Keywords
N-oxides
azidopyrimidine N-oxides
CuAAC
pyrimidines
quantum chemical calculations
tautomers
triazoles
Abstract
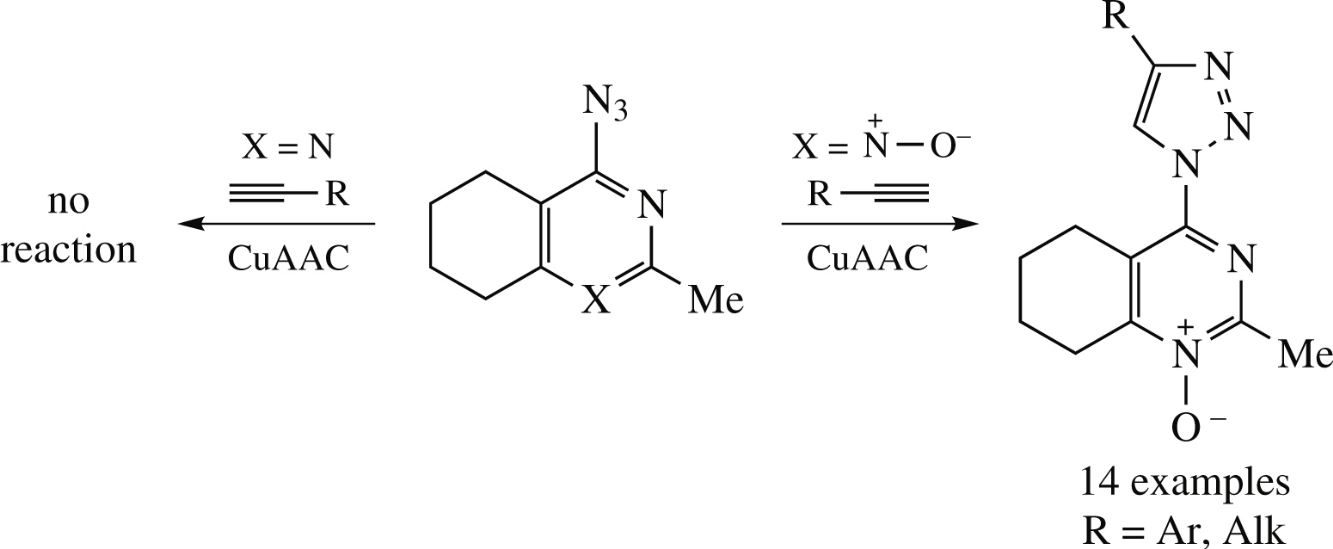
4-Azido-2-methyltetrahydroquinazoline N-oxide cleanly undergoes the copper(i)-catalyzed azide–alkyne cycloaddition (CuAAC) reaction with alkynes to give new conjugates with pyrimidine N-oxide and triazole moieties. Its deoxygenated analogue, 4-azido-2-methyltetrahydroquinazoline, is inert in CuACC process due to the shift of imidoyl azide–tetrazole equilibrium towards the tetrazole tautomer.
References
1.
10.1016/j.mencom.2020.11.008_bib0005
Rewcastle
2008
2.

Baumann M., Baxendale I.R.
Beilstein Journal of Organic Chemistry,
2013
3.

Taylor R.D., MacCoss M., Lawson A.D.
Journal of Medicinal Chemistry,
2014
4.

Agalave S.G., Maujan S.R., Pore V.S.
Chemistry - An Asian Journal,
2011
5.

Dheer D., Singh V., Shankar R.
Bioorganic Chemistry,
2017
6.

Prasher P., Sharma M.
MedChemComm,
2019
7.

Bonandi E., Christodoulou M.S., Fumagalli G., Perdicchia D., Rastelli G., Passarella D.
Drug Discovery Today,
2017
8.

Popova E.A., Trifonov R.E., Ostrovskii V.A.
Russian Chemical Reviews,
2019
9.

Bozorov K., Zhao J., Aisa H.A.
Bioorganic and Medicinal Chemistry,
2019
10.

Rani A., Singh G., Singh A., Maqbool U., Kaur G., Singh J.
RSC Advances,
2020
11.

Ma L., Pang L., Wang B., Zhang M., Hu B., Xue D., Shao K., Zhang B., Liu Y., Zhang E., Liu H.
European Journal of Medicinal Chemistry,
2014
12.

Dofe V.S., Sarkate A.P., Shaikh Z.M., Gill C.H.
Journal of Heterocyclic Chemistry,
2017
13.

Thomann A., Brengel C., Börger C., Kail D., Steinbach A., Empting M., Hartmann R.W.
ChemMedChem,
2016
14.

Meldal M., Tornøe C.W.
Chemical Reviews,
2008
15.

Neumann S., Biewend M., Rana S., Binder W.H.
Macromolecular Rapid Communications,
2019
16.

Minkin V.I., Garnovskii A.D., Elguero J., Katritzky A.R., Denisko O.V.
Advances in Heterocyclic Chemistry,
2000
17.

Gorbunov E.B., Novikova R.K., Plekhanov P.V., Slepukhin P.A., Rusinov G.L., Rusinov V.L., Charushin V.N., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2013
18.

