Keywords
bismuth–iodine polyhedron
bond energy
bond length
Cambridge structural database
density functional theory
intermolecular interactions
iodobismuthates
net energy conservation
Abstract
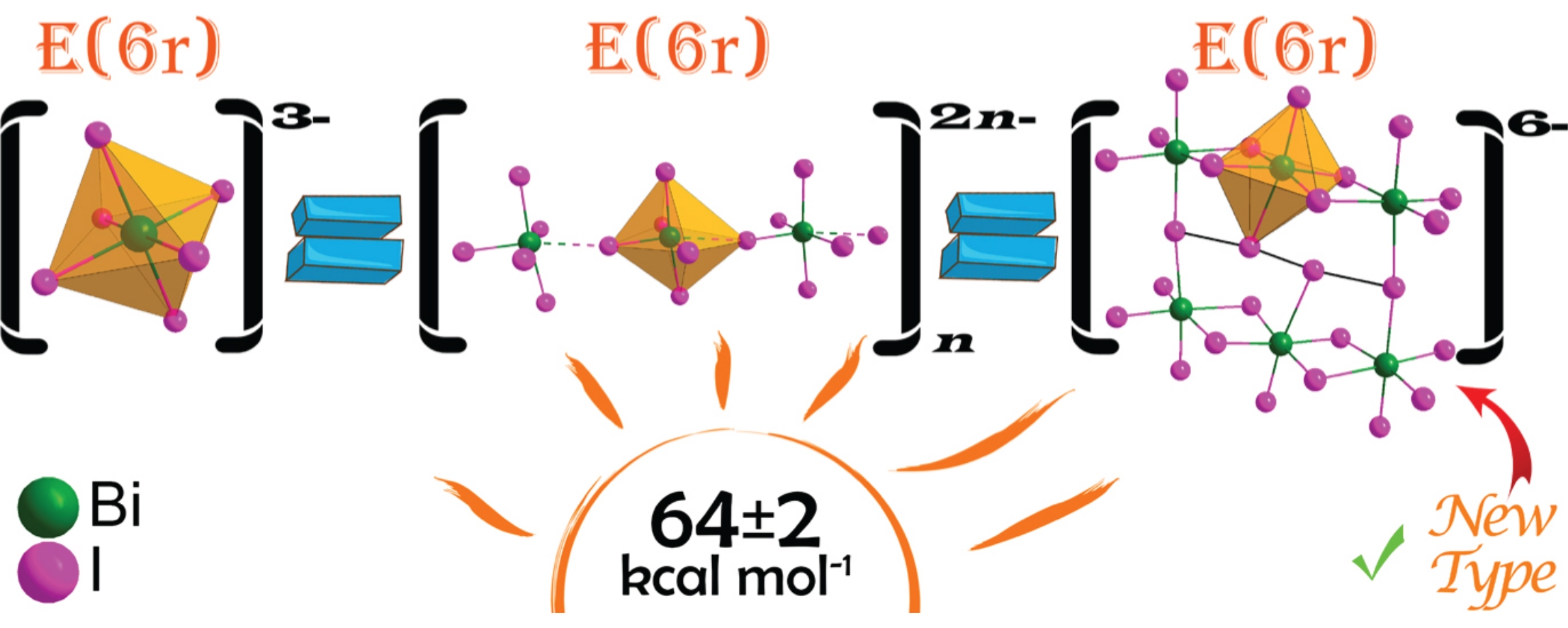
The crystal structure of the new iodobismuthate (PyPy)2(PyPyH)2Bi6I26 was found to consist of unusual hexanuclear [Bi6I26]6− anions containing the linear I42− unit, and the experimental Bi–I bond lengths in this anion were used to obtain the relationship between bond length and bond energy. A statistical analysis of 229 crystal structures of iodobismuthates, based on the quantum chemically estimated strength of Bi–I bonds, revealed that the total energy of the Bi3+ polyhedron remains virtually constant at 64 ± 2kcalmol−1, regardless of its geometry within this family of materials. Thus, the polyhedron geometry flexibly adapts to the relatively weak interactions between iodobismuthate anions and embedded cations.
References
1.

Suarez B., Gonzalez-Pedro V., Ripolles T.S., Sanchez R.S., Otero L., Mora-Sero I.
Journal of Physical Chemistry Letters,
2014
2.

Zhang Y., Liao W., Fu D., Ye H., Chen Z., Xiong R.
Journal of the American Chemical Society,
2015
3.

Wang G., Xu G., Wang M., Cai L., Li W., Guo G.
Chemical Science,
2015
4.
![Semiconductive Nanotube Array Constructed from Giant [PbII 18 I54 (I2 )9 ] Wheel Clusters](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Wang G., Xu G., Liu B., Wang M., Yao M., Guo G.
Angewandte Chemie - International Edition,
2015
5.

Pandey S., Chattopadhyay T., Dev S., Patil Y., Carpenter-Warren C.L., Sinha C.
Polyhedron,
2020
6.

Ben Ali S., Ferretti V., Del Bianco L., Spizzo F., Belhouchet M.
Journal of Molecular Structure,
2020
7.
10.1016/j.mencom.2021.03.007_bib0035
Organic-Inorganic Halide Perovskite Photovoltaics: From Fundamentals to Device Architectures,
2016
8.

