Keywords
1-aminophosphonate
5-oxo-2-phenylimidazolidine
dialkyl chlorophosphite
diversity-oriented synthesis
piperazine-2,5-dione
sodium N-benzylideneglycinate
Abstract
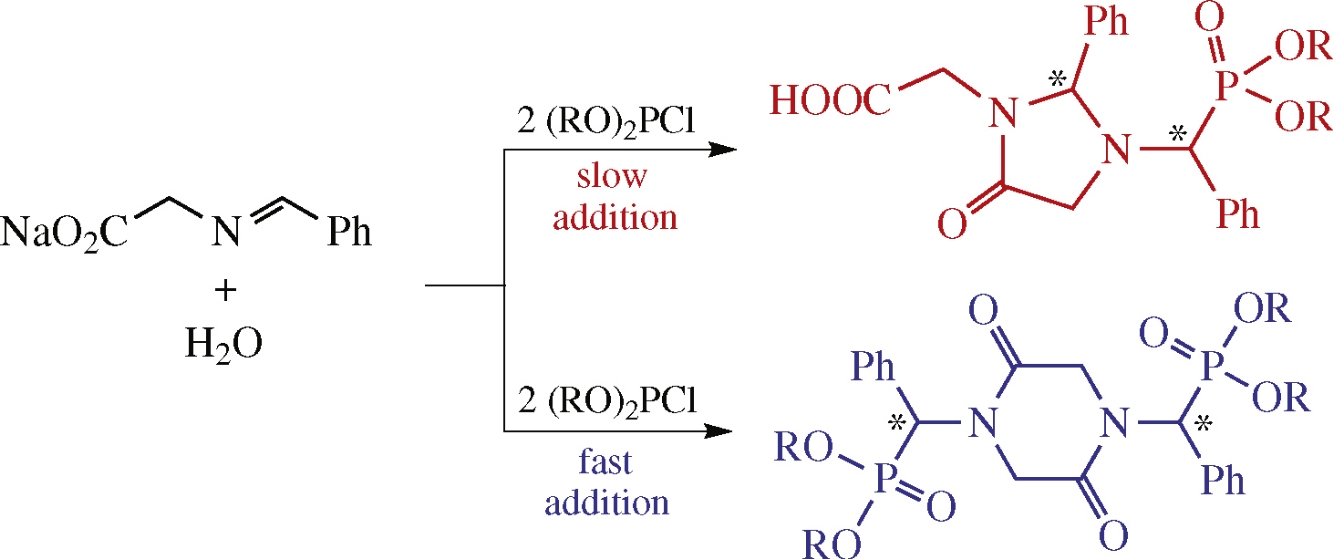
The outcome of reaction of sodium N-benzylideneglycinate containing water in its crystal lattice with dialkyl chlorophosphites depends on the mode of addition of the latter. Upon the simultaneous mixing of the reactants, 1,4-bis[α-(dialkoxyphosphoryl)benzyl]piperazine-2,5-diones are formed from two molecules of each reactant. With the slow addition of dialkyl chlorophosphite, the main reaction product is 2-{3-[α-(dialkoxyphosphoryl)benzyl]-5-oxo-2-phenylimidazolidin-1-yl}acetic acid (‘1: 2 adduct’), its formation comprising 1,4-migration of dialkoxyphosphoryl moiety.
References
1.
10.1016/j.mencom.2021.01.033_bib0005
Kukhar
Aminophosphonic and Aminophosphinic Acids: Chemistry and Biological Activity,
2000
2.

Rasal S.A., Dhavan P.P., Jadhav B.L., Shimpi N.G.
Applied Organometallic Chemistry,
2019
3.

Poola S., Nagaripati S., Tellamekala S., Chintha V., Kotha P., Yagani J.R., Golla N., Cirandur S.R.
Synthetic Communications,
2020
4.

An T.N., Cuong N.V., Quang N.M., Thanh T.V., Alam M.
ChemistrySelect,
2020
5.

Huang X., Wang M., Wang C., Hu W., You Q., Ma T., Jia Q., Yu C., Liao Z., Wang H.
Bioorganic Chemistry,
2020
6.

Huang X., Wang M., You Q., Kong J., Zhang H., Yu C., Wang Y., Wang H., Huang R.
European Journal of Medicinal Chemistry,
2020
7.

Carramiñana V., Ochoa de Retana A.M., Palacios F., de los Santos J.M.
Molecules,
2020
8.

Azzam M.A., El-Boraey H.A., El-Sayed I.E.
Phosphorus, Sulfur and Silicon and the Related Elements,
2019
9.

Ahmed A.A., Awad H.M., El-Sayed I.E., El Gokha A.A.
Journal of the Iranian Chemical Society,
2020
10.

