Keywords
p-toluenesulfonic acid
4-hydroxycoumarin
benzylation
solvent-free
styrene
Abstract
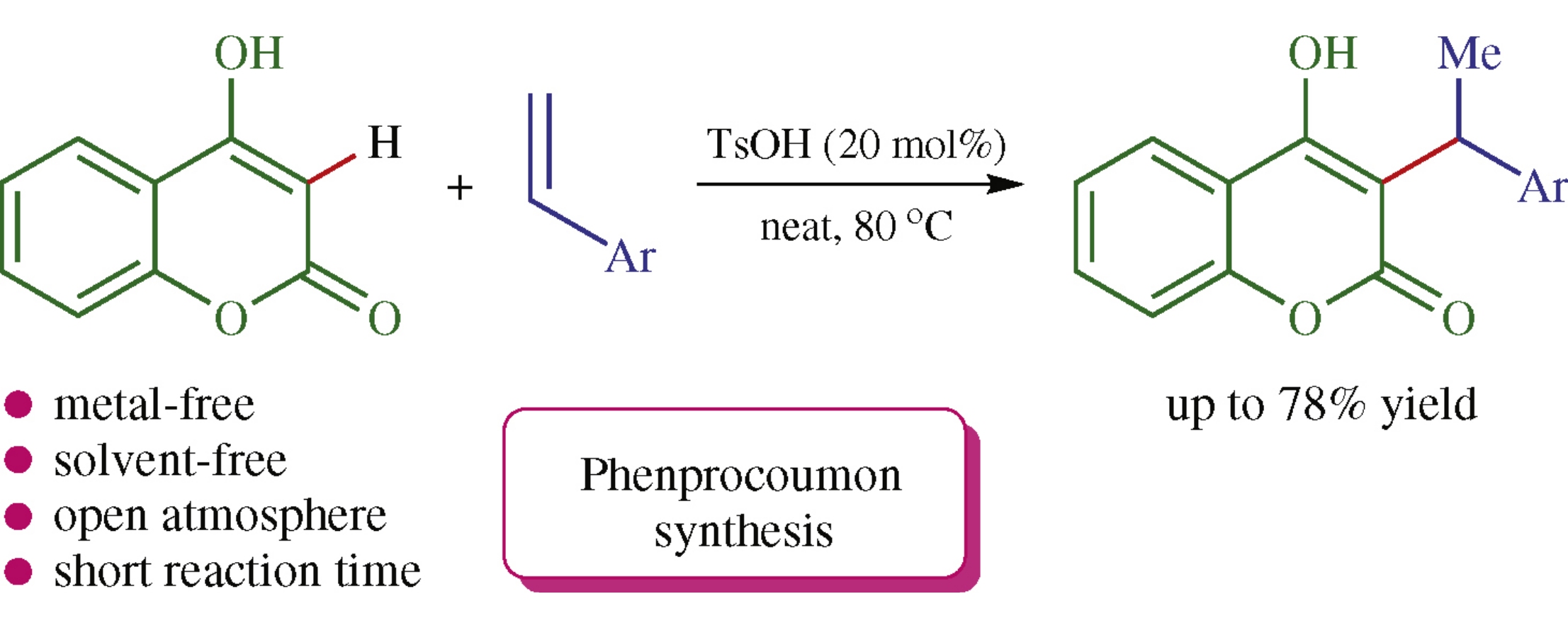
An efficient straightforward solvent-free C(3)-benzylation of 4-hydroxycoumarin with styrenes is performed by heating the reactants in the presence of p-toluenesulfonic acid. By this procedure, benzylated 4-hydroxycoumarin derivatives which exhibit various biological activities were obtained.
References
1.

Lacy A.
Current Pharmaceutical Design,
2004
2.

Medina F.G., Marrero J.G., Macías-Alonso M., González M.C., Córdova-Guerrero I., Teissier García A.G., Osegueda-Robles S.
Natural Product Reports,
2015
3.
10.1016/j.mencom.2021.01.039_bib0015
Athanasellis
Synthesis,
2004
4.

Yu D., Suzuki M., Xie L., Morris-Natschke S.L., Lee K.
Medicinal Research Reviews,
2003
5.

Kirkiacharian B.S., De Clercq E., Kurkjian R., Pannecouque C.
Pharmaceutical Chemistry Journal,
2008
6.

Chohan Z.H., Shaikh A.U., Rauf A., Supuran C.T.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2006
7.

Luchini A.C., Rodrigues-Orsi P., Cestari S.H., Seito L.N., Witaicenis A., Pellizzon C.H., Di Stasi L.C.
Biological and Pharmaceutical Bulletin,
2008
8.
10.1016/j.mencom.2021.01.039_bib0040
Chiarino
Eur. Pat. Appl.,
1988
9.

Cravotto G., Tagliapietra S., Cappello R., Palmisano G., Curini M., Boccalini M.
Archiv der Pharmazie,
2006
10.

Pérez-Cruz F., Serra S., Delogu G., Lapier M., Maya J.D., Olea-Azar C., Santana L., Uriarte E.
Bioorganic and Medicinal Chemistry Letters,
2012
11.

Barot K.P., Jain S.V., Kremer L., Singh S., Ghate M.D.
Medicinal Chemistry Research,
2015
12.

Au N., Rettie A.E.
Drug Metabolism Reviews,
2008
13.

Rullo M., Pisani L.
Chemistry of Heterocyclic Compounds,
2018
14.
Fatykhov R.F., Savchuk M.I., Starnovskaya E.S., Bobkina M.V., Kopchuk D.S., Nosova E.V., Zyryanov G.V., Khalymbadzha I.A., Chupakhin O.N., Charushin V.N., Kartsev V.G.
Mendeleev Communications,
2019
15.

Rueping M., Nachtsheim B., Sugiono E.
Synlett,
2010
16.

Lin X., Dai X., Mao Z., Wang Y.
Tetrahedron,
2009
17.

Thirupathi P., Kim S.S.
Tetrahedron,
2010
18.

Reddy C.R., Srikanth B., Narsimha Rao N., Shin D.
Tetrahedron,
2008
19.

Theerthagiri P., Lalitha A.
Tetrahedron Letters,
2010
20.

Kischel J., Mertins K., Michalik D., Zapf A., Beller M.
Advanced Synthesis and Catalysis,
2007
21.

Angeles-Dunham V.V., Nickerson D.M., Ray D.M., Mattson A.E.
Angewandte Chemie - International Edition,
2014
22.
![Brønsted acidic ionic liquid-catalyzed tandem reaction: an efficient approach towards regioselective synthesis of pyrano[3,2-c]coumarins under solvent-free conditions bearing lower E-factors](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Mahato S., Santra S., Chatterjee R., Zyryanov G.V., Hajra A., Majee A.
Green Chemistry,
2017
23.

Chatterjee R., Santra S., Zyryanov G., Majee A.
Synthesis,
2019
24.

Chatterjee R., Mahato S., Santra S., Zyryanov G.V., Hajra A., Majee A.
ChemistrySelect,
2018
25.

Chatterjee R., Santra S., Zyryanov G.V., Majee A.
Journal of Heterocyclic Chemistry,
2020