Keywords
advanced glycation end-products
aggregation
amyloid structure
protein glycation
thioflavin T
β-casein
Abstract
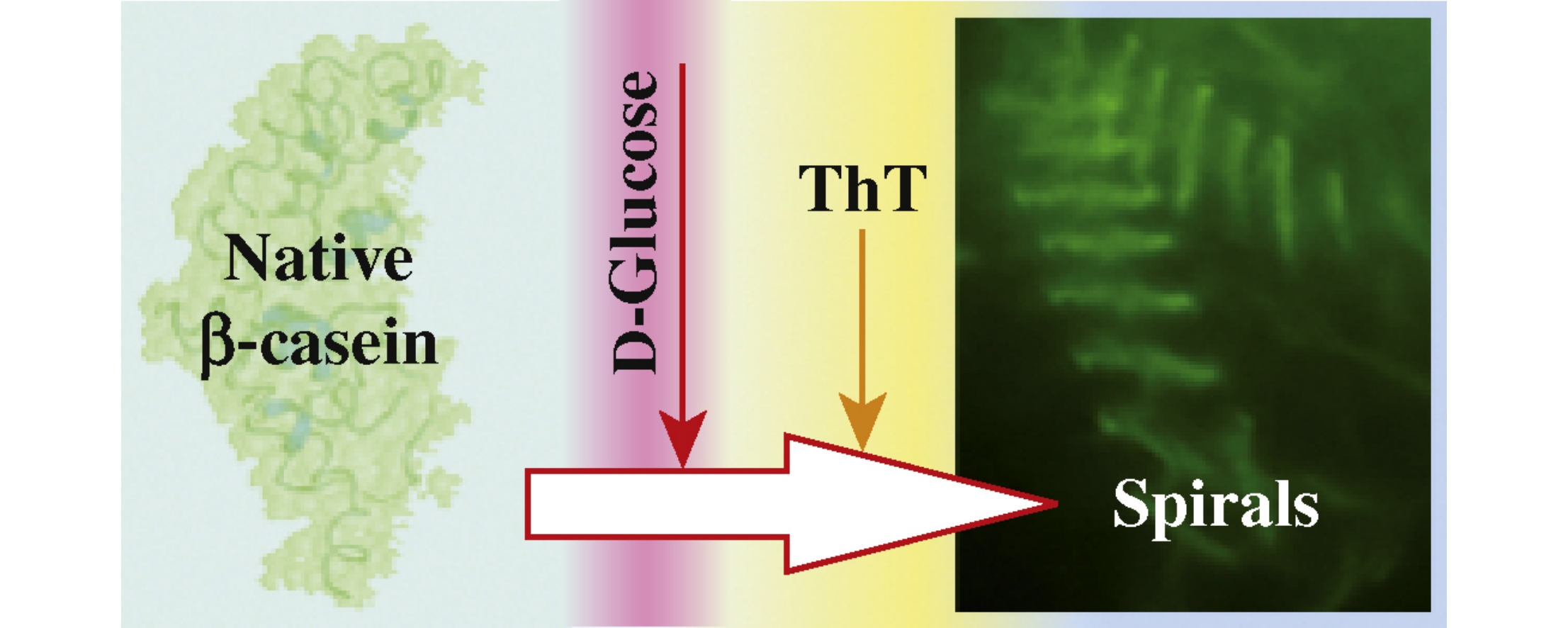
Unusual spiral-like structures in aggregates formed by β-casein glycated in a special way in the presence of thioflavin T are reported. Different glycation agents, temperature, pH, incubation time, and concentrations of protein and modifier were characterized, but only glycated by 200mm glucose for 48h at 37°C without sodium cyanoborohydride β-casein forms spiral structures in the presence of thioflavin T. Thioflavin T affects the size of particles formed by glycated β-casein and also stimulates heat-induced aggregation, indicating that the formation of unusual spiral structures is determined both by the structure of the advanced glycation end products and by the properties of the glycated protein.
References
1.

Adrover M., Mariño L., Sanchis P., Pauwels K., Kraan Y., Lebrun P., Vilanova B., Muñoz F., Broersen K., Donoso J.
Biomacromolecules,
2014
2.

Iannuzzi C., Irace G., Sirangelo I.
Frontiers in Molecular Biosciences,
2014
3.

Li J., Liu D., Sun L., Lu Y., Zhang Z.
Journal of the Neurological Sciences,
2012
4.

Vistoli G., De Maddis D., Cipak A., Zarkovic N., Carini M., Aldini G.
Free Radical Research,
2013
5.

Roldan M., Loebner J., Degen J., Henle T., Antequera T., Ruiz-Carrascal J.
Food Chemistry,
2015
6.
10.1016/j.mencom.2021.01.022_bib0030
Qu
Mol. Nutr. Food Res.,
2017
7.

Zhao D., Le T.T., Larsen L.B., Li L., Qin D., Su G., Li B.
Food Research International,
2017
8.

Cardoso H.B., Wierenga P.A., Gruppen H., Schols H.A.
Food Chemistry,
2018
9.

Geicu O.I., Stanca L., Dinischiotu A., Serban A.I.
Scientific Reports,
2018
10.

O'Mahony J.A., Drapala K.P., Mulcahy E.M., Mulvihill D.M.
International Dairy Journal,
2017
11.

Jindal S., Naeem A.
Journal of Fluorescence,
2013
12.

Yousefi R., Ferdowsi L., Tavaf Z., Sadeghian T., Tamaddon A., Moghtaderi M., Pourpak Z.
Protein and Peptide Letters,
2016
13.

Stroylova Y.Y., Zimny J., Yousefi R., Chobert J., Jakubowski H., Muronetz V.I., Haertlé T.
Biochimica et Biophysica Acta - Proteins and Proteomics,
2011
14.
10.1016/j.mencom.2021.01.022_bib0070
Mercier
Bull. Soc. Chim. Biol.,
1968
15.

Yaylayan V.A., Huyghues‐Despointes A., Feather M.S.
Critical Reviews in Food Science and Nutrition,
1994
16.

Barinova K., Khomyakova E., Semenyuk P., Schmalhausen E., Muronetz V.
Archives of Biochemistry and Biophysics,
2018