Keywords
1-chloromethyl-3,4-dihydroisoquinolines
2-arylethylamines
isoquinolines
recyclization
thiazoles
thioureas
Abstract
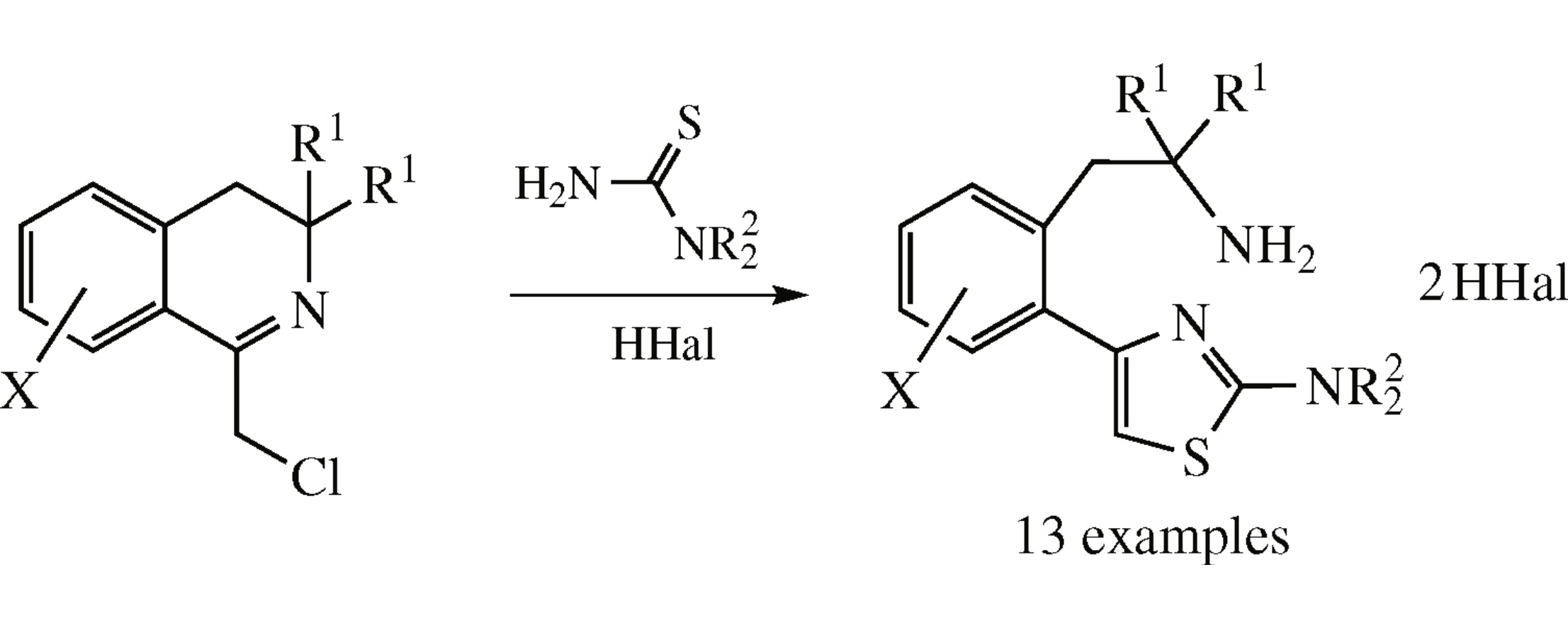
1-Chloromethyl-3,4-dihydroisoquinolines upon treatment with thioureas under acidic conditions undergo recyclization to afford new β-[o-(thiazol-4-yl)aryl]ethylamines.
References
1.

Yao H., Liu J., Xu S., Zhu Z., Xu J.
Expert Opinion on Drug Discovery,
2016
2.

Wang S., Dong G., Sheng C.
Chemical Reviews,
2019
3.
10.1016/j.mencom.2021.01.040_bib0015
Phytochemicals as Lead Compounds for New Drug Discovery,
2020
4.

Karageorgis G., Foley D.J., Laraia L., Waldmann H.
Nature Chemistry,
2020
5.

Gemma S.
Molecules,
2020
6.

Ertl P., Roggo S., Schuffenhauer A.
Journal of Chemical Information and Modeling,
2007
7.
10.1016/j.mencom.2021.01.040_bib0035
Trace Amines and Neurological Disorders: Potential Mechanisms and Risk Factors,
2016
8.

Lindemann L., Meyer C.A., Jeanneau K., Bradaia A., Ozmen L., Bluethmann H., Bettler B., Wettstein J.G., Borroni E., Moreau J., Hoener M.C.
Journal of Pharmacology and Experimental Therapeutics,
2007
9.
www.drugbank.ca.
10.
10.1016/j.mencom.2021.01.040_bib0050
Novel Psychoactive Substances: Policy, Economics and Drug Regulation,
2017
11.
C. Miller, Patent WO/2019/144132, 2019.
12.
K. Ohdachi, Y. Fujimori, N. Makita, N. Koseki, H. Hayashi, Y. Sakamoto, K. Mineno and R. Taga, Patent WO/2020/050409, 2020.
13.

