Keywords
Abstract
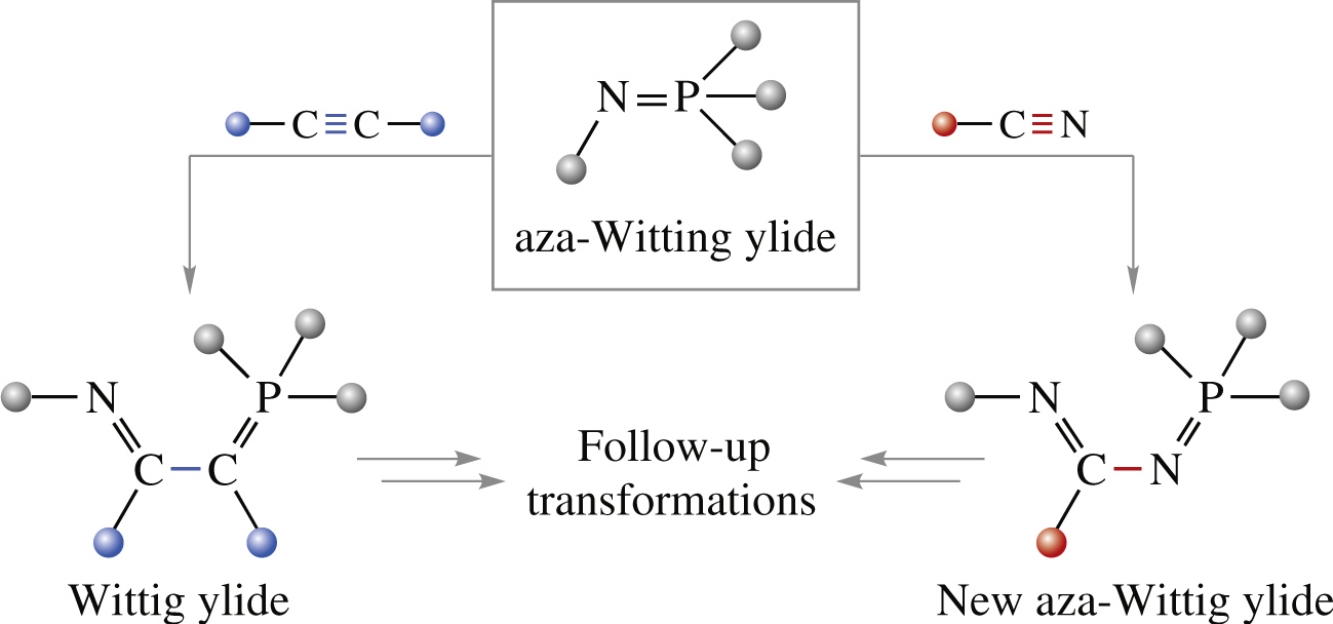
The classic version of aza-Wittig reactions, namely, reactions between phosphazenes and compounds with polar double bonds, is widely employed in organic synthesis to produce C=N bonds. However, only a limited number of aza-Wittig reactions between phosphazenes and compounds with triple bonds is known, which has a lot to do with certain structural features of the reactants, wherein additional activation is required. This review provides a guide to those rare examples of aza-Wittig reactions with acetylenes and nitriles. A unique trait of these atom-economic processes is that they afford Wittig or new aza-Wittig reagents, respectively, which offers new opportunities for employing these reactions in organic syntheses, especially those of N-heterocyclic compounds.
References


























![Ethyl 2-methoxy-6-[(triphenylphosphoranylidene)amino]nicotinate and ethyl 2-methylsulfanyl-6-[(triphenylphosphoranylidene)amino]nicotinate](/storage/images/resized/xG3oC19920mmFVqkoVEGKGaMCeoalzyvWxPkVox5_small_thumb.webp)
![Dimethyl 6-[1,2-bis(methoxycarbonyl)-2-(triphenyl-λ5-phosphanylidene)ethylideneamino]-2-methylsulfanyl-3,4-pyridinedicarboxylate forms a chain of hydrogen-bonded rings](/storage/images/resized/xG3oC19920mmFVqkoVEGKGaMCeoalzyvWxPkVox5_small_thumb.webp)







![[2+2] Cycloaddition and ring expansion reactions of cyclic phosphonium and aminophosphonium salts: synthesis and structure of the first eight-membered ylide-type heterocycles](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)










![Reactions of bis(iminophosphoranes) with palladium(II) dichloride: Metal-induced tautomerization orthopalladation and unexpected platinum-assisted [2 + 2] cycloaddition of an aryl-nitrile with a phosphinimine moiety](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)