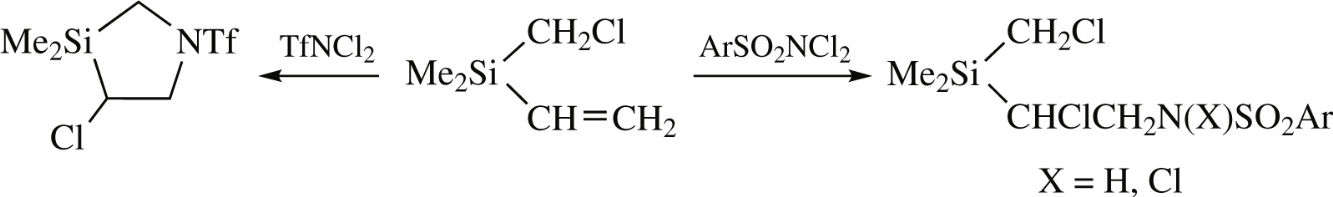Keywords
N,N-dichloro sulfonamides
1,3-azasilolidines
chloroalkyl(dimethyl)vinylsilanes
chloroamidation
heterocyclization
Abstract
The reactions of N,N-dichloroarenesulfonamides with chloromethyl(dimethyl)vinylsilane or of N,N-dichlorotrifluoromethanesulfonamide with chloropropyl(dimethyl)vinylsilanegive the NH- and NCl-containing products of chlorosulfonamidation of the double bonds. In contrast, N,N-(dichloro)trifluoromethanesulfonamide reacts with chloromethyl(dimethyl)vinylsilane to afford 4-chloro-3,3-dimethyl1-[(trifluoroethyl) sulfonyl]-1,3-azasilolidine via intramolecular heterocyclization.
References
1.
![Addition of pseudohalogens to vinyl- and allylsilanes. Synthesis of 1-substituted 2-(alkylsilyl)- and 2-[(alkylsilyl)methyl]aziridines using phase-transfer catalysis](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Lukevics E., Dirnens V., Gol'dberg Y.S., Liepins E., Gavars M., Kalvins I., Shymanskaya M.V.
Organometallics,
1985
2.

Thakur V.V., Talluri S.K., Sudalai A.
Organic Letters,
2003
3.

Landais Y., Zekri E.
European Journal of Organic Chemistry,
2002
4.

Astakhova V.V., Shainyan B.A., Moskalik M.Y., Sterkhova I.V.
Tetrahedron,
2019
5.
Ushakova I.V., Shainyan B.A.
Mendeleev Communications,
2020
6.

Lazareva N.F., Lazarev I.M.
Russian Chemical Bulletin,
2015
7.

Fujii S., Hashimoto Y.
Future Medicinal Chemistry,
2017
8.

Ganin A.S., Moskalik M.Y., Astakhova V.V., Sterkhova I.V., Shainyan B.A.
Tetrahedron,
2020
9.
10.1016/j.mencom.2020.11.035_bib0045
Nazaretyan
Zh. Org. Khim.,
1974
10.

Kirpichenko S.V., Abrosimova A.T., Albanov A.I., Voronkov M.G.
Russian Journal of General Chemistry,
2001