Keywords
condensation
cyanopyrimidine N-oxides
cytotoxic activity
fluorescent properties
quantum chemical calculations
tetrahydroquinazolines
Abstract
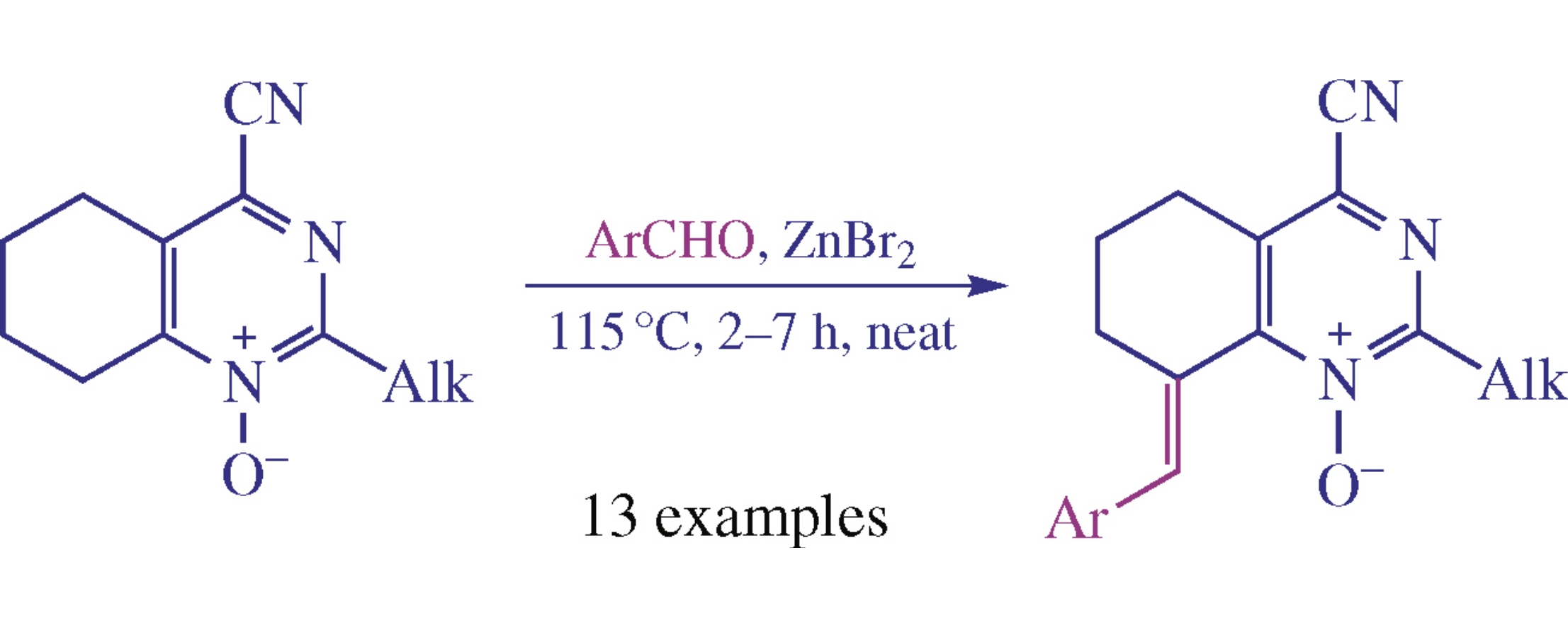
New 8-arylidene-4-cyanotetrahydroquinazoline N-oxides were obtained via the condensation of readily available 4-cyanotetrahydroquinazoline N-oxides with aromatic aldehydes. Some of the synthesized compounds revealed fluorescent properties and moderate cytotoxic activity.
References
1.

Shang X., Morris-Natschke S.L., Liu Y., Guo X., Xu X., Goto M., Li J., Yang G., Lee K.
Medicinal Research Reviews,
2017
2.

Shang X., Morris-Natschke S.L., Yang G., Liu Y., Guo X., Xu X., Goto M., Li J., Zhang J., Lee K.
Medicinal Research Reviews,
2018
3.

Hameed A., Al-Rashida M., Uroos M., Ali S.A., Arshia, Ishtiaq M., Khan K.M.
Expert Opinion on Therapeutic Patents,
2018
4.

Shagufta S., Ahmad I.
MedChemComm,
2017
5.

Long S., Resende D.I., Kijjoa A., Silva A.M., Pina A., Fernández-Marcelo T., Vasconcelos M.H., Sousa E., Pinto M.M.
Marine Drugs,
2018
6.
10.1016/j.mencom.2021.01.011_bib0030
Selvam
Res. Pharm.,
2011
7.
Solyanik G.I.
Experimental Oncology,
2019
8.
Soylem A.E., Assy G.M., Morsi M.G.
Acta Chimica Slovenica,
2016
9.

