Keywords
2-mercaptobenzimidazole
benz[4,5]imidazo[2,1-b][1,3]thiazines
benz[4,5]imidazo[2,1-b]thiazoles
X-ray crystallography
α-chloro ketones
α-chloro oxiranes
Abstract
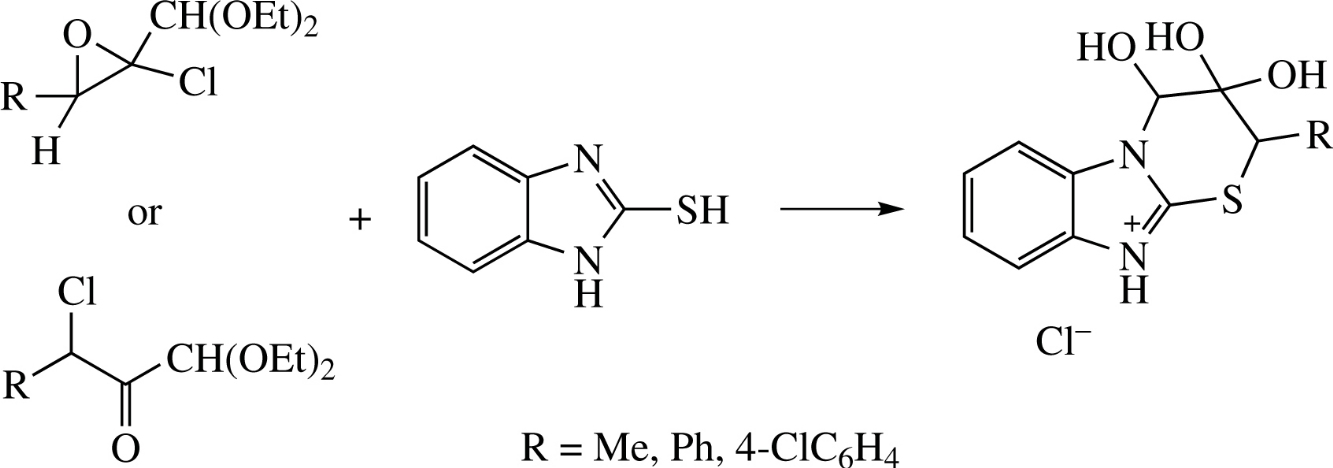
3-Diethoxymethyl-3-hydroxy-3,9-dihydro-2H-benz[4,5]imidazo[2,1-b]thiazol-4-ium salts were synthesized by reaction of chloro oxiranes or isomeric α-chloro ketones with 2-mercaptobenzimidazole. The acetal group of the aforementioned salts undergoes hydrolysis in acidic medium to liberate aldehyde which would react with the NH-moiety of mercaptobenzimidazole, thus leading to cycle expansion with the formation of 3,3,4-trihydroxy-3,4-dihydro-2H-benz[4,5]imidazo[2,1-b][1,3]thiazinium salts. In DMSO solution, the 3,3,4-trihydroxythiazine moiety of the latter compounds is oxidized into 3,4-dioxothiazine one existing in enol form.
References
1.
![CuI nanoparticle-catalyzed synthesis of tetracyclic benzo[e]benzo[4,5]imidazo[1,2-c][1,3]thiazin-6-imine heterocycles by SNAr-type C–S, C–N bond formation from isothiocyanatobenzenes and benzimidazoles](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
2.
![5H-Benzo[d]Benzo[4,5]Imidazo[2,1-b][1,3]Thiazine as a Novel Electron-Acceptor Cored High Triplet Energy Bipolar Host Material for Efficient Solution-Processable Thermally Activated Delayed Fluorescence Organic Light-Emitting Diodes](/storage/images/resized/4QWA67eqfcfyOiA8Wk7YnqroHFqQbTsmDJUYTCTg_small_thumb.webp)
Godumala M., Yoon J., Park S.Y., Lee C., Kim Y., Jeong J., Park S., Woo H.Y., Cho M.J., Choi D.H.
Frontiers in Chemistry,
2020
3.

Matysiak J., Karpińska M.M., Skrzypek A., Wietrzyk J., Kłopotowska D., Niewiadomy A., Paw B., Juszczak M., Rzeski W.
Arabian Journal of Chemistry,
2019
4.

Nikolova I., Slavchev I., Ravutsov M., Dangalov M., Nikolova Y., Zagranyarska I., Stoyanova A., Nikolova N., Mukova L., Grozdanov P., Nikolova R., Shivachev B., Kuz'min V.E., Ognichenko L.N., Galabov A.S., et. al.
Bioorganic Chemistry,
2019
5.

Hamama W.S., Waly M.A., El-Hawary I.I., Zoorob H.H.
Journal of Heterocyclic Chemistry,
2016
6.
![Synthesis, spectral characterization, and antituberculosis activity of thiazino[3,2-A]benzimidazole derivatives](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Gong J., He Y., Cui Z., Guo Y.
Phosphorus, Sulfur and Silicon and the Related Elements,
2016
7.
![Phase-transfer catalytic synthesis and hypocholesterolemic activity of thiazino[3,2-a]benzimidazole and its silicon analog](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Abele E., Abele R., Arsenyan P., Belyakov S., Veveris M., Lukevics E.
Chemistry of Heterocyclic Compounds,
2007
8.
![Synthesis, crystal structure, antioxidant activity and dft study of 2-aryl-2,3-dihydro-4H-[1,3]thiazino[3,2-a]benzimidazol-4-One](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ramos Rodríguez O.A., Magaña Vergara N.E., Mojica Sánchez J.P., Sumaya Martínez M.T., Gómez Sandoval Z., Cruz A., Ramos Organillo Á.
Journal of Molecular Structure,
2020
9.
10.1016/j.mencom.2020.09.041_bib0045
Guseinov
Russ. J. Org. Chem.,
1995
10.

Guseinov F.I., Yudina N.A.
Chemistry of Heterocyclic Compounds,
1998
11.
![Synthesis and molecular structure of 3,7-dimethyl-2-[N-(4-methylpyridyl-2)-4-hydroxy-3-methyl-5-oxopyrrolen-3-yl-2]imidazo[1,2-a]pyridine](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Guseinov F.N., Burangulova R.N., Mukhamedzyanova E.F., Strunin B.P., Sinyashin O.G., Litvinov I.A., Gubaidullin A.T.
Chemistry of Heterocyclic Compounds,
2006
12.

Pistsov M.F., Lavrova O.M., Saifutdinov A.M., Burangulova R.N., Kustov L.M., Guseinov F.I., Musin R.Z.
Russian Journal of General Chemistry,
2017
13.

Guseinov F., Pistsov M., Movsumzade E., Kustov L., Tafeenko V., Chernyshev V., Gurbanov A., Mahmudov K., Pombeiro A.
Crystals,
2017
14.
10.1016/j.mencom.2020.09.041_bib0070
Guseinov
Zh. Org. Khim.,
1994
15.
G. M. Sheldrick, SADABS, Bruker AXS, Madison, WI, 1997.
16.

Altomare A., Cascarano G., Giacovazzo C., Viterbo D.
Acta Crystallographica Section A Foundations of Crystallography,
1991
17.
G. M. Sheldrick, SHELX-97, Programs for Crystal Structure Analysis (Release 97-2), Version 1.2, University of Göttingen, 1997.
18.

Farrugia L.J.
Journal of Applied Crystallography,
1999
19.
APEX2 (Version 2.1), SAINTPlus, Data Reduction and Correction Program (Version 7.31A), Bruker AXS, Madison, WI, 2006.