Keywords
meso-sulfophenyloxy dyes
N-acylsulfonamides
acetylation
cyanine dyes
electroneutral dyes
fluorescently labeled nucleotides
heptamethine dyes
sulfonamides
Abstract
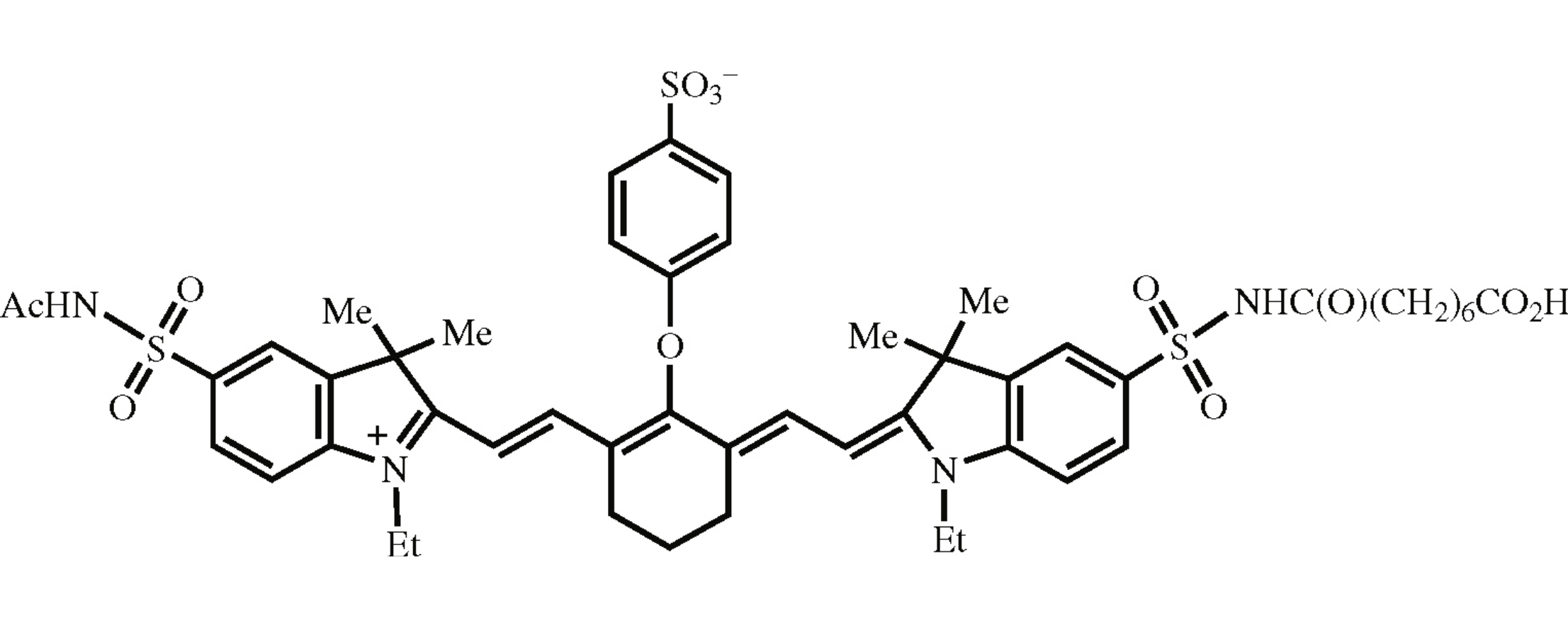
A novel electroneutral rigid meso-sulfophenyloxy substituted heptamethine dye was synthesized in six steps. Selective derivatization of one sulfonamide group with octanedioic acid introduced the carboxy end group attached through the hexamethylene linker, which provided the dye solubility in water. Absorbance of the dye in the near infrared region makes it promising for covalent labeling of biomolecules.
References
1.

Shi C., Wu J.B., Pan D.
Journal of Biomedical Optics,
2016
2.

Hyun H., Owens E.A., Wada H., Levitz A., Park G., Park M.H., Frangioni J.V., Henary M., Choi H.S.
Angewandte Chemie - International Edition,
2015
3.

Owens E.A., Henary M., El Fakhri G., Choi H.S.
Accounts of Chemical Research,
2016
4.

Kasprzak J., Samsel D., Borkowska A., Kecik M.
Acta Physica Polonica A,
2011
5.
Levchenko K.S., Chudov K.A., Zinoviev E.V., Lyssenko K.A., Fakhrutdinov A.N., Demin D.U., Poroshin N.O., Zhukova A.A., Shmelin P.S., Grebennikov E.P.
Mendeleev Communications,
2019
6.

