Keywords
1-azaxanthones
3-formylchromones
alkylidenemalononitriles
heterocyclization
hydroxylamine hydrochloride
Abstract
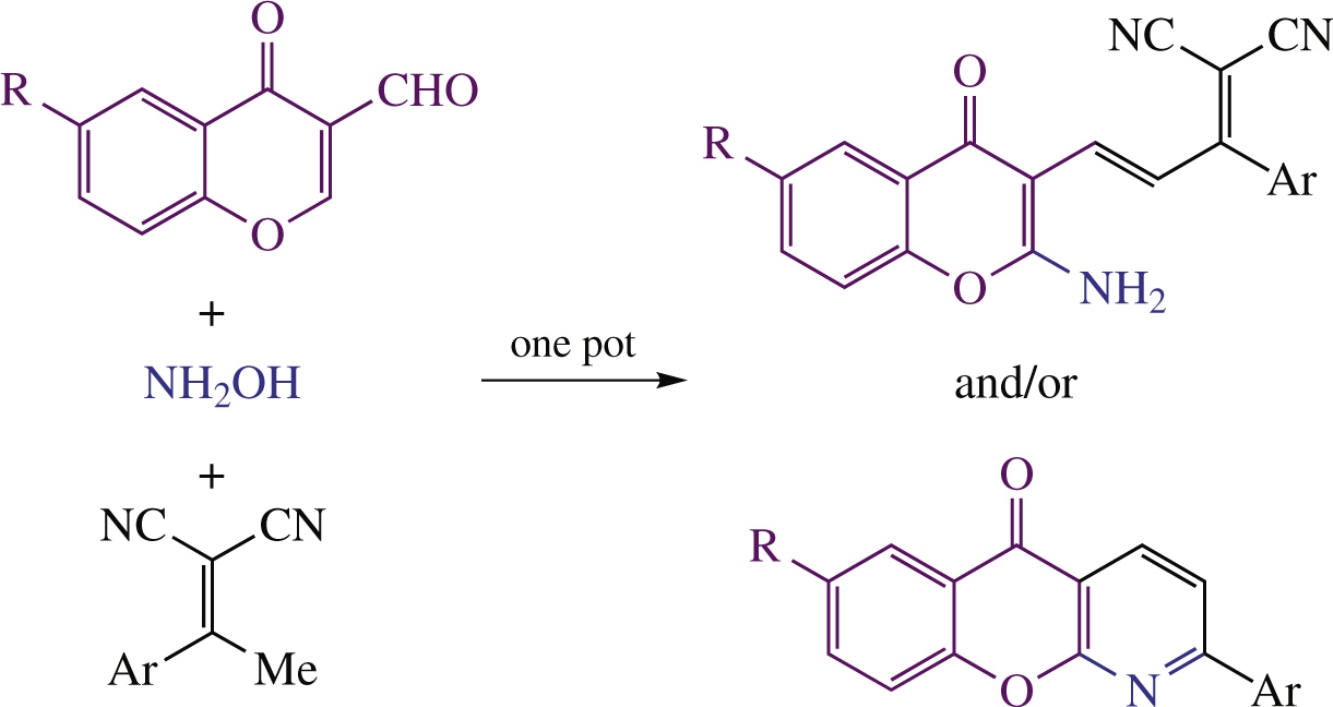
An efficient one-pot synthesis of 2-[3-(2-amino-4-oxo-4Hchromen-3-yl)-1-arylallylidene]malononitriles as intermediates and 2-aryl-5H-chromeno[2,3-b]pyridin-5-ones as final products comprises Et3N-catalyzed condensation of alkylidenemalononitriles, 3-formylchromones and hydroxylamine hydrochloride. The transformation involves stages of 1,4-nucleophilic addition, phenolate–nitrile coupling and Mannich-type heterocyclization reactions.
References
1.
![Synthesis, X-ray Structure, and Pharmacological Activity of Some 6,6-Disubstituted Chromeno[4,3-b]- and Chromeno- [3,4-c]-quinolines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Hegab M.I., Abdel-Fattah A.M., Yousef N.M., Nour H.F., Mostafa A.M., Ellithey M.
Archiv der Pharmazie,
2007
2.
![Chromeno[3,4-c]pyridin-5-ones: Selective Human Dopamine D4 Receptor Antagonists as Potential Antipsychotic Agents](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Unangst P.C., Capiris T., Connor D.T., Heffner T.G., MacKenzie R.G., Miller S.R., Pugsley T.A., Wise L.D.
Journal of Medicinal Chemistry,
1997
3.

