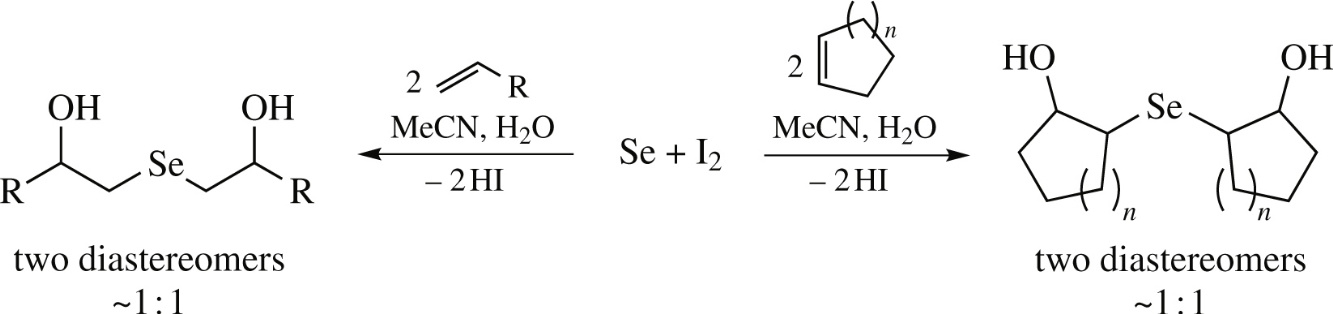Keywords
alkenes
b-hydroxy selenides
iodine-assisted reactions
organoselenium compounds
selenenylation
Abstract
β,β′-Dihydroxy selenides are obtained by the iodine-assisted reaction of elemental selenium with alkenes in aqueous acetonitrile in up to 83% yield and ∼1: 1 diastereomeric ratio. The X-ray analysis of bis(2-hydroxycyclohex-1-yl) selenide revealed it to be the trans,trans-isomer indicating that the reaction occurred stereoselectively as anti-addition.
References
1.

Ścianowski J., Rafiński Z., Wojtczak A., Burczyński K.
Tetrahedron Asymmetry,
2009
2.

Kumakura F., Mishra B., Priyadarsini K.I., Iwaoka M.
European Journal of Organic Chemistry,
2010
3.

Arai K., Kumakura F., Takahira M., Sekiyama N., Kuroda N., Suzuki T., Iwaoka M.
Journal of Organic Chemistry,
2015
4.

Tanini D., Degl'Innocenti A., Capperucci A.
European Journal of Organic Chemistry,
2014
5.

Tanini D., D'Esopo V., Chen D., Barchielli G., Capperucci A.
Phosphorus, Sulfur and Silicon and the Related Elements,
2016
6.

Tanini D., Tiberi C., Gellini C., Salvi P.R., Capperucci A.
Advanced Synthesis and Catalysis,
2018
7.

Leng T., Wu G., Zhou Y., Gao W., Ding J., Huang X., Liu M., Wu H.
Advanced Synthesis and Catalysis,
2018
8.

Wang X., Li H., Yan J.
Chinese Chemical Letters,
2018
9.

EPP O., LADENSTEIN R., WENDEL A.
FEBS Journal,
1983
10.
10.1016/j.mencom.2020.11.023_bib0050
Santi
Organoselenium Compounds in Biology and Medicine: Synthesis, Biological and Therapeutic Treatments,
2017
11.

Kurkutov E.O., Potapov V.A., Amosova S.V.
Russian Journal of Organic Chemistry,
2018
12.

Gopal M., Milne J.
Inorganic Chemistry,
1992
13.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007