Keywords
betulinic acid
glycoside
glycosylation
lupane
mannopyranose
phosphonium salts
saponins
triterpenes
Abstract
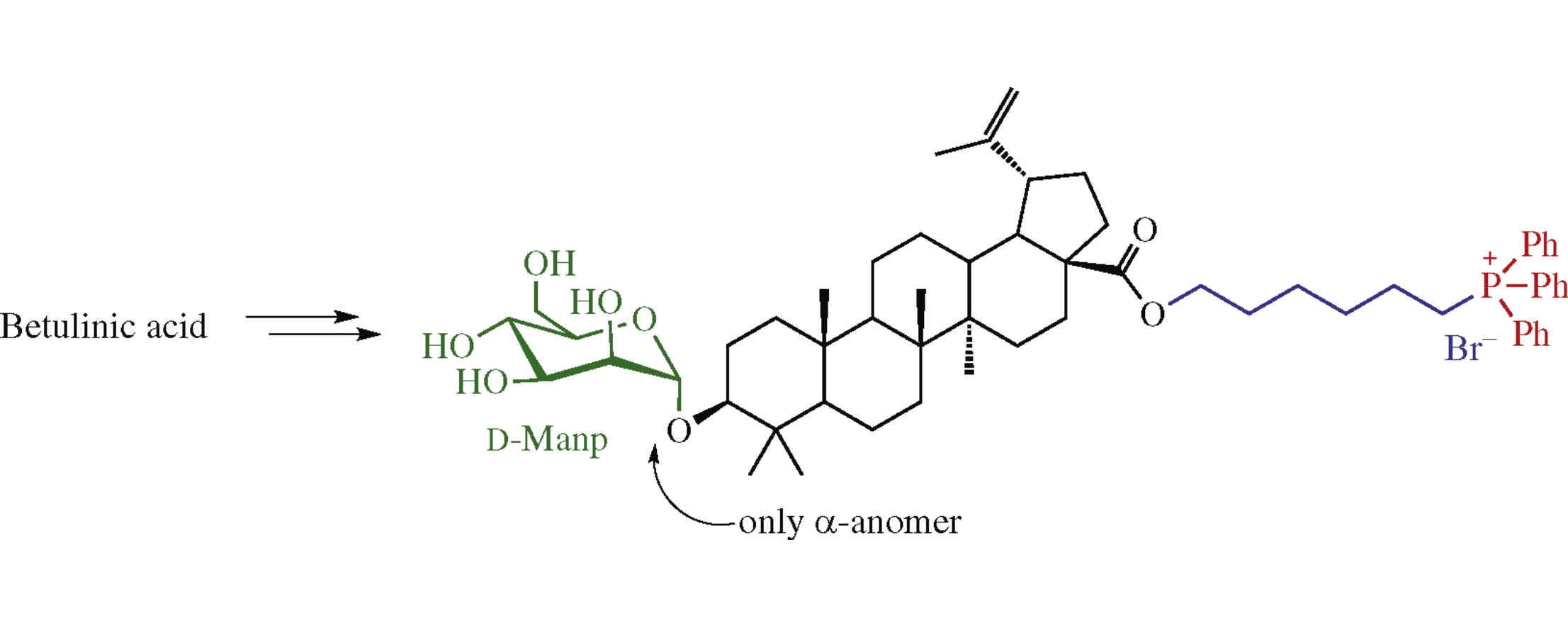
A general approach to lupane monodesmosides containing a C-28 phosphonioalkyl group is based on the initial glycosylation of triterpene acid ω-haloalkyl esters at the C-3 position. The subsequent phosphorylation with triphenylphosphine and deprotection in the carbohydrate moiety finalize the synthesis.
References
1.
10.1016/j.mencom.2021.01.034_sbref0005a
2018
2.
10.1016/j.mencom.2021.01.034_sbref0005b
1974
3.

4.

Sheng H., Sun H.
Natural Product Reports,
2011
5.

Safayhi H., Sailer E.-.
Planta Medica,
1997
6.

Pádua T.A., de Abreu B.S., Costa T.E., Nakamura M.J., Valente L.M., Henriques M.D., Siani A.C., Rosas E.C.
Archives of Pharmacal Research,
2014
7.

Smina T.P., Mathew J., Janardhanan K.K., Devasagayam T.P.
Environmental Toxicology and Pharmacology,
2011
8.

Cichewicz R.H., Kouzi S.A.
Medicinal Research Reviews,
2003
9.
10.1016/j.mencom.2021.01.034_bib0035
Alqahtani
Curr. Med. Chem.,
2013
10.

Laszczyk M.
Planta Medica,
2009
11.

M.R. Patlolla J., V. Rao C.
Current Pharmaceutical Biotechnology,
2011
12.

Prasad S., Kalra N., Shukla Y.
Molecular Nutrition and Food Research,
2007
13.

Hussain Shaik A., Rasool S.N., Kareem M.A., Krushna G.S., Akhtar P.M., Devi K.L.
Journal of Medicinal Food,
2012
14.

Sánchez M., Theoduloz C., Schmeda-Hirschmann G., Razmilic I., Yáñez T., Rodríguez J.A.
Life Sciences,
2006
15.

