Keywords
clathrochelates
electrocatalyst
hydrogen evolution reaction
macrocyclic compounds
polymer electrolyte membrane
water electrolysis
Abstract
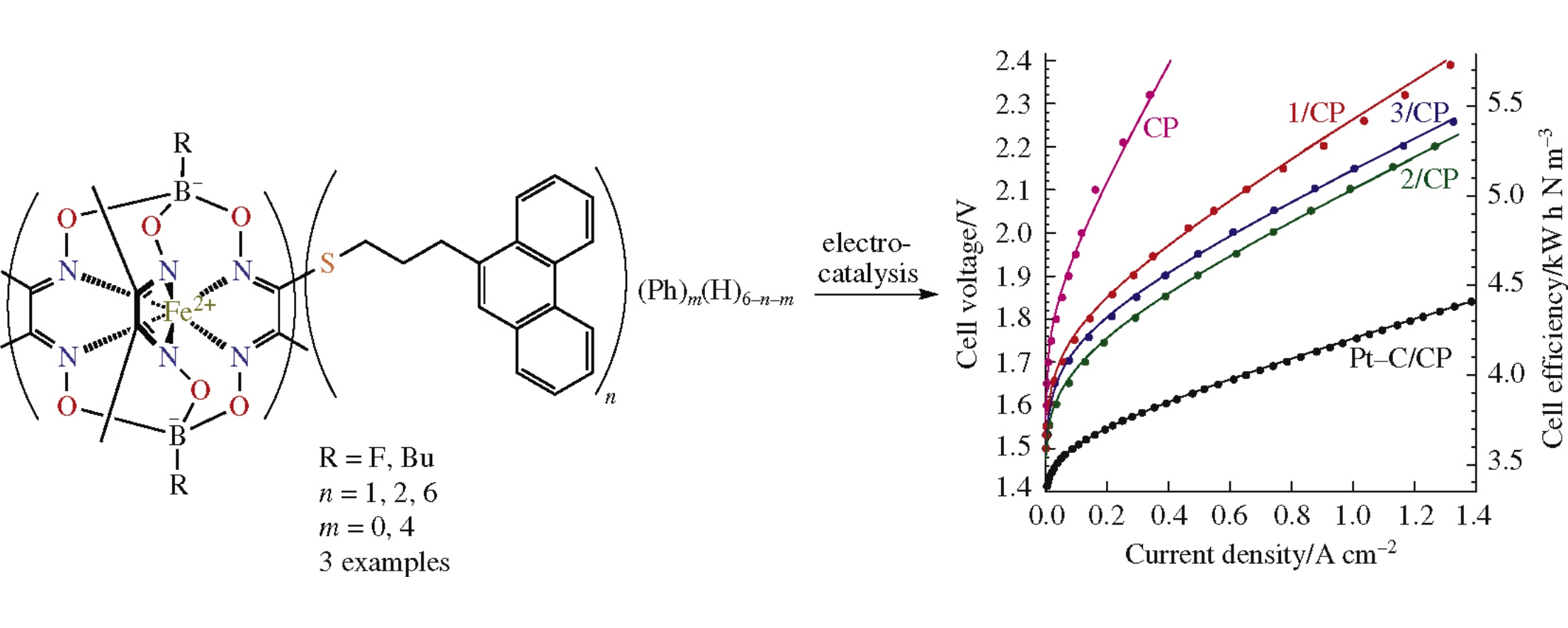
Hydrogen-evolving cathodes were prepared using a series of iron(ii) clathrochelates bearing various number of terminal phenanthrenyl groups via physisorption on carbon paper and employed in the polymer electrolyte membrane water electrolysis cells instead of typically used platinum. In situ electrochemical activation of the cathodes was carried out, after that the cells performance and durability were evaluated. These clathrochelate complexes represent a promising alternative to platinum as hydrogen-evolving cathode electrocatalysts.
References
1.

Ayers K.
Current Opinion in Electrochemistry,
2019
2.

Vincent I., Bessarabov D.
Renewable and Sustainable Energy Reviews,
2018
3.

Zhang B., Fan L., Ambre R.B., Liu T., Meng Q., Timmer B.J., Sun L.
Joule,
2020
4.

Sun X., Xu K., Fleischer C., Liu X., Grandcolas M., Strandbakke R., Bjørheim T., Norby T., Chatzitakis A.
Catalysts,
2018
5.

Liu Q., Liu X., Zheng L., Shui J.
Angewandte Chemie - International Edition,
2018
6.
10.1016/j.mencom.2021.01.005_bib0030
Voloshin
Cage Metal Complexes. Clathrochelates Revisited,
2017
7.

Pantani O., Naskar S., Guillot R., Millet P., Anxolabéhère-Mallart E., Aukauloo A.
Angewandte Chemie - International Edition,
2008
8.

Dolganov A.V., Belov A.S., Novikov V.V., Vologzhanina A.V., Mokhir A., Bubnov Y.N., Voloshin Y.Z.
Dalton Transactions,
2013
9.

Thoi V.S., Sun Y., Long J.R., Chang C.J.
Chemical Society Reviews,
2013
10.

Grigoriev S.A., Pushkarev A.S., Pushkareva I.V., Millet P., Belov A.S., Novikov V.V., Belaya I.G., Voloshin Y.Z.
International Journal of Hydrogen Energy,
2017
11.

