Keywords
1,2,5-oxadiazoles
dinitrogen tetraoxide
NO-donors
oxidative cyclization
pharmacologically oriented compounds
pyrazolo[3,4-d]pyridazine 5,6-dioxides
Abstract
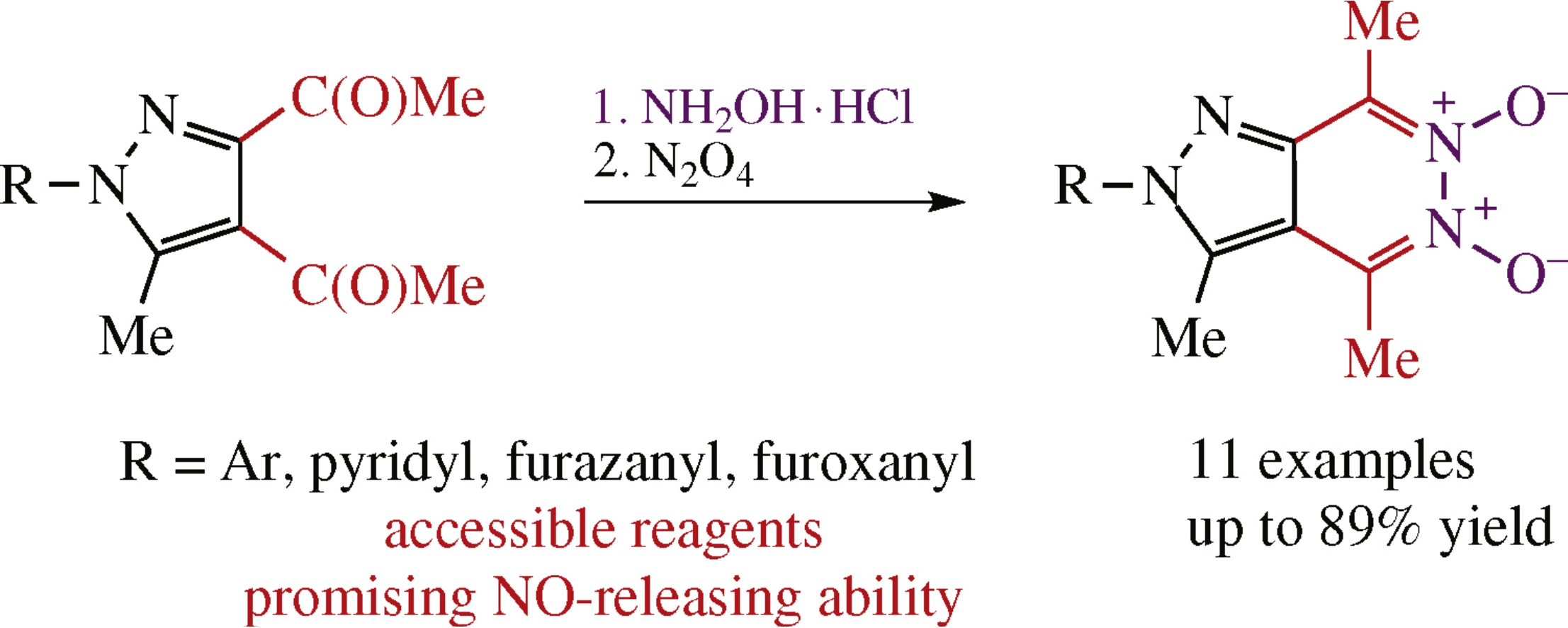
A simple and effective protocol for the synthesis of 2H-pyrazolo[3,4-d]pyridazine 5,6-dioxides includes a transformation of accessible 3,4-diacetyl-5-methyl-1H-pyrazoles into the corresponding 1,4-dioximes followed by their N2O4- mediated oxidative cyclization. All transformations proceed under mild conditions in good to high yields. According to the Griess assay, synthesized 2H-pyrazolo[3,4-d]pyridazine 5,6-dioxides showed promising NO-donor profiles producing NO in a wide range of concentrations.
References
1.

