Keywords
diazo compounds
diazo transfer
maleimides
Michael acceptors
parallel synthesis
privileged structures
Wittig reaction
Abstract
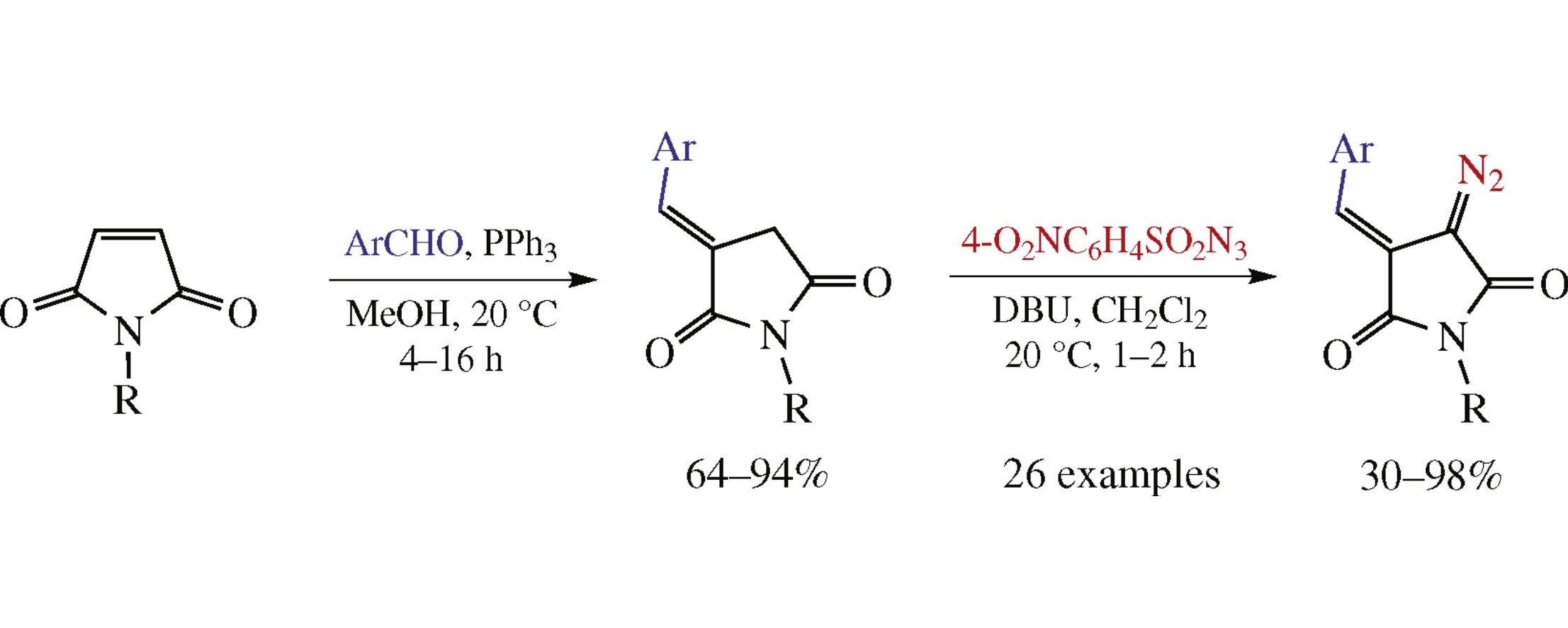
A practically convenient synthesis of (E)-3-arylidene-4-diazopyrrolidine-2,5-diones from N-substituted maleimides via the Wittig reaction and the Regitz diazo transfer has been developed. In all 26 cases studied, only one chromatographic purification was required with no need for aqueous workup, which makes this protocol amenable to producing the emerging class of diazo compounds in parallel format.
References
1.

Baell J.B., Nissink J.W.
ACS Chemical Biology,
2017
2.

Jackson P.A., Widen J.C., Harki D.A., Brummond K.M.
Journal of Medicinal Chemistry,
2016
3.

Bauer R.A.
Drug Discovery Today,
2015
4.

Baell J., Walters M.A.
Nature,
2014
5.

Deng X., Kong L., Zhao Y., He J., Peng L., Li Y., Zhao Q.
Natural Products and Bioprospecting,
2012
6.

Luo K., Bao Y., Liu F., Xiao C., Li K., Zhang C., Huang R., Lin J., Zhang J., Jin Y.
European Journal of Medicinal Chemistry,
2019
7.

Shalaby E.M., Girgis A.S., Farag H., Mabied A.F., Fitch A.N.
RSC Advances,
2016
8.

Maga G., Falchi F., Radi M., Botta L., Casaluce G., Bernardini M., Irannejad H., Manetti F., Garbelli A., Samuele A., Zanoli S., Esté J.A., Gonzalez E., Zucca E., Paolucci S., et. al.
ChemMedChem,
2011
9.

Kalia D., Malekar P.V., Parthasarathy M.
Angewandte Chemie - International Edition,
2015
10.

Laha D., Bhat R.G.
Asian Journal of Organic Chemistry,
2020
11.
E. Chupakhin, M. Gecht, A. Ivanov, G. Kantin, D. Dar’in and M. Krasavin, Synthesis, in press.
12.

Hedaya E., Theodoropulos S.
Tetrahedron,
1968
13.

Bulletin of the Korean Chemical Society,
2004
14.

15.
https://www.molinspiration.com/.
16.
Zhmurov P.A., Dar’in D.V., Bakulina O.Y., Krasavin M.
Mendeleev Communications,
2020