Cinelli M.A., Reidl C.T., Li H., Chreifi G., Poulos T.L., Silverman R.B.
Journal of Medicinal Chemistry,
2020
14.

Appukkuttan P., Orts A., Chandran R. ., Goeman J. ., Van der Eycken J., Dehaen W., Van der Eycken E.
European Journal of Organic Chemistry,
2004
15.
![8-(Heteroaryl)phenalkyl-1-Phenyl-1,3,8-triazaspiro[4.5]decan-4-ones as Opioid Receptor Modulators](/storage/images/resized/doUaFUZdxUEQjLi1TwZjGHi8HXYNWWSk04dSC6Xh_small_thumb.webp)
Jordan A., Orsini M., Middleton S., Connolly P., Brenneman D., Pan K., Reitz A.
Medicinal Chemistry,
2005
16.

Ross T.M., Battista K., Bignan G.C., Brenneman D.E., Connolly P.J., Liu J., Middleton S.A., Orsini M., Reitz A.B., Rosenthal D.I., Scott M.K., Vaidya A.H.
Bioorganic and Medicinal Chemistry Letters,
2015
17.

Peshkov A.A., Peshkov V.A., Pereshivko O.P., Van Hecke K., Kumar R., V. Van der Eycken E.
Journal of Organic Chemistry,
2015
18.
![One-pot synthesis of polyhydropyrido[1,2- a ]indoles and tetracyclic quinazolinones from 2-arylindoles using copper-mediated oxidative tandem reactions](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Yamashita M., Nishizono Y., Himekawa S., Iida A.
Tetrahedron,
2016
19.

Dai C., Huang Z., Liu L., Han Y., Shi D., Zhao Y.
European Journal of Organic Chemistry,
2020
20.
L. Jianqi, G. Zhengsong, Z. Qingwei, M. Zhilong, Z. Zixue and G. Zheng, Patent CN110498789, 2019.
21.
B. J. Morrow, R.C. Foitzik, M.A. Camerine, H.R. Lagiakos, S.R. Walker, Y.E. B. Bozikis, G.I. Stevenson, A.N. Cuzzupe and P. A. Stupple, Patent WO/2019/043139, 2019.
22.

Fournier P., Arbour M., Cauchon E., Chen A., Chefson A., Ducharme Y., Falgueyret J., Gagné S., Grimm E., Han Y., Houle R., Lacombe P., Lévesque J., MacDonald D., Mackay B., et. al.
Bioorganic and Medicinal Chemistry Letters,
2012
23.

Kusuma B.R., Zhang L., Sundstrom T., Peterson L.B., Dobrowsky R.T., Blagg B.S.
Journal of Medicinal Chemistry,
2012
24.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Demidov O.P., Sochnev V.S., Borodkina I.G., Drobin Y.D., Spasov A.A.
Mendeleev Communications,
2020
25.

Kartsev V.G., Zubenko A.A., Morkovnik A.S., Divaeva L.N.
Tetrahedron Letters,
2015
26.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Kartsev V.G., Suponitsky K.Y., Klimenko A.I.
Mendeleev Communications,
2018
27.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Kartsev V.G., Borodkin G.S., Klimenko A.I.
Mendeleev Communications,
2018
28.
CrysAlis Pro, version 1.171.38.41, Rigaku Oxford Diffraction, 2015.
29.
10.1016/j.mencom.2021.01.040_bib0135
Sheldrick
Acta Crystallogr.,
2015
30.
10.1016/j.mencom.2021.01.040_bib0140
Sheldrick
Acta Crystallogr.,
2015
31.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
32.

Zhao M., Ren Z., Yu L., Wang Y., Guan Z.
Organic Letters,
2016
33.

Khatik G.L., Datusalia A.K., Ahsan W., Kaur P., Vyas M., Mittal A., Nayak S.K.
Current Drug Discovery Technologies,
2018
34.

Agarwal S., Kalal P., Gandhi D., Prajapat P.
Current Drug Discovery Technologies,
2017
35.

Kumawat M.K.
Current Drug Discovery Technologies,
2017
36.

Shakya A., Bhat H.R., Ghosh S.K.
Current Drug Discovery Technologies,
2017
37.

Kashyap A., Adhikari N., Das A., Shakya A., Ghosh S.K., Singh U.P., Bhat H.R.
Current Drug Discovery Technologies,
2018
38.

Sharma D., Malhotra A., Bansal R.
Current Drug Discovery Technologies,
2018