Lee H., Mason J.C., Achilefu S.
Journal of Organic Chemistry,
2006
7.
10.1016/j.mencom.2021.01.021_sbref0010b
Liu
2014
8.

Lin C., Usama S., Burgess K.
Molecules,
2018
9.

Chipon B., Clavé G., Bouteiller C., Massonneau M., Renard P., Romieu A.
Tetrahedron Letters,
2006
10.
Thavornpradit S., Usama S.M., Park G.K., Shrestha J.P., Nomura S., Baek Y., Choi H.S., Burgess K.
Theranostics,
2019
11.

Mironov A.F., Zhdanova K.A., Bragina N.A.
Russian Chemical Reviews,
2018
12.
Zhdanova K.A., Ezhov A.V., Bragina N.A., Mironov A.F.
Mendeleev Communications,
2018
13.

Amiot C., Xu S., Liang S., Pan L., Zhao J.
Sensors,
2008
14.
Deng G., Li S., Sun Z., Li W., Zhou L., Zhang J., Gong P., Cai L.
Theranostics,
2018
15.

Vus K., Tarabara U., Kurutos A., Ryzhova O., Gorbenko G., Trusova V., Gadjev N., Deligeorgiev T.
Molecular BioSystems,
2017
16.

Okoh O.A., Critchley M.E., Bisby R.H., Lawrence C.L., Wainwright M., Smith R.B.
Coloration Technology,
2019
17.
10.1016/j.mencom.2021.01.021_sbref0020b
Henary
Heterocycl. Commun.,
2013
18.

Podrugina T.A., Pavlova A.S., Doroshenko I.A., Kuz’min V.A., Kostyukov A.A., Shtil’ A.A.
Russian Chemical Bulletin,
2018
19.
Prokhorov V.V., Pozin S.I., Perelygina O.M., Mal’tsev E.I.
Mendeleev Communications,
2019
20.
James N.S., Chen Y., Joshi P., Ohulchanskyy T.Y., Ethirajan M., Henary M., Strekowsk L., Pandey R.K.
Theranostics,
2013
21.

Braun A.B., Wehl I., Kölmel D.K., Schepers U., Bräse S.
Chemistry - A European Journal,
2019
22.
Shershov V.E., Kuznetsova V.E., Lapa S.A., Spitsyn M.A., Guseinov T.O., Tkachev Y.V., Zasedatelev A.S., Chudinov A.V.
Mendeleev Communications,
2017
23.

Spitsyn M.A., Kuznetsova V.E., Shershov V.E., Emelyanova М.А., Guseinov T.O., Lapa S.A., Nasedkina T.V., Zasedatelev A.S., Chudinov A.V.
Dyes and Pigments,
2017
24.

Shershov V.E., Spitsyn M.A., Kuznetsova V.E., Timofeev E.N., Ivashkina O.A., Abramov I.S., Nasedkina T.V., Zasedatelev A.S., Chudinov A.V.
Dyes and Pigments,
2013
25.
G. Caputo and C. L. Della, Patent EP 1491591 A1, 2004.
26.

Malet-Sanz L., Madrzak J., Ley S.V., Baxendale I.R.
Organic and Biomolecular Chemistry,
2010
27.
Tacic A., Nikolic V., Nikolic L., Savic I.
Advanced technologies,
2017
28.

Flanagan, J.H., Khan S.H., Menchen S., Soper S.A., Hammer R.P.
Bioconjugate Chemistry,
1997
29.

Lipowska M., Patonay G., Strekowski L.
Synthetic Communications,
1993
30.

Gayton J., Autry S., Fortenberry R., Hammer N., Delcamp J.
Molecules,
2018
31.

Berredjem M., Bouchareb F., Kaki S.A., Dekhil M., Aouf N.
Arabian Journal of Chemistry,
2017
32.
![[66] Acetylation](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Riordan J.F., Vallee B.L.
Methods in Enzymology,
1967
33.

Trávnícek M., Potácek M.
Molecules,
1999
34.
10.1016/j.mencom.2021.01.021_bib0075
Massah
Turk. J. Chem.,
2007
35.

Song F., Peng X., Lu E., Zhang R., Chen X., Song B.
Journal of Photochemistry and Photobiology A: Chemistry,
2004
36.

Levitz A., Marmarchi F., Henary M.
Molecules,
2018