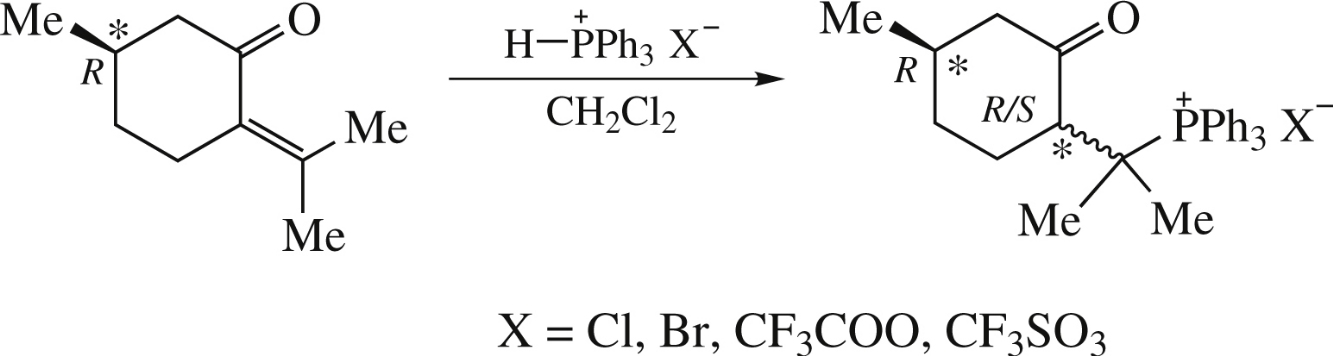Keywords
p-menthane
R-pulegone
addition reaction
phosphonium salt
terpenes
terpenoids
triphenylphosphine
α,β-enones
Abstract
The reaction of the R-pulegone with P–H-phosphonium salts gives the corresponding 8-phosphonio-p-menthan-3-one salts with high regio- and stereoselectivity. The structure of salts was determined by NMR and IR spectroscopy, mass spectrometry and X-ray diffraction analysis.
References
1.

Hobuß D., Hasenjäger J., Driessen-Hölscher B., Baro A., Axenov K.V., Laschat S., Frey W.
Inorganica Chimica Acta,
2011
2.

Wiese B., Knühl G., Flubacher D., Prieß J.W., Ulriksen (nee Laursen) B., Brödner K., Helmchen G.
European Journal of Organic Chemistry,
2005
3.

Malik K.M., Newman P.D.
Dalton Transactions,
2003
4.

Fernández-Pérez H., Etayo P., Panossian A., Vidal-Ferran A.
Chemical Reviews,
2011
5.

Harwood L.M., Julia M.
Synthesis,
1980
6.

Araki S., Hatano M., Butsugan Y.
Journal of Organic Chemistry,
1986
7.

Yang P., Yao M., Li J., Li Y., Li A.
Angewandte Chemie - International Edition,
2016
8.

Brocksom T.J., Coelho F., Deprés J., Greene A.E., Freire de Lima M.E., Hamelin O., Hartmann B., Kanazawa A.M., Wang Y.
Journal of the American Chemical Society,
2002
9.
10.1016/j.mencom.2020.11.003_sbref0020a
Dhanya
Anti-Cancer Agents Med Chem.,
2017
10.

Cho D.Y., Cho H., Kwon K., Yu M., Lee E., Huh K.M., Lee D.H., Kang H.C.
Advanced Functional Materials,
2015
11.

Bachowska B., Kazmierczak-Baranska J., Cieslak M., Nawrot B., Szczęsna D., Skalik J., Bałczewski P.
ChemistryOpen,
2012
12.

Kanazawa A., Ikeda T., Endo T.
Antimicrobial Agents and Chemotherapy,
1994
13.

Wu T., Xie A., Tan S., Cai X.
Colloids and Surfaces B: Biointerfaces,
2011
14.

Pinto T.C., Banerjee A., Nazarov P.A.
Bulletin of Russian State Medical University,
2018
15.

Heller A., Brockhoff G., Goepferich A.
European Journal of Pharmaceutics and Biopharmaceutics,
2012
16.

Lu P., Bruno B.J., Rabenau M., Lim C.S.
Journal of Controlled Release,
2016
17.
10.1016/j.mencom.2020.11.003_sbref0030c
Smith
Discov. Med.,
2011
18.

Ross M.F., Kelso G.F., Blaikie F.H., James A.M., Cochemé H.M., Filipovska A., Da Ros T., Hurd T.R., Smith R.A., Murphy M.P.
Biochemistry (Moscow),
2005
19.

Bittner Fialová S., Kello M., Čoma M., Slobodníková L., Drobná E., Holková I., Garajová M., Mrva M., Zachar V., Lukáč M.
Molecules,
2019
20.
Khasiyatullina N.R., Mironov V.F., Gumerova S.K., Voloshina A.D., Sapunova A.S.
Mendeleev Communications,
2019
21.

Spivak A.Y., Nedopekina D.A., Shakurova E.R., Khalitova R.R., Gubaidullin R.R., Odinokov V.N., Dzhemilev U.M., Bel’skii Y.P., Bel’skaya N.V., Stankevich S.A., Korotkaya E.V., Khazanov V.A.
Russian Chemical Bulletin,
2013
22.

Tsepaeva O.V., Nemtarev A.V., Abdullin T.I., Grigor’eva L.R., Kuznetsova E.V., Akhmadishina R.A., Ziganshina L.E., Cong H.H., Mironov V.F.
Journal of Natural Products,
2017
23.

Tsepaeva O.V., Nemtarev A.V., Mironov V.F.
Russian Journal of Organic Chemistry,
2017
24.

Reaction of Methyl (2-Methylidene)-3-oxolup-20(29)-en-28-oate with Dimethyl Trimethylsilyl Phosphite
Tsepaeva O.V., Nemtarev A.V., Abdullin T.I., Kuznetsova E.V., Mironov V.F.
Russian Journal of General Chemistry,
2018
25.
10.1016/j.mencom.2020.11.003_sbref0045e
Antipin
Eur. J. Clin. Invest.,
2018
26.
10.1016/j.mencom.2020.11.003_sbref0045f
Antipin
Eur. J. Clin. Invest.,
2019
27.
10.1016/j.mencom.2020.11.003_sbref0045g
Tsepaeva
Anti-Cancer Agents Med Chem.,
2019
28.

Testai L., Strobykina I., Semenov V.V., Semenova M., Pozzo E.D., Martelli A., Citi V., Martini C., Breschi M.C., Kataev V.E., Calderone V.
International Journal of Molecular Sciences,
2017
29.

Beaumont A.J., Kiely C., Denise Rooney A.
Journal of Fluorine Chemistry,
2001
30.

Oumzil H., Ghoulami S., Rhajaoui M., Ilidrissi A., Fkih-Tetouani S., Faid M., Benjouad A.
Phytotherapy Research,
2002
31.

Flamini G., Cioni P.L., Puleio R., Morelli I., Panizzi L.
Phytotherapy Research,
1999
32.

González-Chávez M.M., Cárdenas-Ortega N.C., Méndez-Ramos C.A., Pérez-Gutiérrez S.
Molecules,
2011
33.
10.1016/j.mencom.2020.11.003_bib0070
Kim
J. Appl. Entomol.,
2006
34.

Krause L., Herbst-Irmer R., Sheldrick G.M., Stalke D.
Journal of Applied Crystallography,
2015
35.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
36.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
37.

Farrugia L.J.
Journal of Applied Crystallography,
2012
38.

Parsons S., Flack H.D., Wagner T.
Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials,
2013