Pellet N., Gao P., Gregori G., Yang T., Nazeeruddin M.K., Maier J., Grätzel M.
Angewandte Chemie - International Edition,
2014
9.

Green M.A., Ho-Baillie A., Snaith H.J.
Nature Photonics,
2014
10.

Yang W.S., Park B., Jung E.H., Jeon N.J., Kim Y.C., Lee D.U., Shin S.S., Seo J., Kim E.K., Noh J.H., Seok S.I.
Science,
2017
11.

Kojima A., Teshima K., Shirai Y., Miyasaka T.
Journal of the American Chemical Society,
2009
12.

Shin S.S., Yeom E.J., Yang W.S., Hur S., Kim M.G., Im J., Seo J., Noh J.H., Seok S.I.
Science,
2017
13.

Yan K., Long M., Zhang T., Wei Z., Chen H., Yang S., Xu J.
Journal of the American Chemical Society,
2015
14.
NREL, Best Research-Cell Efficiency Chart, https://www.nrel.gov/pv/cell-efficiency.html.
15.

Zhao J., Cai B., Luo Z., Dong Y., Zhang Y., Xu H., Hong B., Yang Y., Li L., Zhang W., Gao C.
Scientific Reports,
2016
16.

Adonin S.A., Sokolov M.N., Fedin V.P.
Coordination Chemistry Reviews,
2016
17.
Kotov V.Y., Ilyukhin A.B., Buikin P.A., Yorov K.E.
Mendeleev Communications,
2019
18.

Saidaminov M.I., Abdelhady A.L., Murali B., Alarousu E., Burlakov V.M., Peng W., Dursun I., Wang L., He Y., Maculan G., Goriely A., Wu T., Mohammed O.F., Bakr O.M.
Nature Communications,
2015
19.

Chen Y., Yang Z., Guo C., Ni C., Ren Z., Li H., Lang J.
European Journal of Inorganic Chemistry,
2010
20.

Buikin P.A., Ilyukhin A.B., Simonenko N.P., Laurinavichyute V.K., Kotov V.Y.
Polyhedron,
2018
21.
![Large Spontaneous Polarization and Clear Hysteresis Loop of a Room-Temperature Hybrid Ferroelectric Based on Mixed-Halide [BiI3Cl2] Polar Chains and Methylviologen Dication](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Leblanc N., Mercier N., Zorina L., Simonov S., Auban-Senzier P., Pasquier C.
Journal of the American Chemical Society,
2011
22.
![The motley family of polar compounds (MV)[M(X5−xX′x)] based on anionic chains of trans-connected M(III)(X,X′)6 octahedra (M=Bi, Sb; X, X′=Cl, Br, I) and methylviologen (MV) dications](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Leblanc N., Mercier N., Allain M., Toma O., Auban-Senzier P., Pasquier C.
Journal of Solid State Chemistry,
2012
23.

Kotov V.Y., Ilyukhin A.B., Korlyukov A.A., Smol’yakov A.F., Kozyukhin S.A.
New Journal of Chemistry,
2018
24.

Saparov B., Mitzi D.B.
Chemical Reviews,
2016
25.

Li T., Hu Y., Morrison C.A., Wu W., Han H., Robertson N.
Sustainable Energy and Fuels,
2017
26.
Buikin P.A., Ilyukhin A.B., Baranchikov A.E., Yorov K.E., Kotov V.Y.
Mendeleev Communications,
2018
27.

Kresse G., Hafner J.
Physical Review B,
1993
28.

Adamo C., Barone V.
Journal of Chemical Physics,
1999
29.

Grimme S., Antony J., Ehrlich S., Krieg H.
Journal of Chemical Physics,
2010
30.
10.1016/j.mencom.2021.03.007_bib0150
Matta
The Quantum Theory of Atoms in Molecules: From Solid State to DNA and Drug Design,
2007
31.

Espinosa E., Molins E., Lecomte C.
Chemical Physics Letters,
1998
32.
Lyssenko K.A.
Mendeleev Communications,
2012
33.

Ananyev I.V., Karnoukhova V.A., Dmitrienko A.O., Lyssenko K.A.
Journal of Physical Chemistry A,
2017
34.

Ananyev I.V., Bushmarinov I.S., Ushakov I.E., Aitkulova A.I., Lyssenko K.A.
RSC Advances,
2015
35.
Panova M.V., Medvedev M.G., Bushmarinov I.S., Ananyev I.V., Lyssenko K.A.
Mendeleev Communications,
2017
36.

Groom C.R., Bruno I.J., Lightfoot M.P., Ward S.C.
Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials,
2016
37.
![(Bzl4P)2[Bi2I8] - ein Iodobismutat mit fünffach koordiniertem Bi3+-Ion](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Krautscheid H.
Zeitschrift fur Anorganische und Allgemeine Chemie,
1999
38.
![Synthesis and structure of bismuth-containing complexes [Ph3PMe] 2 + [BiI5]2− and [Ph3PMe] 2 + [BiI5 · C5H5N]2− · C5H5N](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Sharutin V.V., Egorova I.V., Sharutina O.K., Boyarkina E.A.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2008