Keywords
2H-chromen-2-imines
Antibacterial activity
benzo[f]chromen-3-imines
heterocyclization
hybrid molecules
Knoevenagel reaction
nitriles
salicylic aldehydes
Abstract
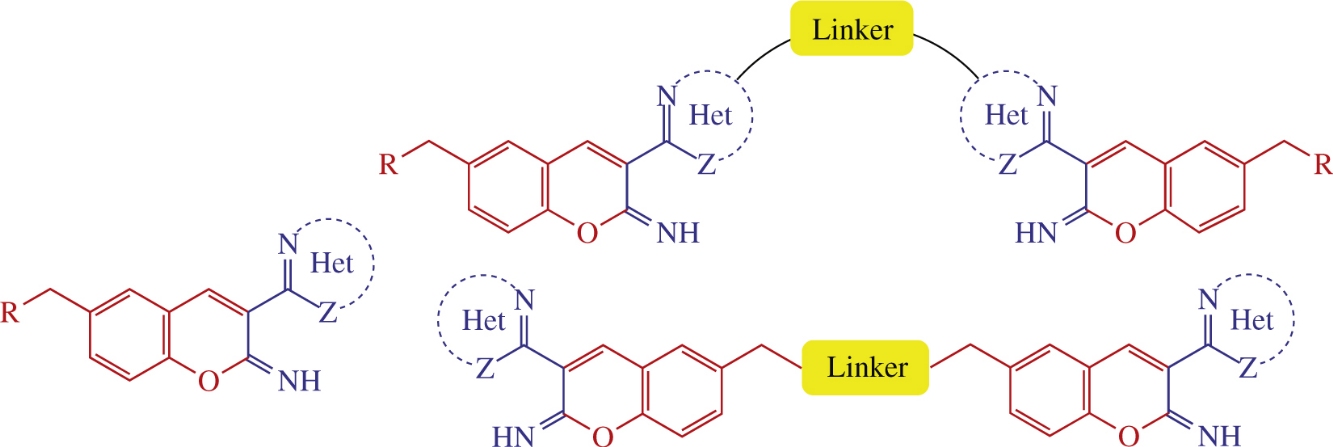
Novel mono- and bis(2H-chromen-2-imine) hybrids linked to different heteroarene units were prepared by cyclocondensation of 2-hydroxybenzaldehydes with 2-heteroarylacetonitriles. Their in vitro antibacterial activities against different strains of Gram-positive and negative bacteria were assessed. Piperazine-linked bis(3-heteroaryl-2Hchromen-2-imines) exhibited the best antibacterial efficacies against E. coli, S. aureus and S. mutans strains with MIC values of 1.4–20.8μm.
References
1.

Kontogiorgis C., Detsi A., Hadjipavlou-Litina D.
Expert Opinion on Therapeutic Patents,
2012
2.

Kostova I., Bhatia S., Grigorov P., Balkansky S., S. Parmar V., K. Prasad A., Saso L.
Current Medicinal Chemistry,
2011
3.

Sanad S.M., Mekky A.E.
Synthetic Communications,
2020
4.

Cho J., Hwang T., Chang T., Lim Y., Sung P., Lee T., Chen J.
Food Chemistry,
2012
5.

Kim N.H., Kim S., Oh J.S., Lee S., Kim Y.K.
Biochemical and Biophysical Research Communications,
2012
6.

Gomez-Outes A., Luisa Suarez-Gea M., Calvo-Rojas G., Lecumberri R., Rocha E., Pozo-Hernandez C., Isabel Terleira-Fernandez A., Vargas-Castrillon E.
Current Drug Discovery Technologies,
2012
7.
10.1016/j.mencom.2020.11.024_bib0035
Sanad
J. Iran. Chem. Soc.,
2020
8.

Anand P., Singh B., Singh N.
Bioorganic and Medicinal Chemistry,
2012
9.

Matos M.J., Viña D., Janeiro P., Borges F., Santana L., Uriarte E.
Bioorganic and Medicinal Chemistry Letters,
2010
10.

Elnaggar D.H., Abdel Hafez N.A., Rashdan H.R., Abdelwahed N.A., Awad H.M., Ali K.A.
Mini-Reviews in Medicinal Chemistry,
2019
11.