Schweiker S.S., Tauber A.L., Kam C.M., Eyckens D.J., Henderson L.C., Levonis S.M.
ChemistrySelect,
2020
11.

Bazine I., Cheraiet Z., Bensegueni R., Bensouici C., Boukhari A.
Journal of Heterocyclic Chemistry,
2020
12.

Shaikh S., Dhavan P., Pavale G., Ramana M.M., Jadhav B.L.
Bioorganic Chemistry,
2020
13.
10.1016/j.mencom.2021.01.033_sbref0030b
Shaikh
Mol. Divers.,
2020
14.

Shaik M.S., Nadiveedhi M.R., Gundluru M., Poola S., Allagadda R., Chippada A.R., Cirandur S.R.
Synthetic Communications,
2020
15.

Marenin K.S., Agafontsev A.M., Bryleva Y.A., Gatilov Y.V., Glinskaya L.A., Piryazev D.A., Tkachev A.V.
ChemistrySelect,
2020
16.

Kaur G., Shamim M., Bhardwaj V., Gupta V.K., Banerjee B.
Synthetic Communications,
2020
17.

Lakoud S.G., Aissa R., Guillot R., Toffano M., Aribi‐Zouioueche L.
ChemistrySelect,
2020
18.

Kolli M.K., Palani E., Govindasamy C., Katta V.R.
Research on Chemical Intermediates,
2018
19.

Kunde S.P., Kanade K.G., Karale B.K., Akolkar H.N., Arbuj S.S., Randhavane P.V., Shinde S.T., Shaikh M.H., Kulkarni A.K.
RSC Advances,
2020
20.

Hudson H.R., Tajti Á., Bálint E., Czugler M., Karaghiosoff K., Keglevich G.
Synthetic Communications,
2020
21.

Shilpa T., Ann Harry N., Ujwaldev S.M., Anilkumar G.
ChemistrySelect,
2020
22.

Smolobochkin A.V., Gazizov A.S., Doszhanova K.A., Kuandykova A.B., Jiyembayev B.Z., Burilov A.R., Pudovik M.A., Cherkasov R.A.
Russian Journal of General Chemistry,
2020
23.

Keglevich G., Rádai Z.
Tetrahedron Letters,
2020
24.

Maestro A., Marigorta E.M., Palacios F., Vicario J.
Asian Journal of Organic Chemistry,
2020
25.
10.1016/j.mencom.2021.01.033_sbref0050a
Litvinov
Russ. J. Gen. Chem.,
1999
26.

Mironov V.F., Gubaidullin A.T., Ivkova G.A., Litvinov I.A., Buzykin B.I., Burnaeva L.M., Konovalova I.V.
Russian Journal of General Chemistry,
2001
27.

Mironov V.F., Gubaidullin A.T., Burnaeva L.M., Litvinov I.A., Ivkova G.A., Romanov S.V., Zyablikova† T.A., Konovalov A.I., Konovalova I.V.
Russian Journal of General Chemistry,
2004
28.
![Intramolecular transformation of 2-benzylideneaminoethoxybenzo[d]-1,3,2- dioxaphosphorin-4-one into 3,4-benzo-10-phenyl-1-aza-5,7-dioxa-6-phosphabicyclo- [4.3.1]decane-2,6-dione](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Dimukhametov M.N., Abdrakhmanova L.M., Mironov V.F., Musin R.Z.
Russian Journal of General Chemistry,
2010
29.
Dimukhametov M.N., Ivkova G.A., Litvinov I.A., Fayzullin R.R., Mironov V.F.
Mendeleev Communications,
2019
30.

Dimukhametov M.N., Bayandina E.V., Davydova E.Y., Zyablikova T.A., Dobrynin A.B., Litvinov I.A., Al"fonsov V.A.
Russian Chemical Bulletin,
2001
31.
![A convenient synthesis and spatial structure of 2-aryl-2-oxo-2- phenylbenzo[e]-1,4,2 oxazaphosphinanes](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Dimukhametov M.N., Mironov V.F., Mironova E.V., Krivolapov D.B., Dobrynin A.B., Litvinov I.A., Musin R.Z.
Russian Chemical Bulletin,
2013
32.
Dimukhametov M.N., Abaskalova M.A., Davydova E.Y., Bayandina E.V., Dobrynin A.B., Litvinov I.A., Alfonsov V.A.
Mendeleev Communications,
2004
33.
Dimukhametov M.N., Musin R.Z., Buzykin B.I., Latypov S.K., Mironov V.F.
Mendeleev Communications,
2005
34.
10.1016/j.mencom.2021.01.033_bib0070
Sheldrick
Acta Crystallogr.,
2015
35.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
36.

Macrae C.F., Edgington P.R., McCabe P., Pidcock E., Shields G.P., Taylor R., Towler M., van de Streek J.
Journal of Applied Crystallography,
2006