Azide−Tetrazole Equilibrium of C-6 Azidopurine Nucleosides and Their Ligation Reactions with Alkynes
Lakshman M.K., Singh M.K., Parrish D., Balachandran R., Day B.W.
Journal of Organic Chemistry,
2010
19.
![Selective 15N-Labeling and Analysis of 13C−15N J Couplings as an Effective Tool for Studying the Structure and Azide−Tetrazole Equilibrium in a Series of Tetrazolo[1,5-b][1,2,4]triazines and Tetrazolo[1,5-a]pyrimidines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Deev S.L., Shenkarev Z.O., Shestakova T.S., Chupakhin O.N., Rusinov V.L., Arseniev A.S.
Journal of Organic Chemistry,
2010
20.

Khalymbadzha I.A., Shestakova T.S., Deev S.L., Rusinov V.L., Chupakhin O.N., Shenkarev Z.O., Arseniev A.S.
Russian Chemical Bulletin,
2013
21.

Shestakova T.S., Shenkarev Z.O., Deev S.L., Chupakhin O.N., Khalymbadzha I.A., Rusinov V.L., Arseniev A.S.
Journal of Organic Chemistry,
2013
22.

Pleshkova N.V., Nikolaenkova E.B., Krivopalov V.P., Mamatyuk V.I.
Russian Chemical Bulletin,
2017
23.

Thomann A., Zapp J., Hutter M., Empting M., Hartmann R.W.
Organic and Biomolecular Chemistry,
2015
24.

Synthesis and fluorescent properties of N(9)-alkylated 2-amino-6-triazolylpurines and 7-deazapurines
Šišuļins A., Bucevičius J., Tseng Y., Novosjolova I., Traskovskis K., Bizdēna Ē., Chang H., Tumkevičius S., Turks M.
Beilstein Journal of Organic Chemistry,
2019
25.

Zaķis J.M., Ozols K., Novosjolova I., Vilšķe̅rsts R., Mishnev A., Turks M.
Journal of Organic Chemistry,
2020
26.
Astakhov A.V., Suponitsky K.Y., Chernyshev V.M.
Mendeleev Communications,
2018
27.

Nikolaenkova Е.B., Aleksandrova N.V., Mamatyuk V.I., Krivopalov V.P.
Russian Chemical Bulletin,
2018
28.

Sedenkova K.N., Averina E.B., Grishin Y.K., Kutateladze A.G., Rybakov V.B., Kuznetsova T.S., Zefirov N.S.
Journal of Organic Chemistry,
2012
29.

Sedenkova K.N., Averina E.B., Grishin Y.K., Bacunov A.B., Troyanov S.I., Morozov I.V., Deeva E.B., Merkulova A.V., Kuznetsova T.S., Zefirov N.S.
Tetrahedron Letters,
2015
30.

Sedenkova K.N., Dueva E.V., Averina E.B., Grishin Y.K., Osolodkin D.I., Kozlovskaya L.I., Palyulin V.A., Savelyev E.N., Orlinson B.S., Novakov I.A., Butov G.M., Kuznetsova T.S., Karganova G.G., Zefirov N.S.
Organic and Biomolecular Chemistry,
2015
31.

Bis(gem-dihalocyclopropanes): synthesis and heterocyclization upon treatment with nitronium triflate
Sedenkov K.N., Averin E.B., Grishin Y.K., Kuznetsova T.S., Zefirov N.S.
Russian Chemical Bulletin,
2016
32.

Sedenkova K.N., Averina E.B., Grishin Y.K., Kolodyazhnaya J.V., Rybakov V.B., Vasilenko D.A., Steglenko D.V., Minkin V.I., Kuznetsova T.S., Zefirov N.S.
Tetrahedron Letters,
2017
33.

Sedenkova K.N., Averina E.B., Grishin Y.K., Kolodyazhnaya J.V., Rybakov V.B., Kuznetsova T.S., Hughes A., Gomes G.D., Alabugin I.V., Zefirov N.S.
Organic and Biomolecular Chemistry,
2017
34.
Sedenkova K.N., Averina E.B., Nazarova A.A., Grishin Y.K., Karlov D.S., Zamoyski V.L., Grigoriev V.V., Kuznetsova T.S., Palyulin V.A.
Mendeleev Communications,
2018
35.
Sedenkova K.N., Nazarova A.A., Khvatov E.V., Dueva E.V., Orlov A.A., Osolodkin D.I., Grishin Y.K., Kuznetsova T.S., Palyulin V.A., Averina E.B.
Mendeleev Communications,
2018
36.

Sedenkova K.N., Kolodyazhnaya J.V., Vasilenko D.A., Gracheva Y.A., Kharitonoshvili E.V., Grishin Y.K., Chistov A.A., Rybakov V.B., Holt T., Kutateladze A.G., Kuznetsova T.S., Milaeva E.R., Averina E.B.
Dyes and Pigments,
2019
37.

Sedenkova K.N., Terekhin A.V., Abdrashitova I.V., Vasilenko D.A., Sadovnikov K.S., Gracheva Y.A., Grishin Y.K., Holt T., Kutateladze A.G., Kuznetsova T.S., Milaeva E.R., Averina E.B.
Tetrahedron Letters,
2020
38.

Nazarova A.A., Sedenkova K.N., Karlov D.S., Lavrov M.I., Grishin Y.K., Kuznetsova T.S., Zamoyski V.L., Grigoriev V.V., Averina E.B., Palyulin V.A.
MedChemComm,
2019
39.

Sedenkova K.N., Averina E.B., Grishin Y.K., Kuznetsova T.S., Zefirov N.S.
Tetrahedron Letters,
2014