El-Subbagh H.I., Hassan G.S., El-Messery S.M., Al-Rashood S.T., Al-Omary F.A., Abulfadl Y.S., Shabayek M.I.
European Journal of Medicinal Chemistry,
2014
10.

Behera R.K., Behera A.K., Pradhan R., Pati A., Patra M.
Synthetic Communications,
2006
11.

Singh N., Pandey S.K., Anand N., Dwivedi R., Singh S., Sinha S.K., Chaturvedi V., Jaiswal N., Srivastava A.K., Shah P., Siddiqui M.I., Tripathi R.P.
Bioorganic and Medicinal Chemistry Letters,
2011
12.

Fathalla M., Shehta W., Assay M.G., Abd Alsalam E.
Research on Chemical Intermediates,
2018
13.

Majumdar P., Mohanta P.P., Sahu S., Behera A.K.
Synthetic Communications,
2018
14.

Raghav N., Jangra S., Kumar A., Bhattacharyya S.
International Journal of Biological Macromolecules,
2017
15.

Meng Q., Xu H., Xu J., Hao G., Gao X., Hu X., Rong L., Cai P.
Journal of Heterocyclic Chemistry,
2016
16.

Zakeri M., Nasef M.M., Abouzari-Lotf E.
Journal of Molecular Liquids,
2014
17.
Deli J., Lóránd T., Szabó D., Földesi A., Zschunke A.
Collection of Czechoslovak Chemical Communications,
2010
18.

Sevenard D.V., Khomutov O.G., Koryakova O.V., Sattarova V.V., Kodess M.I., Stelten J., Loop I., Lork E., Pashkevich K.I., Röschenthaler G.
Synthesis,
2000
19.

Sedenkova K.N., Averina E.B., Grishin Y.K., Kutateladze A.G., Rybakov V.B., Kuznetsova T.S., Zefirov N.S.
Journal of Organic Chemistry,
2012
20.

Sedenkova K.N., Averina E.B., Grishin Y.K., Bacunov A.B., Troyanov S.I., Morozov I.V., Deeva E.B., Merkulova A.V., Kuznetsova T.S., Zefirov N.S.
Tetrahedron Letters,
2015
21.

Sedenkova K.N., Dueva E.V., Averina E.B., Grishin Y.K., Osolodkin D.I., Kozlovskaya L.I., Palyulin V.A., Savelyev E.N., Orlinson B.S., Novakov I.A., Butov G.M., Kuznetsova T.S., Karganova G.G., Zefirov N.S.
Organic and Biomolecular Chemistry,
2015
22.

Bis(gem-dihalocyclopropanes): synthesis and heterocyclization upon treatment with nitronium triflate
Sedenkov K.N., Averin E.B., Grishin Y.K., Kuznetsova T.S., Zefirov N.S.
Russian Chemical Bulletin,
2016
23.

Sedenkova K.N., Averina E.B., Grishin Y.K., Kolodyazhnaya J.V., Rybakov V.B., Vasilenko D.A., Steglenko D.V., Minkin V.I., Kuznetsova T.S., Zefirov N.S.
Tetrahedron Letters,
2017
24.

Sedenkova K.N., Averina E.B., Grishin Y.K., Kolodyazhnaya J.V., Rybakov V.B., Kuznetsova T.S., Hughes A., Gomes G.D., Alabugin I.V., Zefirov N.S.
Organic and Biomolecular Chemistry,
2017
25.
Sedenkova K.N., Averina E.B., Nazarova A.A., Grishin Y.K., Karlov D.S., Zamoyski V.L., Grigoriev V.V., Kuznetsova T.S., Palyulin V.A.
Mendeleev Communications,
2018
26.
Sedenkova K.N., Nazarova A.A., Khvatov E.V., Dueva E.V., Orlov A.A., Osolodkin D.I., Grishin Y.K., Kuznetsova T.S., Palyulin V.A., Averina E.B.
Mendeleev Communications,
2018
27.

Sedenkova K.N., Kolodyazhnaya J.V., Vasilenko D.A., Gracheva Y.A., Kharitonoshvili E.V., Grishin Y.K., Chistov A.A., Rybakov V.B., Holt T., Kutateladze A.G., Kuznetsova T.S., Milaeva E.R., Averina E.B.
Dyes and Pigments,
2019
28.
Nazarova A.A., Sedenkova K.N., Vasilenko D.A., Grishin Y.K., Kuznetsova T.S., Averina E.B.
Mendeleev Communications,
2020
29.

Sedenkova K.N., Terekhin A.V., Abdrashitova I.V., Vasilenko D.A., Sadovnikov K.S., Gracheva Y.A., Grishin Y.K., Holt T., Kutateladze A.G., Kuznetsova T.S., Milaeva E.R., Averina E.B.
Tetrahedron Letters,
2020
30.

Sedenkova K.N., Averina E.B., Grishin Y.K., Kuznetsova T.S., Zefirov N.S.
Tetrahedron Letters,
2014
31.

Neese F.
Wiley Interdisciplinary Reviews: Computational Molecular Science,
2017