Keywords
naphthalene
organic solar cells
rhodanine
Stille cross-coupling
thiophene
Abstract
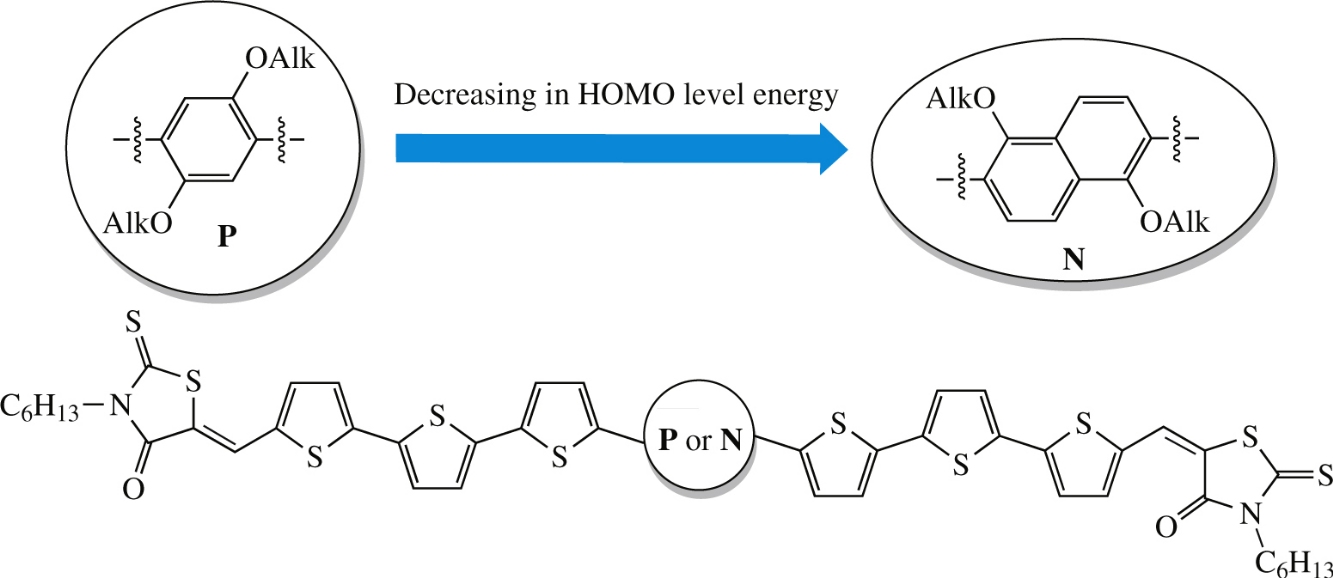
New small molecules bearing either phenylene or naphthalene core in combination with thiophene and rhodanine periphery moieties were synthesized by the Stille cross-coupling reaction. Their evaluation for prototypes of organic solar cells showed that replacing phenylene moiety by the naphthalene one caused decrease in the HOMO energy, while the open-circuit voltage and fill factors of solar cells based on these materials were significantly improved to manifest maximum power conversion efficiencies to 5.3%.
References
1.

Liu Q., Jiang Y., Jin K., Qin J., Xu J., Li W., Xiong J., Liu J., Xiao Z., Sun K., Yang S., Zhang X., Ding L.
Science Bulletin,
2020
2.

Zhou R., Jiang Z., Yang C., Yu J., Feng J., Adil M.A., Deng D., Zou W., Zhang J., Lu K., Ma W., Gao F., Wei Z.
Nature Communications,
2019
3.

Sun X., Zhou Y., Wu W., Liu Y., Tian W., Yu G., Qiu W., Chen S., Zhu D.
Journal of Physical Chemistry B,
2006
4.

Lin Y., Zhan X.
Accounts of Chemical Research,
2015
5.

Collins S.D., Ran N.A., Heiber M.C., Nguyen T.
Advanced Energy Materials,
2017
6.

Roncali J., Leriche P., Blanchard P.
Advanced Materials,
2014
7.

