Keywords
Acetylene
aminophenols
nucleophilic addition
superbases
vinylation
vinyloxyanilines
Abstract
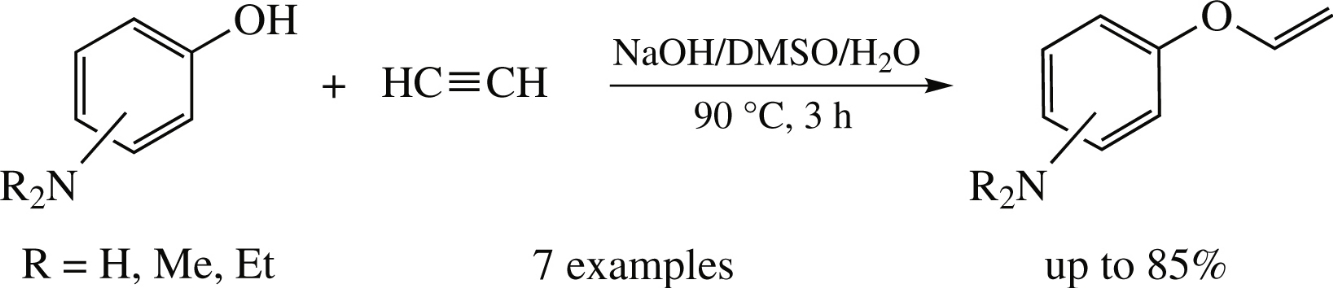
An efficient atom-economic chemoselective synthesis of vinyloxyanilines from aminophenols and acetylene catalyzed by sodium aminophenolates in aqueous DMSO (90°C, 3h) has been developed.
References
1.
10.1016/j.mencom.2020.11.033_sbref0005a
Jai
Chem. Pharm. Res.,
2018
2.

Alpuche-García A., Dávila-González X., Arregui L., Beltrán H.I.
RSC Advances,
2017
3.
![Reaction of bis[(2-chlorocarbonyl)phenyl] Diselenide with Phenols, Aminophenols, and Other Amines towards Diphenyl Diselenides with Antimicrobial and Antiviral Properties](/storage/images/resized/MjH1ITP7lMYGxeqUZfkt2BnVLgjkk413jwBV97XX_small_thumb.webp)
Giurg M., Gołąb A., Suchodolski J., Kaleta R., Krasowska A., Piasecki E., Piętka-Ottlik M.
Molecules,
2017
4.

Kinjo T., Tadokoro T., Tokushige A., Zamami T., Taira S., Ikehara Y., Tsuhako C., Ohtsu H., Ueda S., Kakinohana M.
Anesthesia and Analgesia,
2019
5.

Wasserman I., Poeran J., Zubizarreta N., Babby J., Serban S., Goldberg A.T., Greenstein A.J., Memtsoudis S.G., Mazumdar M., Leibowitz A.B.
Anesthesiology,
2018
6.

Ahmadi A., Khalili M., Ahmadian S., Shahghobadi N., Nahri-Niknafs B.
Pharmaceutical Chemistry Journal,
2014
7.

8.

Bushmakina I.M., Drozdova N.I., Golenko N.S., Martynova M.A.
Pharmaceutical Chemistry Journal,
2009
9.

Shadyro O., Ksendzova G., Polozov G., Sorokin V., Boreko E., Savinova O., Dubovik B., Bizunok N.
Bioorganic and Medicinal Chemistry Letters,
2008
10.

da França S., Dario M., Esteves V., Baby A., Velasco M.
Cosmetics,
2015
11.
Otutu J.O., Okoro D., Ossai E.K.
Journal of Applied Sciences,
2008
12.
10.1016/j.mencom.2020.11.033_sbref0020c
Focal Encyclopedia of Photography,
2007
13.
10.1016/j.mencom.2020.11.033_sbref0020d
Konopelnik
Mater. Sci.,
2002
14.
10.1016/j.mencom.2020.11.033_sbref0025a
Zhang
Organometallics,
2020
15.

Kolyvanov N.A., Oparina L.A., Tarasova O.A., Albanov A.I., Gusarova N.K., Trofimov B.A.
Tetrahedron Letters,
2020
16.

Unexpected acid-catalyzed ferrocenylmethylation of diverse nucleophiles with vinyloxymethylferrocene
Oparina L.A., Artem'ev A.V., Vysotskaya O.V., Tarasova O.A., Shagun V.A., Bagryanskaya I.Y., Trofimov B.A.
Tetrahedron,
2016
17.

