Keywords
adamantane
chlorambucil
colchicine
dual target profile
heterobivalent compounds
lung carcinoma A549
tubulin
twin drugs
Abstract
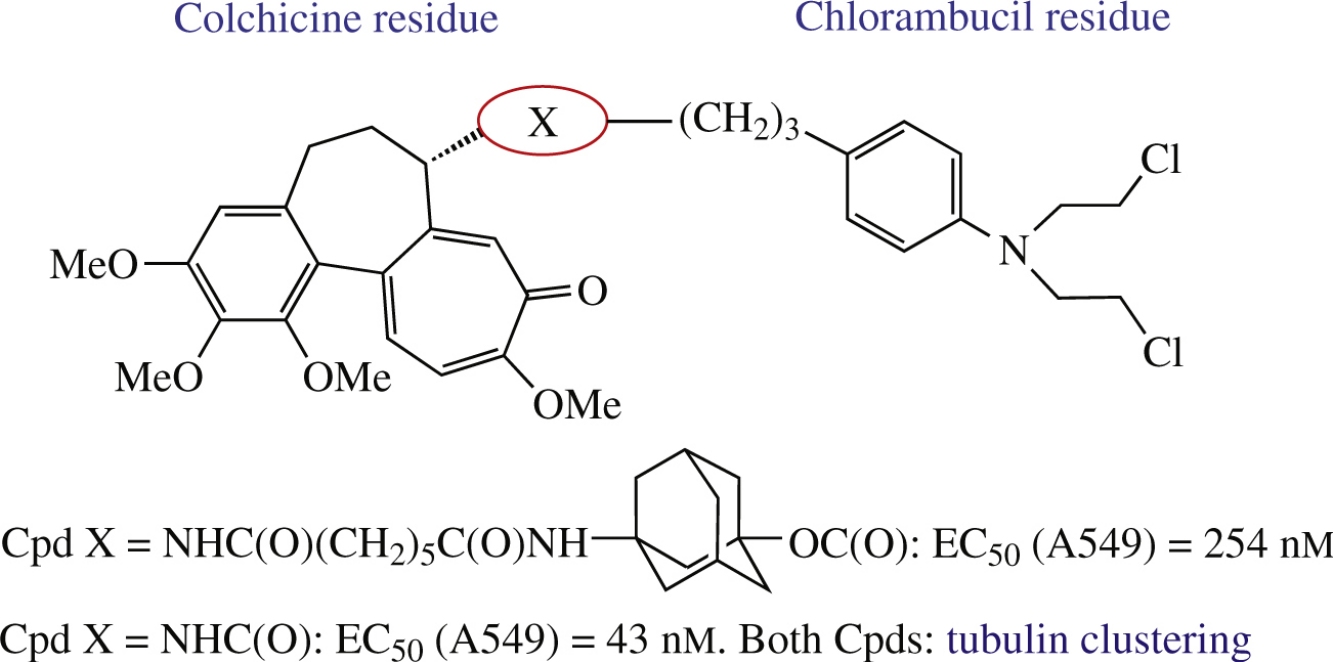
Two new heterobivalent molecules were designed by linking a DNA-alkylating agent chlorambucil with tubulin-targeting compound colchicine or its adamantyl containing C(7)-derivative. Target compounds were synthesized via acid coupling with N-deacetylcolchicine. According to preliminary biotests, both conjugates caused microtubule depolymerization and strong tubulin clustering, and colchicine–chlorambucil conjugate was found to be one order of magnitude more potent and to exhibit some selectivity to A549 carcinoma cells.
References
1.
10.1016/j.mencom.2020.11.005_bib0005
The Practice of Medicinal Chemistry,
2003
2.

Schmidt A., Bölck B., Jedig M., Steinritz D., Balszuweit F., Kehe K., Bloch W.
Toxicology,
2009
3.

La Regina G., Coluccia A., Naccarato V., Silvestri R.
European Journal of Pharmaceutical Sciences,
2019
4.

Haider K., Rahaman S., Yar M.S., Kamal A.
Expert Opinion on Therapeutic Patents,
2019
5.

Chen Y., Jia Y., Song W., Zhang L.
Frontiers in Pharmacology,
2018
6.

Pathak R.K., Wen R., Kolishetti N., Dhar S.
Molecular Cancer Therapeutics,
2017
7.
Zefirov N.A., Gadert L., Fatkulin A.R., Shibilev V.M., Butov G.M., Mokhov V.M., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2020
8.
E. V. Nurieva, N.A. Zefirov, N.S. Temnyakova, S.A. Kuznetsov and O. N. Zefirova, Izv. Akad. Nauk, Ser. Khim., 2020, in press.
9.

Thomopoulou P., Sachs J., Teusch N., Mariappan A., Gopalakrishnan J., Schmalz H.
ACS Medicinal Chemistry Letters,
2015
10.

Kim S., Cho S., Kim H., Seok H., Kim S., Kyu Kwon T., Chang J.
Experimental and Molecular Medicine,
2013
11.

Prota A.E., Danel F., Bachmann F., Bargsten K., Buey R.M., Pohlmann J., Reinelt S., Lane H., Steinmetz M.O.
Journal of Molecular Biology,
2014
12.

Trott O., Olson A.J.
Journal of Computational Chemistry,
2009
13.

Pettersen E.F., Goddard T.D., Huang C.C., Couch G.S., Greenblatt D.M., Meng E.C., Ferrin T.E.
Journal of Computational Chemistry,
2004
14.

Zefirova O.N., Nurieva E.V., Wobith B., Schulz S., Zefirov N.A., Kuznetsov S.A.
Pure and Applied Chemistry,
2020
15.

Bagnato J.D., Eilers A.L., Horton R.A., Grissom C.B.
Journal of Organic Chemistry,
2004
16.

Gerlier D., Thomasset N.
Journal of Immunological Methods,
1986
17.
Zefirov N.A., Evteeva Y.A., Krasnoperova A.I., Mamaeva A.V., Milaeva E.R., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2020
18.

Lavrushkina E.A., Shibilev V.M., Zefirov N.A., Shevtsova E.F., Shevtsov P.N., Kuznetsov S.A., Zefirova O.N.
Russian Chemical Bulletin,
2020
19.
Zefirov N.A., Hoppe M., Kuznetsova I.V., Chernyshov N.A., Grishin Y.K., Maloshitskaya O.A., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2018
20.

Kalinina M.A., Skvortsov D.A., Rubtsova M.P., Komarova E.S., Dontsova O.A.
Molecular Imaging and Biology,
2017
21.

Proshin A.N., Trofimova T.P., Zefirova O.N., Petrova L.N., Skvortsov D.A., Bachurin S.O.
Russian Chemical Bulletin,
2019