Keywords
Antibacterial activity
catalytic activity
chitosan
Mannich reaction
propargylic amines
pyridoxal
zinc(II) complexes
Abstract
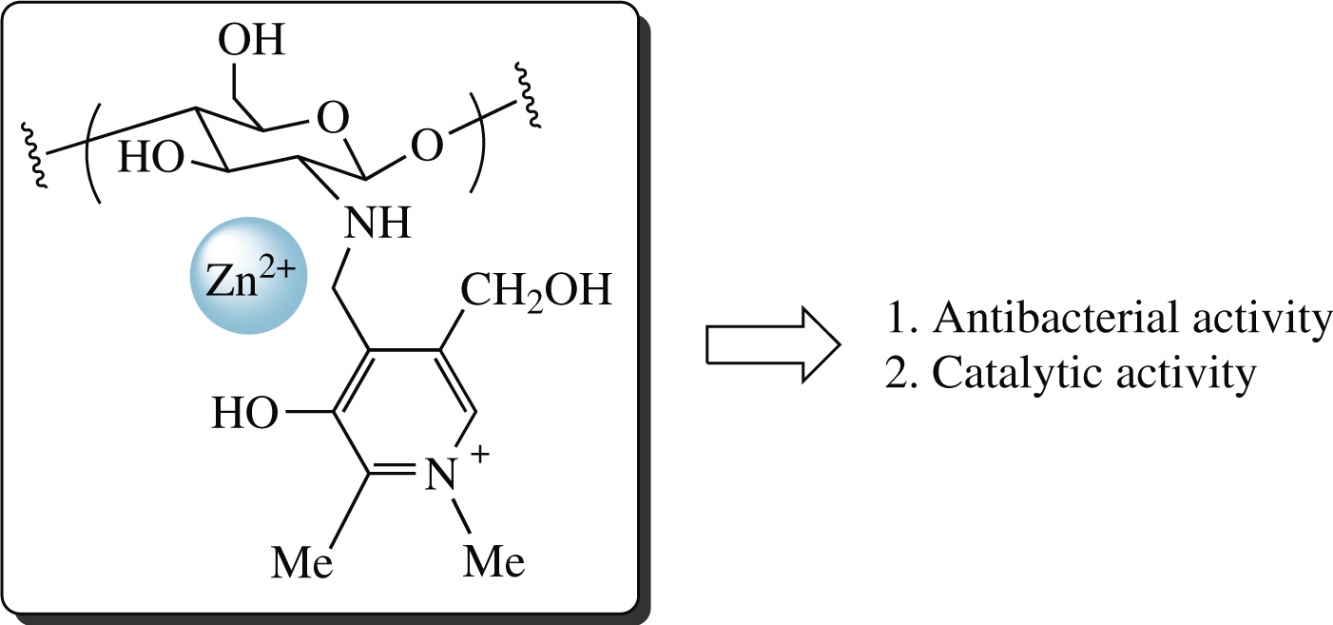
Ultrasonic treatment of the reaction between chitosan and aromatic aldehyde, 4-formyl-3-hydroxy-5-hydroxymethyl-1,2-dimethylpyridin-1-ium iodide, resulted in its significant acceleration and increase in degree of derivatization. The CH=N bond of the prepared Schiff base was reduced, and the formed polymeric material was converted into the corresponding zinc(II) composite by treatment with ZnCl2 and NaOH. The obtained composite is non-toxic, possesses high antibacterial activity and can be employed as the catalyst in the Mannich reaction between benzaldehyde, phenylacetylene and piperidine.
References
2.

Raik S., Andranovitš S., Petrova V., Xu Y., Lam J., Morris G., Brodskaia A., Casettari L., Kritchenkov A., Skorik Y.
Polymers,
2018
3.

Zemskova L.A., Shlyk D.K., Voit A.V., Barinov N.N.
Russian Chemical Bulletin,
2019
4.

Alexandrova V.A., Revina A.A., Busev S.A., Sadykova V.S.
Russian Chemical Bulletin,
2018
5.
Sedyakina N.E., Silaeva A.O., Krivoshchepov A.F., Avramenko G.V.
Mendeleev Communications,
2018
6.
Sedyakina N.E., Feldman N.B., Lutsenko S.V., Avramenko G.V.
Mendeleev Communications,
2018
7.
Yaroslavov A.A., Efimova A.A., Mulashkin F.D., Rudenskaya G.N., Krivtsov G.G.
Mendeleev Communications,
2018
8.

Zargar V., Asghari M., Dashti A.
ChemBioEng Reviews,
2015
9.

Pillai C.K., Paul W., Sharma C.P.
Progress in Polymer Science,
2009
10.
10.1016/j.mencom.2020.09.030_bib0050
Muzzarelli
2005
11.

Kritchenkov A.S., Egorov A.R., Artemjev A.A., Kritchenkov I.S., Volkova O.V., Kiprushkina E.I., Zabodalova L.A., Suchkova E.P., Yagafarov N.Z., Tskhovrebov A.G., Kurliuk A.V., Shakola T.V., Khrustalev V.N.
International Journal of Biological Macromolecules,
2020
12.
10.1016/j.mencom.2020.09.030_bib0060
Parhi
2019
13.
10.1016/j.mencom.2020.09.030_bib0065
Morin-Crini
2019
14.

Kritchenkov A.S., Egorov A.R., Dysin A.P., Volkova O.V., Zabodalova L.A., Suchkova E.P., Kurliuk A.V., Shakola T.V.
International Journal of Biological Macromolecules,
2019
15.

Kritchenkov A.S., Egorov A.R., Artemjev A.A., Kritchenkov I.S., Volkova O.V., Kurliuk A.V., Shakola T.V., Rubanik V.V., Rubanik V.V., Tskhovrebov A.G., Yagafarov N.Z., Khrustalev V.N.
International Journal of Biological Macromolecules,
2020
16.

Kritchenkov A.S., Egorov A.R., Volkova O.V., Kritchenkov I.S., Kurliuk A.V., Shakola T.V., Khrustalev V.N.
International Journal of Biological Macromolecules,
2019
17.

Kritchenkov A.S., Skorik Y.A.
Russian Chemical Bulletin,
2017
18.

Antony R., Arun T., Manickam S.T.
International Journal of Biological Macromolecules,
2019
19.
10.1016/j.mencom.2020.09.030_bib0095
Yu
Asian J. Chem.,
2007
20.

Parsaee Z., Haratipour P., Lariche M.J., Vojood A.
Ultrasonics Sonochemistry,
2018
21.
22.

Al-Blewi F.F., Rezki N., Al-Sodies S.A., Bardaweel S.K., Sabbah D.A., Messali M., Aouad M.R.
Chemistry Central Journal,
2018
23.

Kritchenkov A.S., Luzyanin K.V., Bokach N.A., Kuznetsov M.L., Gurzhiy V.V., Kukushkin V.Y.
Organometallics,
2013
24.

Ramu E., Varala R., Sreelatha N., Adapa S.R.
Tetrahedron Letters,
2007
25.

Iwanejko J., Wojaczyńska E., Olszewski T.K.
Current Opinion in Green and Sustainable Chemistry,
2018
26.

Qiu Y., Qin Y., Ma Z., Xia W.
Chemistry Letters,
2014