Trofimov B., Oparina L., Gusarova N., Vysotskaya O., Artem’ev A., Kolyvanov N.
Synthesis,
2014
18.

Tunge J., Pahadi N.
Synlett,
2009
19.

20.

Martín Castro A.M.
Chemical Reviews,
2004
21.

Shimomoto H., Fukami D., Irita T., Katsukawa K., Nagai T., Kanaoka S., Aoshima S.
Journal of Polymer Science, Part A: Polymer Chemistry,
2012
22.
10.1016/j.mencom.2020.11.033_sbref0030c
Login
Vinyl Ether Monomers and Polymers. Kirk-Othmer Encyclopedia of Chemical Technology,
2000
23.

Shostakovskii M.F., Skvortsova G.G., Samoilova M.Y.
Polymer Science U S S R,
1964
24.

Kundu D., Maity P., Ranu B.C.
Organic Letters,
2014
25.

Jouvin K., Bayle A., Legrand F., Evano G.
Organic Letters,
2012
26.

27.

Willis M.C., Taylor D., Gillmore A.T.
Chemical Communications,
2003
28.

Wan Z., Jones C.D., Koenig T.M., Pu Y.J., Mitchell D.
Tetrahedron Letters,
2003
29.

McKinley N.F., O'Shea D.F.
Journal of Organic Chemistry,
2004
30.

Lam P.Y., Vincent G., Bonne D., Clark C.G.
Tetrahedron Letters,
2003
31.

Blouin M., Frenette R.
Journal of Organic Chemistry,
2001
32.

Okimoto Y., Sakaguchi S., Ishii Y.
Journal of the American Chemical Society,
2002
33.

Yu L., Tang N., Sheng S., Chen R., Liu X., Cai M.
Chinese Journal of Chemistry,
2012
34.

Zhang J., Sheng S., Liu X., Lin S.
Journal of Chemical Research,
2009
35.

Fu G., Yu L., Mao X., Wu D.
Journal of Chemical Research,
2008
36.
10.1016/j.mencom.2020.11.033_sbref0060d
Sheng
Chin. Chem. Lett.,
2005
37.

Teong S.P., Chua A.Y., Deng S., Li X., Zhang Y.
Green Chemistry,
2017
38.

Werner G., Rodygin K.S., Kostin A.A., Gordeev E.G., Kashin A.S., Ananikov V.P.
Green Chemistry,
2017
39.

Rattanangkool E., Vilaivan T., Sukwattanasinitt M., Wacharasindhu S.
European Journal of Organic Chemistry,
2016
40.
10.1016/j.mencom.2020.11.033_sbref0070a
Skvortsova
Zh. Org. Khim.,
1965
41.
10.1016/j.mencom.2020.11.033_sbref0070b
Mikhant’ev
Zh. Obshch. Khim.,
1964
42.
10.1016/j.mencom.2020.11.033_sbref0070c
Shostakovskii
Zh. Obshch. Khim.,
1961
43.
10.1016/j.mencom.2020.11.033_sbref0070d
Mikhant’ev
Zh. Obshch. Khim.,
1961
44.

Trofimov B.A., Oparina L.A., Kolyvanov N.A., Vysotskaya O.V., Gusarova N.K.
Russian Journal of Organic Chemistry,
2015
45.
B. A. Trofimov, N.K. Gusarova, L.A. Oparina, N.A. Kolyvanov and O. V. Vysotskaya, Patent RU 2640808, 2018.
46.

Yuan Y., Thomé I., Kim S.H., Chen D., Beyer A., Bonnamour J., Zuidema E., Chang S., Bolm C.
Advanced Synthesis and Catalysis,
2010
47.

Olmstead W.N., Margolin Z., Bordwell F.G.
Journal of Organic Chemistry,
1980
48.
Schmidt E.Y., Semenova N.V., Ivanova E.V., Bidusenko I.A., Trofimov B.A.
Mendeleev Communications,
2020
49.
Schmidt E.Y., Semenova N.V., Ivanova E.V., Ushakov I.A., Trofimov B.A.
Mendeleev Communications,
2020
50.
Orel V.B., Vitkovskaya N.M., Absalyamov D.Z., Schmidt E.Y., Trofimov B.A.
Mendeleev Communications,
2019