Giannouli V., Kostakis I.K., Pouli N., Marakos P., Kousidou O.C., Tzanakakis G.N., Karamanos N.K.
Journal of Medicinal Chemistry,
2007
4.

Rashid M.A., Rasool N., Appel B., Adeel M., Karapetyan V., Mkrtchyan S., Reinke H., Fischer C., Langer P.
Tetrahedron,
2008
5.

Nohara A., Kuriki H., Saijo T., Sugihara H., Kanno M., Sanno Y.
Journal of Medicinal Chemistry,
1977
6.
![Synthesis and anti-inflammatory activities of 4H-chromene and chromeno[2,3-b]pyridine derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Chung S., Huang W., Huang C., Liu F., Huang R., Wu C., Lee A.
Research on Chemical Intermediates,
2015
7.

KUBO K., UKAWA K., KUZUNA S., NOHARA A.
Chemical and Pharmaceutical Bulletin,
2011
8.
![Studies on Antianaphylactic Agents. 7. Synthesis of Antiallergic 5-Oxo-5H-[1]benzopyrano[2,3-b]pyridines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Nohara A., Ishiguro T., Ukawa K., Sugihara H., Maki Y., Sanno Y.
Journal of Medicinal Chemistry,
1985
9.
![Synthesis of 1,2,3,4-tetrahydro-5H-[1]benzopyrano[3,4-c]pyridin-5-ones. II. Substitution at the 3-position with 2-aminoethyl and 2-aminopropyl side chains](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Connor D.T., Unangst P.C., Schwender C.F., Sorenson R.J., Carethers M.E., Puchalski C., Brown R.E.
Journal of Heterocyclic Chemistry,
1984
10.

Marquise N., Harford P.J., Chevallier F., Roisnel T., Dorcet V., Gagez A., Sablé S., Picot L., Thiéry V., Wheatley A.E., Gros P.C., Mongin F.
Tetrahedron,
2013
11.

Dubrovskiy A.V., Larock R.C.
Organic Letters,
2010
12.

Ibad M.F., Abid O., Adeel M., Nawaz M., Wolf V., Villinger A., Langer P.
Journal of Organic Chemistry,
2010
13.

Sosnovskikh V.Y., Irgashev R.A., Demkovich I.A.
Russian Chemical Bulletin,
2008
14.

Villani F.J., Mann T.A., Wefer E.A., Hannon J., Larca L.L., Landon M.J., Spivak W., Vashi D., Tozzi S.
Journal of Medicinal Chemistry,
1975
15.
![Novel Chromeno[2,3-b]pyridines from Basic Rearrangement of 4-Oxo-chromene-3-carbonitrile](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Ibrahim M.A.
Synthetic Communications,
2009
16.
![Efficient synthesis of benzopyrano[2,3-b]pyridines by sequential reactions of 1,3-bis-silyl enol ethers with 3-cyanobenzopyrylium triflates](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Langer P., Appel B.
Tetrahedron Letters,
2003
17.
Dimitriadou E., Raftopoulou M., Kasapidou P.M., Tsoleridis C.A., Stephanidou-Stephanatou J., Hadjipavlou-Litina D.J., Kontogiorgis C., Pritsa A., Papadopoulos A.
Arkivoc,
2014
18.

Barnes D.M., Haight A.R., Hameury T., McLaughlin M.A., Mei J., Tedrow J.S., Riva Toma J.D.
Tetrahedron,
2006
19.
![An Unexpected Phosphine-Catalyzed [3 + 2] Annulation. Synthesis of Highly Functionalized Cyclopentenes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Lu Z., Zheng S., Zhang X., Lu X.
Organic Letters,
2008
20.

Panwar R., Shally S., Shaw R., Elagamy A., Pratap R.
Organic and Biomolecular Chemistry,
2018
21.

Reddy G.J., Latha D., Thirupathaiah C., Rao K.S.
Organic Preparations and Procedures International,
2004
22.

Waldmann H., Karunakar G.V., Kumar K.
Organic Letters,
2008
23.

Liu X., Chen P., Wang B., Li Y., Wang S., Li Z.
Bioorganic and Medicinal Chemistry Letters,
2007
24.

Korotaev V.Y., Barkov A.Y., Kutyashev I.B., Safrygin A.V., Sosnovskikh V.Y.
Tetrahedron,
2014