2006
28.

Rit A., Spaniol T.P., Maron L., Okuda J.
Angewandte Chemie - International Edition,
2013
29.

Lummis P.A., Momeni M.R., Lui M.W., McDonald R., Ferguson M.J., Miskolzie M., Brown A., Rivard E.
Angewandte Chemie - International Edition,
2014
30.

Al-Rafia S.M., Malcolm A.C., Liew S.K., Ferguson M.J., Rivard E.
Journal of the American Chemical Society,
2010
31.

DeGroot M.W., Corrigan J.F.
Organometallics,
2005
32.
Yambulatov D.S., Petrov P.A., Nelyubina Y.V., Starikova A.A., Pavlov A.A., Aleshin D.Y., Nikolaevskii S.A., Kiskin M.A., Sokolov M.N., Eremenko I.L.
Mendeleev Communications,
2020
33.

Roy M.M., Ferguson M.J., Rivard E.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2016
34.

Lee K., Huang Y., Corrigan J.F.
Chemical Communications,
2019
35.
![Homoleptic trimethylsilylchalcogenolato zincates [Zn(ESiMe3)3]− and stannanides [Sn(ESiMe3)3]− (E = S, Se): precursors in solution-based low-temperature binary metal chalcogenide and Cu2ZnSnS4 (CZTS) synthesis](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Guschlbauer J., Vollgraff T., Sundermeyer J.
Dalton Transactions,
2020
36.
10.1016/j.mencom.2021.03.009_bib0085
Sheldrick
Acta Crystallogr.,
2015
37.
10.1016/j.mencom.2021.03.009_bib0090
Sheldrick
Acta Crystallogr.,
2015
38.

Bergerhoff G., Berndt M., Brandenburg K.
Journal of Research of the National Institute of Standards and Technology,
1996
39.

Roy M.M., Ferguson M.J., McDonald R., Rivard E.
Chemistry - A European Journal,
2016
40.

Chernyshev V.M., Denisova E.A., Eremin D.B., Ananikov V.P.
Chemical Science,
2020