Keywords
amides
peptide synthesis
prostate cancer
prostate specific membrane antigen
solid-phase synthesis
target drug delivery
Abstract
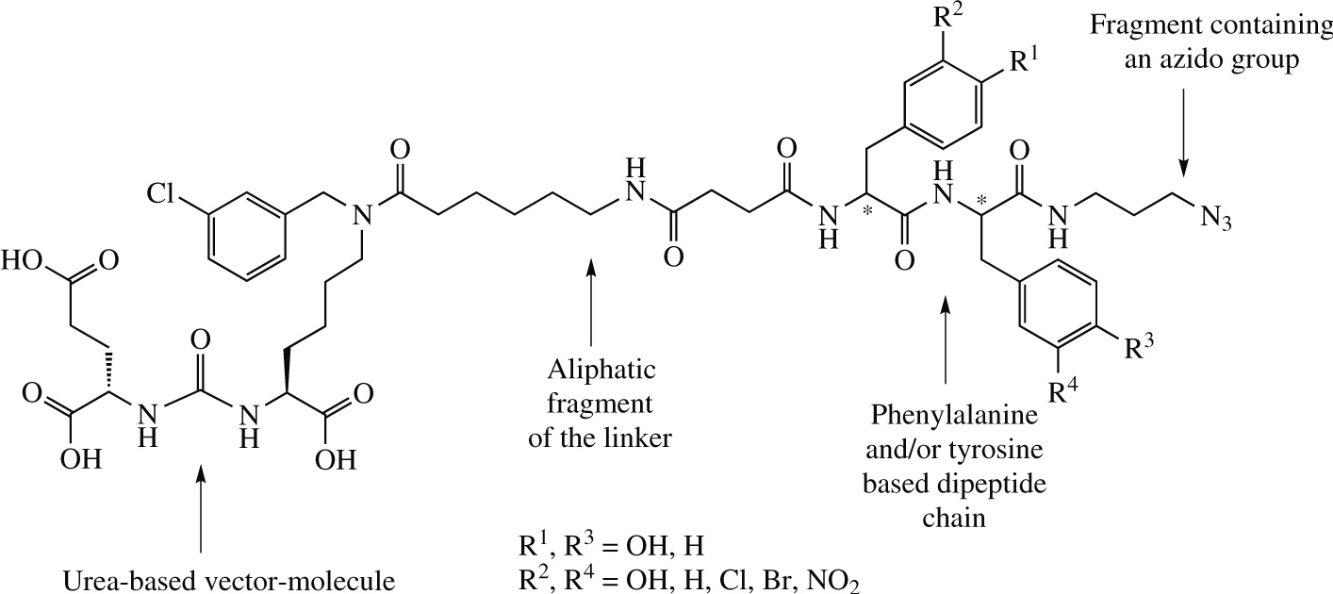
Selective ligands of an urea-based prostate specific membrane antigen with a phenylalanine/tyrosine-based dipeptide linker and with a mingled chiral centers configuration and/or substituted aromatic fragments were prepared in seven steps by liquid- and in six steps by solid-phase synthesis. In vitro test for inhibiting the cleavage of N-acetylaspartylglutamate revealed the optimum linker containing l-phenylalanine in the structure on the N-terminus of a dipeptide chain.
References
1.

Siegel R.L., Miller K.D., Jemal A.
Ca-A Cancer Journal for Clinicians,
2019
2.
10.1016/j.mencom.2020.11.022_bib0010
Chekhonin
Voprosy Med. Khim.,
2002
3.

Uspenskaya A.A., Machulkin A.É., Mazhuga A.G., Beloglazkina E.K.
Pharmaceutical Chemistry Journal,
2019
4.

Glutamate Carboxypeptidase II in Diagnosis and Treatment of Neurologic Disorders and Prostate Cancer
Barinka C., Rojas C., Slusher B., Pomper M.
Current Medicinal Chemistry,
2012
5.

Ivanenkov Y.A., Machulkin A.E., Garanina A.S., Skvortsov D.A., Uspenskaya A.A., Deyneka E.V., Trofimenko A.V., Beloglazkina E.K., Zyk N.V., Koteliansky V.E., Bezrukov D.S., Aladinskaya A.V., Vorobyeva N.S., Puchinina M.M., Riabykh G.K., et. al.
Bioorganic and Medicinal Chemistry Letters,
2019
6.

Machulkin A.E., Ivanenkov Y.A., Aladinskaya A.V., Veselov M.S., Aladinskiy V.A., Beloglazkina E.K., Koteliansky V.E., Shakhbazyan A.G., Sandulenko Y.B., Majouga A.G.
Journal of Drug Targeting,
2016
7.

Jayaprakash S., Wang X., Heston W., Kozikowski A.
ChemMedChem,
2006
8.

Machulkin A.E., Skvortsov D.A., Ivanenkov Y.A., Ber A.P., Kavalchuk M.V., Aladinskaya A.V., Uspenskaya A.A., Shafikov R.R., Plotnikova E.A., Yakubovskaya R.I., Nimenko E.A., Zyk N.U., Beloglazkina E.K., Zyk N.V., Koteliansky V.E., et. al.
Bioorganic and Medicinal Chemistry Letters,
2019
9.

Synthesis and Biological Analysis of Prostate-Specific Membrane Antigen-Targeted Anticancer Prodrugs
Kularatne S.A., Venkatesh C., Santhapuram H.R., Wang K., Vaitilingam B., Henne W.A., Low P.S.
Journal of Medicinal Chemistry,
2010
10.
![Radiosynthesis of a new PSMA targeting ligand ([18F]FPy-DUPA-Pep)](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Malik N., Machulla H., Solbach C., Winter G., Reske S.N., Zlatopolskiy B.
Applied Radiation and Isotopes,
2011
11.

Kularatne S.A., Zhou Z., Yang J., Post C.B., Low P.S.
Molecular Pharmaceutics,
2009
12.
A. Machulkin, A. Uspenskaya, A. Ber, S. Petrov, I. Saltykova, Y. Ivanenkov, D. Skvortsov, A. Erofeev, P. Gorelkin, E. Beloglazkina, E. Belov, E. Khazanova and A. Mazhuga, Patent RU 2697519C1, 2019.
13.

Lundt B.F., Johansen N.L., Vølund A., Markussen J.
International journal of peptide & protein research,
2009
14.

Shendage D.M., Fröhlich R., Haufe G.
Organic Letters,
2004
15.
10.1016/j.mencom.2020.11.022_bib0075
Shin
Synlett,
2009
16.
10.1016/j.mencom.2020.11.022_bib0080
Lygo
1-Hydroxybenzotriazole,
2013
17.

Pearson D.A., Blanchette M., Baker M.L., Guindon C.A.
Tetrahedron Letters,
1989
18.
10.1016/j.mencom.2020.11.022_bib0090
Peptide Synthesis and Applications,
2005
19.
10.1016/j.mencom.2020.11.022_bib0095
Peptide Synthesis Protocols,
1995
20.

Guillier F., Orain D., Bradley M.
Chemical Reviews,
2000
21.

CHATZI K.B., GATOS D., STAVROPOULOS G.
International journal of peptide & protein research,
2009
22.

FUJIWARA Y., AKAJI K., KISO Y.
Chemical and Pharmaceutical Bulletin,
2011
23.
10.1016/j.mencom.2020.11.022_bib0115
Horoszewicz
Cancer Res.,
1983
24.

Ben Jemaa A., Sallami S., Céraline J., Oueslati R.
Cell Biology International,
2013