Kostyuchenko A.S., Ulyankin E.B., Zheleznova T.Y., Chernenko S.A., Shatsauskas A.L., Abaidulina D.R., Bystrushkin M.O., Samsonenko A.L., Fisyuk A.S.
Chemistry of Heterocyclic Compounds,
2019
8.
Solodukhin A.N., Luponosov Y.N., Buzin M.I., Peregudova S.M., Svidchenko E.A., Ponomarenko S.A.
Mendeleev Communications,
2018
9.
Lukovskaya E.V., Dyachenko N.V., Khoroshutin A.V., Bobyleva A.A., Anisimov A.V., Karnoukhova V.A., Jonusauskas G., Fedorov Y.V., Fedorova O.A.
Mendeleev Communications,
2018
10.
Malov V.V., Ghosh T., Nair V.C., Maslov M.M., Katin K.P., Unni K.N., Tameev A.R.
Mendeleev Communications,
2019
11.
![Synthesis and photovoltaic properties of new thieno[3,2-b]indole-based dyes](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Steparuk A.S., Irgashev R.A., Rusinov G.L., Krivogina E.V., Lazarenko P.I., Kozyukhin S.A.
Russian Chemical Bulletin,
2019
12.

13.

Li Y., Guo Q., Li Z., Pei J., Tian W.
Energy and Environmental Science,
2010
14.

Li H., Lu K., Wei Z.
Advanced Energy Materials,
2017
15.

Je H., Hong J., Kwon H., Kim N.Y., Park C.E., Kwon S., An T.K., Kim Y.
Dyes and Pigments,
2018
16.

Li Z., He G., Wan X., Liu Y., Zhou J., Long G., Zuo Y., Zhang M., Chen Y.
Advanced Energy Materials,
2011
17.

Roquet S., Cravino A., Leriche P., Alévêque O., Frère P., Roncali J.
Journal of the American Chemical Society,
2006
18.

Suman S., Singh S.P.
Journal of Materials Chemistry A,
2019
19.

Wang Z., Xu X., Li Z., Feng K., Li K., Li Y., Peng Q.
Advanced Electronic Materials,
2016
20.

A. V., Liu S., Wong K.
Materials Science and Engineering: R: Reports,
2018
21.

Kim I., Jo J., Yun J.
Nanomaterials,
2016
22.

Yao H., Ye L., Zhang H., Li S., Zhang S., Hou J.
Chemical Reviews,
2016
23.
![Benzo[1,2-b:4,5-b′]dithiophene (BDT)-based small molecules for solution processed organic solar cells](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Li M., Ni W., Wan X., Zhang Q., Kan B., Chen Y.
Journal of Materials Chemistry A,
2015
24.

Zhou J., Zuo Y., Wan X., Long G., Zhang Q., Ni W., Liu Y., Li Z., He G., Li C., Kan B., Li M., Chen Y.
Journal of the American Chemical Society,
2013
25.

Zhao J., Xia B., Lu K., Deng D., Yuan L., Zhang J., Zhu L., Zhu X., Li H., Wei Z.
RSC Advances,
2016
26.

Baek M., Park H., Dutta P., Lee W., Kang I., Lee S.
Journal of Polymer Science, Part A: Polymer Chemistry,
2013
27.
![A potential naphtho[2,1-b:3,4-b′]dithiophene-based polymer with large open circuit voltage for efficient use in organic solar cells](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Kim Y.J., Cheon Y.R., Jang J., Kim Y., Park C.E.
Journal of Materials Chemistry C,
2015
28.

Qin H., Cai D., Wang M., Ma Y., Yin Z., Tang C., Chen S., Zheng Q.
Polymer,
2015
29.

Chochos C.L., Katsouras A., Drakopoulou S., Miskaki C., Krassas M., Tzourmpakis P., Kakavelakis G., Sprau C., Colsmann A., Squeo B.M., Gregoriou V.G., Kymakis E., Avgeropoulos A.
Journal of Polymer Science, Part A: Polymer Chemistry,
2017
30.

Sun X., Zhang Z., Hou R., Huang M., Zhao B., Tan S.
Dyes and Pigments,
2017