Prashanth T., Avin B.R., Thirusangu P., Ranganatha V.L., Prabhakar B.T., Sharath Chandra J.N., Khanum S.A.
Biomedicine and Pharmacotherapy,
2019
12.

Cagide F., Borges F., Gomes L.R., Low J.N.
Journal of Molecular Structure,
2015
13.

Ibrar A., Tehseen Y., Khan I., Hameed A., Saeed A., Furtmann N., Bajorath J., Iqbal J.
Bioorganic Chemistry,
2016
14.
10.1016/j.mencom.2020.11.024_bib0070
Yaghmaei
Chem.,
2017
15.

Neyts J., Clercq E.D., Singha R., Chang Y.H., Das A.R., Chakraborty S.K., Hong S.C., Tsay S., Hsu M., Hwu J.R.
Journal of Medicinal Chemistry,
2009
16.

Singh L.R., Avula S.R., Raj S., Srivastava A., Palnati G.R., Tripathi C.K., Pasupuleti M., Sashidhara K.V.
Journal of Antibiotics,
2017
17.

Sethi P., Bansal Y., Bansal G.
Medicinal Chemistry Research,
2017
18.

Popova S.A., Shevchenko O.G., Chukicheva I.Y., Kutchin A.V.
Chemistry and Biodiversity,
2019
19.

S. Zambare A., A. Kalam Khan F., P. Zambare S., D. Shinde S., N. Sangshetti J.
Current Organic Chemistry,
2015
20.
![Synthesis of novel bis(chromenes) and bis(chromeno[3,4-C]pyridine) incorporating piperazine moiety](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Mekky A.E., Sanad S.M.
Synthetic Communications,
2019
21.

Shakil M.R., Meguerdichian A.G., Tasnim H., Shirazi-Amin A., Seraji M.S., Suib S.L.
Inorganic Chemistry,
2019
22.

Hashem H.E., Amr A.E., Nossier E.S., Elsayed E.A., Azmy E.M.
Molecules,
2020
23.

Hassan A.Y., Sarg M.T., Hussein E.M.
Journal of Heterocyclic Chemistry,
2019
24.
10.1016/j.mencom.2020.11.024_bib0120
Makowska
Pharmazie,
2019
25.
![Convenient synthesis of linear 2H,6H-pyrano[3,2-g] chromenes from natural occurring compound; visnagin](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Abdel-Aziem A., El-Sawy E.R., Kirsch G.
Synthetic Communications,
2019
26.

Radwan A.S., Khalid M.A.
Journal of Heterocyclic Chemistry,
2019
27.

Khidre R.E., Radini I.A.
Journal of Heterocyclic Chemistry,
2019
28.

Makowska A., Sączewski F., Bednarski P., Sączewski J., Balewski Ł.
Molecules,
2018
29.

Meng T., Qin Q., Wang Z., Peng L., Zou H., Gan Z., Tan M., Wang K., Liang F.
Journal of Inorganic Biochemistry,
2018
30.

Sanad S.M., Ahmed A.A., Mekky A.E.
Archiv der Pharmazie,
2020
31.
10.1016/j.mencom.2020.11.024_bib0155
Sanad
J. Heterocycl. Chem.,
2020
32.

North M., Young C.
ChemSusChem,
2011
33.

Ahmed A.A., Mekky A.E., Elwahy A.H., Sanad S.M.
Synthetic Communications,
2020
34.
![Microwave Assisted Three Component One‐pot Synthesis of Bis(aminoazolo[1,5‐ a ]pyrimidines) and Bis(aminoazino[1,2‐ a ]benzimidazoles) Bearing Thiazole Moiety](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Mekky A.E., Sanad S.M., Ahmed A.A.
ChemistrySelect,
2019
35.

Mekky A.E., Sanad S.M.
Bioorganic Chemistry,
2020
36.

Mekky A.E., Sanad S.M.
Journal of Heterocyclic Chemistry,
2019
37.

Mohammad H., Reddy P.V., Monteleone D., Mayhoub A.S., Cushman M., Seleem M.N.
European Journal of Medicinal Chemistry,
2015