Ghaffari Moghaddam M., Bin H. Ahmad F., Samzadeh-Kermani A.
Pharmacology & Pharmacy,
2012
16.

Yogeeswari P., Sriram D.
Current Medicinal Chemistry,
2005
17.

Mullauer F.B., Kessler J.H., Medema J.P.
Anti-Cancer Drugs,
2010
18.
10.1016/j.mencom.2021.01.034_sbref0065a
Csuk
Expert Opin. Ther. Pat.,
2014
19.
10.1016/j.mencom.2021.01.034_sbref0065b
Saxena
CBI,
2004
20.

Urban M., Sarek J., Klinot J., Korinkova G., Hajduch M.
Journal of Natural Products,
2004
21.

Baratto L.C., Porsani M.V., Pimentel I.C., Pereira Netto A.B., Paschke R., Oliveira B.H.
European Journal of Medicinal Chemistry,
2013
22.

Kommera H., Kaluđerović G.N., Kalbitz J., Dräger B., Paschke R.
European Journal of Medicinal Chemistry,
2010
23.

Mullauer F.B., Kessler J.H., Medema J.P.
Apoptosis : an international journal on programmed cell death,
2008
24.

Smith R.A., Hartley R.C., Murphy M.P.
Antioxidants and Redox Signaling,
2011
25.

Spivak A.Y., Nedopekina D.A., Shakurova E.R., Khalitova R.R., Gubaidullin R.R., Odinokov V.N., Dzhemilev U.M., Bel’skii Y.P., Bel’skaya N.V., Stankevich S.A., Korotkaya E.V., Khazanov V.A.
Russian Chemical Bulletin,
2013
26.

Spivak A.Y., Keiser J., Vargas M., Gubaidullin R.R., Nedopekina D.A., Shakurova E.R., Khalitova R.R., Odinokov V.N.
Bioorganic and Medicinal Chemistry,
2014
27.

Spivak A.Y., Nedopekina D.A., Khalitova R.R., Gubaidullin R.R., Odinokov V.N., Bel’skii Y.P., Bel’skaya N.V., Khazanov V.A.
Medicinal Chemistry Research,
2017
28.

Nedopekina D.A., Gubaidullin R.R., Odinokov V.N., Maximchik P.V., Zhivotovsky B., Bel'skii Y.P., Khazanov V.A., Manuylova A.V., Gogvadze V., Spivak A.Y.
MedChemComm,
2017
29.

Tsepaeva O.V., Nemtarev A.V., Abdullin T.I., Grigor’eva L.R., Kuznetsova E.V., Akhmadishina R.A., Ziganshina L.E., Cong H.H., Mironov V.F.
Journal of Natural Products,
2017
30.
10.1016/j.mencom.2021.01.034_sbref0080f
Antipin
Eur. J. Clin. Inv.,
2018
31.

Reaction of Methyl (2-Methylidene)-3-oxolup-20(29)-en-28-oate with Dimethyl Trimethylsilyl Phosphite
Tsepaeva O.V., Nemtarev A.V., Abdullin T.I., Kuznetsova E.V., Mironov V.F.
Russian Journal of General Chemistry,
2018
32.

Tsepaeva O.V., Nemtarev A.V., Mironov V.F.
Russian Journal of Organic Chemistry,
2017
33.
10.1016/j.mencom.2021.01.034_sbref0080i
Antipin
Eur. J. Clin. Invest.,
2019
34.

Tsepaeva O.V., Nemtarev A.V., Salikhova T.I., Abdullin T.I., Grigor`eva L.R., Khozyainova S.A., Mironov V.F.
Anti-Cancer Agents in Medicinal Chemistry,
2020
35.

Ye Y., Zhang T., Yuan H., Li D., Lou H., Fan P.
Journal of Medicinal Chemistry,
2017
36.

Ponomaryov D.V., Grigor´eva L.R., Nemtarev A.V., Tsepaeva O.V., Mironov V.F., Antipin I.S.
Russian Chemical Bulletin,
2020
37.
10.1016/j.mencom.2021.01.034_sbref0085a
Oral Bioavailability: Basic Principles, Advanced Concepts, and Applications,
2011
38.

J. C. Furtado N., Pirson L., Edelberg H., M. Miranda L., Loira-Pastoriza C., Preat V., Larondelle Y., André C.
Molecules,
2017
39.

Vincken J., Heng L., de Groot A., Gruppen H.
Phytochemistry,
2007
40.

Gauthier C., Legault J., Lebrun M., Dufour P., Pichette A.
Bioorganic and Medicinal Chemistry,
2006
41.
42.

Schmidt R.R.
Pure and Applied Chemistry,
1989
43.

Korda A., Pakulski Z., Cmoch P., Gwardiak K., Karczewski R.
Tetrahedron,
2017
44.

Korda A., Pakulski Z., Cmoch P., Gwardiak K., Karczewski R.
Carbohydrate Research,
2018
45.
10.1016/j.mencom.2021.01.034_bib0110
Levy
The Organic Chemistry of Sugars,
2005