Jadala C., Sathish M., Anchi P., Tokala R., Lakshmi U.J., Reddy V.G., Shankaraiah N., Godugu C., Kamal A.
ChemMedChem,
2019
2.

Ivasiv V., Albertini C., Gonçalves A.E., Rossi M., Bolognesi M.L.
Current Topics in Medicinal Chemistry,
2019
3.

Harrison J.R., Brand S., Smith V., Robinson D.A., Thompson S., Smith A., Davies K., Mok N., Torrie L.S., Collie I., Hallyburton I., Norval S., Simeons F.R., Stojanovski L., Frearson J.A., et. al.
Journal of Medicinal Chemistry,
2018
4.
Zyryanov G.V., Kopchuk D.S., Kovalev I.S., Santra S., Rahman M., Khasanov A.F., Krinochkin A.P., Taniya O.S., Chupakhin O.N., Charushin V.N.
Mendeleev Communications,
2020
5.
10.1016/j.mencom.2021.01.012_sbref0010a
Granik
Oksid azota (NO): Novyi put’ k poisku lekarstv (Nitric Oxide (NO): New Way to Drug Discovery),
2004
6.

Willmot M., Gray L., Gibson C., Murphy S., Bath P.M.
Nitric Oxide - Biology and Chemistry,
2005
7.
10.1016/j.mencom.2021.01.012_sbref0010c
Nitric Oxide Donors: For Pharmaceutical and Biological Applications,
2005
8.

Krause P., Wätzig E., Acil H., König S., Unthan-Fechner K., Tsikas D., Probst I.
Nitric Oxide - Biology and Chemistry,
2010
9.

Serafim R.A., Pernichelle F.G., Ferreira E.I.
Expert Opinion on Drug Discovery,
2017
10.

Zhilin E.S., Fershtat L.L., Bystrov D.M., Kulikov A.S., Dmitrienko A.O., Ananyev I.V., Makhova N.N.
European Journal of Organic Chemistry,
2019
11.
Larin A.A., Fershtat L.L., Ustyuzhanina N.E., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2018
12.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2018
13.

Makhova N.N., Fershtat L.L.
Tetrahedron Letters,
2018
14.

Fershtat L.L., Makhova N.N.
Russian Chemical Reviews,
2016
15.
Kulikov A.S., Larin A.A., Fershtat L.L., Anikina L.V., Pukhov S.A., Klochkov S.G., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Arkivoc,
2017
16.
10.1016/j.mencom.2021.01.012_sbref0015g
Zhilin
ChemPhotoChem,
2020
17.

Fershtat L.L., Makhova N.N.
ChemMedChem,
2017
18.

Matsubara R., Takazawa S., Ando A., Hayashi M., Tohda R., Tsubaki M.
Asian Journal of Organic Chemistry,
2017
19.

Seymour C.P., Tohda R., Tsubaki M., Hayashi M., Matsubara R.
Journal of Organic Chemistry,
2017
20.

Matsubara R., Ando A., Hayashi M.
Tetrahedron Letters,
2017
21.

Makhova N.N., Belen’kii L.I., Gazieva G.A., Dalinger I.L., Konstantinova L.S., Kuznetsov V.V., Kravchenko A.N., Krayushkin M.M., Rakitin O.A., Starosotnikov A.M., Fershtat L.L., Shevelev S.A., Shirinian V.Z., Yarovenko V.N.
Russian Chemical Reviews,
2020
22.

Lazzarato L., Cena C., Rolando B., Marini E., Lolli M.L., Guglielmo S., Guaita E., Morini G., Coruzzi G., Fruttero R., Gasco A.
Bioorganic and Medicinal Chemistry,
2011
23.

Borretto E., Lazzarato L., Spallotta F., Cencioni C., D’Alessandra Y., Gaetano C., Fruttero R., Gasco A.
ACS Medicinal Chemistry Letters,
2013
24.

Guglielmo S., Cortese D., Vottero F., Rolando B., Kommer V.P., Williams D.L., Fruttero R., Gasco A.
European Journal of Medicinal Chemistry,
2014
25.