Zelinskii G.E., Pavlov A.A., Belov A.S., Belaya I.G., Vologzhanina A.V., Nelyubina Y.V., Efimov N.N., Zubavichus Y.V., Bubnov Y.N., Novikov V.V., Voloshin Y.Z.
ACS Omega,
2017
12.

Anxolabéhère-Mallart E., Costentin C., Fournier M., Nowak S., Robert M., Savéant J.
Journal of the American Chemical Society,
2012
13.

Anxolabéhère-Mallart E., Costentin C., Fournier M., Robert M.
Journal of Physical Chemistry C,
2014
14.

Lassalle-Kaiser B., Zitolo A., Fonda E., Robert M., Anxolabéhère-Mallart E.
ACS Energy Letters,
2017
15.

Varzatskii O.A., Oranskiy D.A., Vakarov S.V., Chornenka N.V., Belov A.S., Vologzhanina A.V., Pavlov A.A., Grigoriev S.A., Pushkarev A.S., Millet P., Kalinichenko V.N., Voloshin Y.Z., Dedov A.G.
International Journal of Hydrogen Energy,
2017
16.

Voloshin Y.Z., Chornenka N.V., Varzatskii O.A., Belov A.S., Grigoriev S.A., Pushkarev A.S., Millet P., Kalinichenko V.N., Belaya I.G., Bugaenko M.G., Dedov A.G.
Electrochimica Acta,
2018
17.

Voloshin Y.Z., Chornenka N.V., Belov A.S., Grigoriev S.A., Pushkarev A.S., Millet P., Kalinichenko V.N., Oranskiy D.A., Dedov A.G.
Journal of the Electrochemical Society,
2019
18.

Pushkarev A.S., Solovyev M.A., Grigoriev S.A., Pushkareva I.V., Voloshin Y.Z., Chornenka N.V., Belov A.S., Millet P., Kalinichenko V.N., Dedov A.G.
International Journal of Hydrogen Energy,
2020
19.

Alia S.M., Anderson G.C.
Journal of the Electrochemical Society,
2019
20.

Siracusano S., Baglio V., Di Blasi A., Briguglio N., Stassi A., Ornelas R., Trifoni E., Antonucci V., Aricò A.S.
International Journal of Hydrogen Energy,
2010
21.

Weiß A., Siebel A., Bernt M., Shen T.-., Tileli V., Gasteiger H.A.
Journal of the Electrochemical Society,
2019
22.

Ito H., Maeda T., Nakano A., Kato A., Yoshida T.
Electrochimica Acta,
2013
23.

Voloshin Y.Z., Buznik V.M., Dedov A.G.
Pure and Applied Chemistry,
2020
24.

Carmo M., Fritz D.L., Mergel J., Stolten D.
International Journal of Hydrogen Energy,
2013
25.

Bender G., Carmo M., Smolinka T., Gago A., Danilovic N., Mueller M., Ganci F., Fallisch A., Lettenmeier P., Friedrich K.A., Ayers K., Pivovar B., Mergel J., Stolten D.
International Journal of Hydrogen Energy,
2019
26.
10.1016/j.mencom.2021.01.005_bib0130
Bockris
Modern Electrochemistry. An Introduction to an Interdisciplinary Area,
1970
27.

Lettenmeier P., Wang L., Golla-Schindler U., Gazdzicki P., Cañas N.A., Handl M., Hiesgen R., Hosseiny S.S., Gago A.S., Friedrich K.A.
Angewandte Chemie - International Edition,
2015
28.

Oh H., Nong H.N., Reier T., Gliech M., Strasser P.
Chemical Science,
2015
29.

Deng J., Ren P., Deng D., Yu L., Yang F., Bao X.
Energy and Environmental Science,
2014
30.

Tavakkoli M., Kallio T., Reynaud O., Nasibulin A.G., Johans C., Sainio J., Jiang H., Kauppinen E.I., Laasonen K.
Angewandte Chemie - International Edition,
2015
31.

Zubavichus Y.V., Grigor’ev S.A., Pushkarev A.S., Borisov M.M., Bugaenko M.G., Voloshin Y.Z., Dedov A.G.
Nanotechnologies in Russia,
2020
32.
Dedov A.G., Voloshin Y.Z., Belov A.S., Loktev A.S., Bespalov A.S., Buznik V.M.
Mendeleev Communications,
2019
33.

Shinagawa T., Garcia-Esparza A.T., Takanabe K.
Scientific Reports,
2015
34.

Al Cheikh J., Villagra A., Ranjbari A., Pradon A., Antuch M., Dragoe D., Millet P., Assaud L.
Applied Catalysis B: Environmental,
2019
35.

Anantharaj S., Ede S.R., Karthick K., Sam Sankar S., Sangeetha K., Karthik P.E., Kundu S.
Energy and Environmental Science,
2018
36.

Aßmann P., Gago A.S., Gazdzicki P., Friedrich K.A., Wark M.
Current Opinion in Electrochemistry,
2020
37.

Kim J., Kim J., Kim H., Ahn S.H.
ACS applied materials & interfaces,
2019
38.

Ng J.W., Hellstern T.R., Kibsgaard J., Hinckley A.C., Benck J.D., Jaramillo T.F.
ChemSusChem,
2015
39.

Kim J.H., Kim H., Kim J., Lee H.J., Jang J.H., Ahn S.H.
Journal of Power Sources,
2018