Keywords
N-dealkylation
allylic amines
hydride transfer
methylalumoxane
propargylic amines
zinc iodide
Abstract
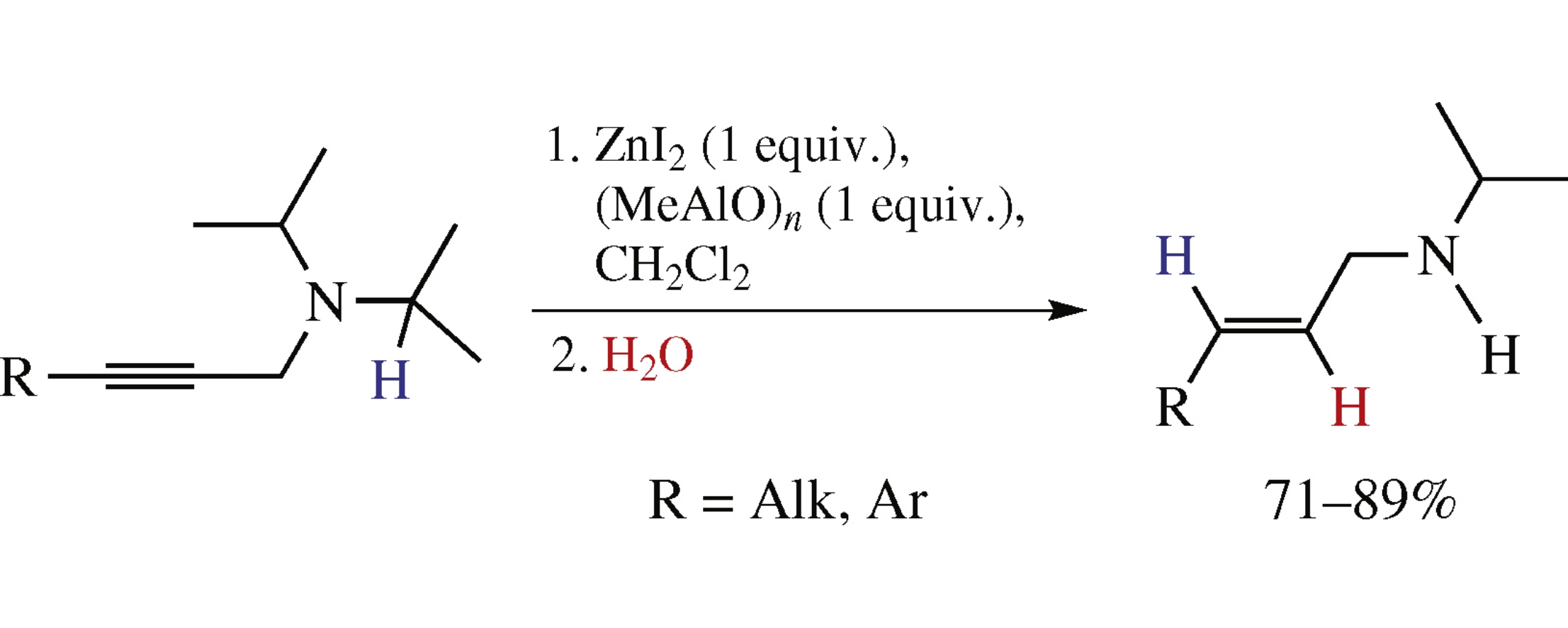
The reaction of N,N-diisopropyl-N-propargylamines with Et3Al/MXn system (MXn=CuCl, CuBr, ZnCl2, ZnI2, CdI2, TiCl4, ZrCl4, VCl5, TaCl5) in CH2Cl2 proceeds as an intramolecular red-ox hydride transfer affording N-dealkylated N-isopropyl-N-allylamines, allenes being formed as side products. The highest chemoselectivities and yields (71–89%) were attained by the application of new ZnI2/(MeAlO)n system.
References
1.

Ramazanov I., Yaroslavova A., Yaubasarov N., Dzhemilev U.
Synlett,
2017
2.

Ramazanov I.R., Kadikova R.N., Saitova Z.R., Dzhemilev U.M.
Russian Chemical Bulletin,
2020
3.

4.

Kuang J., Tang X., Ma S.
Organic Chemistry Frontiers,
2015
5.

Huang X., Ma S.
Accounts of Chemical Research,
2019
6.

Liu Q., Tang X., Cai Y., Ma S.
Organic Letters,
2017
7.

Tang X., Zhu C., Cao T., Kuang J., Lin W., Ni S., Zhang J., Ma S.
Nature Communications,
2013
8.

Crabbé P., Fillion H., André D., Luche J.
Journal of the Chemical Society Chemical Communications,
1979
9.

Kuang J., Ma S.
Journal of Organic Chemistry,
2009
10.
Kadikova G.N., D’yakonov V.A., Nasretdinov R.N., Dzhemileva L.U., Dzhemilev U.M.
Mendeleev Communications,
2020