Zhilin E.S., Bystrov D.M., Ananyev I.V., Fershtat L.L., Makhova N.N.
Chemistry - A European Journal,
2019
26.
![Vasorelaxant and antiplatelet activity of 4,7-dimethyl-1,2,5-oxadiazolo[3,4-d]pyridazine 1,5,6-trioxide: Role of soluble guanylate cyclase, nitric oxide and thiols](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kots A.Y., Grafov M.A., Khropov Y.V., Betin V.L., Belushkina N.N., Busygina O.G., Yazykova M.Y., Ovchinnikov I.V., Kulikov A.S., Makhova N.N., Medvedeva N.A., Bulargina T.V., Severina I.S.
British Journal of Pharmacology,
2000
27.

NAKADATE M., SUEYOSHI S., SUZUKI I.
Chemical and Pharmaceutical Bulletin,
2011
28.

Rozen S., Shaffer A.
Organic Letters,
2017
29.
10.1016/j.mencom.2021.01.012_sbref0050a
Spyroudis
Synthesis,
1976
30.

Igeta H., Ohsawa A., Arai H.
Heterocycles,
1978
31.

Ohsawa A., Arai H., Igeta H., Akimoto T., Tsuji A., Iitaka Y.
Journal of Organic Chemistry,
1979
32.
![[1,2,5]Oxadiazolo[3,4-d]pyridazine 1,5,6-trioxides: efficient synthesis via the reaction of 3,4-bis(hydroxyimino)methyl)-1,2,5-oxadiazole 2-oxides with a mixture of concentrated nitric and trifluoroacetic acids and structural characterization](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ogurtsov V.A., Dorovatovskii P.V., Zubavichus Y.V., Khrustalev V.N., Fakhrutdinov A.N., Zlotin S.G., Rakitin O.A.
Tetrahedron Letters,
2018
33.
![Synthesis and structural investigation of 4,4′-dimethyl-[3,3′-bi(1,2,5-oxadiazole)] 5,5′-dioxide](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Obruchnikova N.V., Novikov R.A., Zlotin S.G., Dorovatovskii P.V., Khrustalev V.N., Rakitin O.A.
Russian Chemical Bulletin,
2018
34.

Ivanova O.A., Averina E.B., Kuznetsova T.S., Zefirov N.S.
Chemistry of Heterocyclic Compounds,
2000
35.
10.1016/j.mencom.2021.01.012_sbref0055d
Fruttero
Eur. J. Org. Chem.,
1988
36.

Ansari A., Ali A., Asif M., Shamsuzzaman S.
New Journal of Chemistry,
2017
37.
![Design, Synthesis, and Physicochemical and Pharmacological Profiling of 7-Hydroxy-5-oxopyrazolo[4,3-b]pyridine-6-carboxamide Derivatives with Antiosteoarthritic Activity In Vivo](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Mugnaini C., Kostrzewa M., Bryk M., Mahmoud A.M., Brizzi A., Lamponi S., Giorgi G., Ferlenghi F., Vacondio F., Maccioni P., Colombo G., Mor M., Starowicz K., Di Marzo V., Ligresti A., et. al.
Journal of Medicinal Chemistry,
2020
38.
Epishina M.A., Kulikov A.S., Fershtat L.L., Ananyev I.V., Makhova N.N.
Mendeleev Communications,
2019
39.

Althagafi I., Elghalban M.G., Saad F., Al-Fahemi J.H., El-Metwaly N.M., Bondock S., Almazroai L., Saleh K.A., Al-Hazmi G.A.
Journal of Molecular Liquids,
2017
40.

Shawali A.S., Haboub A.J.
Journal of Heterocyclic Chemistry,
2013
41.

Abdel-Galil F., Sami Shawali A., Mahmoud Hassaneen H., Shetta A., Osman A.
Heterocycles,
1982
42.

Sami Shawali A., Mohmoud Hassaneen H., Osman Abdelhamid A., Pagni R.
Heterocycles,
1982
43.

Naidek K.P., Hoffmeister D.M., Pazinato J., Westphal E., Gallardo H., Nakamura M., Araki K., Toma H.E., Winnischofer H.
European Journal of Inorganic Chemistry,
2014