Keywords
antimicrobial activity
atrane
cinnamate
cobalt complex
crystal structure
thermal stability
triethanolamine
zinc complex
Abstract
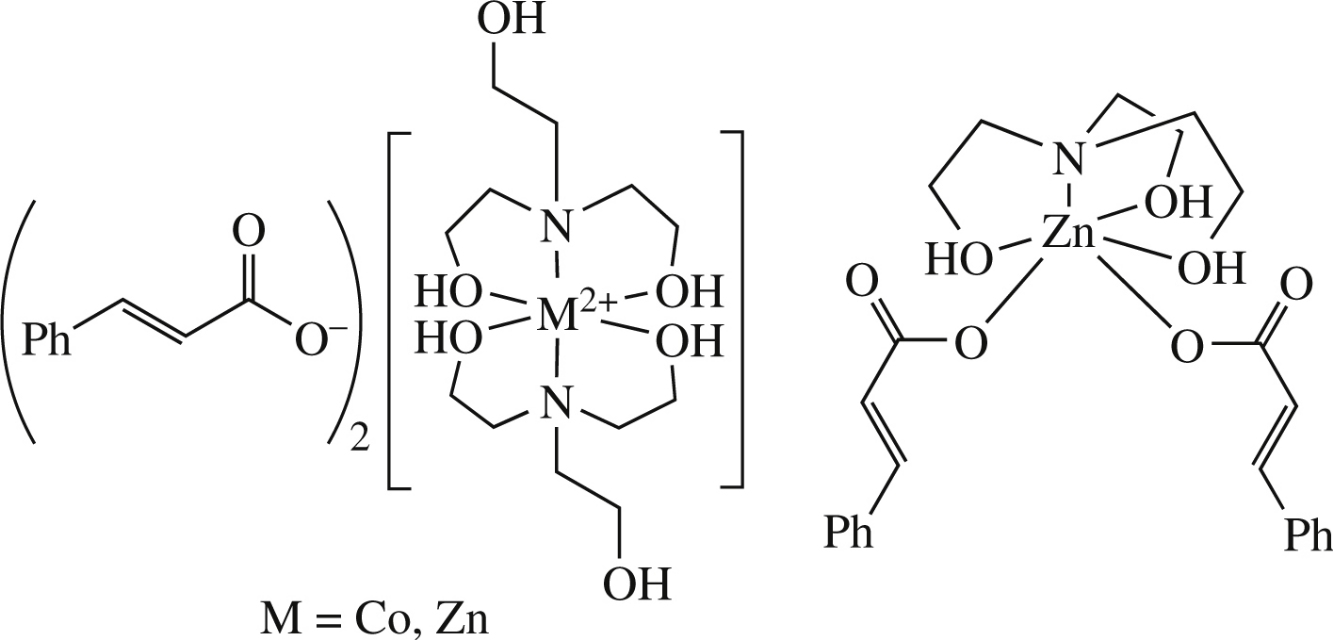
New hydrometallatranes, viz. complexes of triethanolamine with Znii and Coii cinnamates, have been synthesized and characterized by IR spectroscopy, ESI mass spectrometry, elemental and thermal analyses. The structure of cobalt complex has been solved using single-crystal X-ray diffraction. The compounds synthesized and their analogues have been tested for antimicrobial activity.
References
1.

Voronkov M.G., Baryshok V.P.
Herald of the Russian Academy of Sciences,
2010
2.

Voronkov M.G., Kolesnikova O.P., Rasulov M.M., Mirskova A.N.
Pharmaceutical Chemistry Journal,
2007
3.

Voronkov M.G., Baryshok V.P.
Pharmaceutical Chemistry Journal,
2004
4.
Adamovich S.N., Oborina E.N., Ushakov I.A.
Mendeleev Communications,
2019
5.
Smirnov V.I., Zelbst E.A., Kuznetzova G.A., Sterkhova I.V.
Mendeleev Communications,
2018
6.

Ignatyev I., Kondratenko Y., Fundamensky V., Kochina T.
Transition Metal Chemistry,
2017
7.

Topcu Y., Yilmaz V.T., Thöne C.
Acta Crystallographica Section E Structure Reports Online,
2001
8.

Mirskova A.N., Adamovich S.N., Mirskov R.G., Schilde U.
Chemistry Central Journal,
2013
9.

Kondratenko Y., Fundamensky V., Ignatyev I., Zolotarev A., Kochina T., Ugolkov V.
Polyhedron,
2017
10.

Kondratenko Y., Zolotarev A.A., Ignatyev I., Ugolkov V., Kochina T.
Transition Metal Chemistry,
2019
11.

Escovar R.M., Thurston J.H., Ould-Ely T., Kumar A., Whitmire K.H.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2005
12.

Karabach Y.Y., Kirillov A.M., da Silva M.F., Kopylovich M.N., Pombeiro A.J.
Crystal Growth and Design,
2006
13.
10.1016/j.mencom.2020.09.029_bib0065
Kolesnikova
Byulleten’ Sibirskogo Otdeleniya Rossiiskoi Akademii Meditsinskikh Nauk,
2009
14.

Adamovich S.N., Oborina E.N.
Russian Chemical Bulletin,
2019
15.

Abzaeva K.A., Rasulov M.M., Zhigacheva I.V.
Russian Chemical Bulletin,
2019
16.

Kirillov A.M., Kopylovich M.N., Kirillova M.V., Haukka M., da Silva M.F., Pombeiro A.J.
Angewandte Chemie,
2005
17.

Wang D., Kuang D., Zhang F., Tang S., Jiang W.
European Journal of Organic Chemistry,
2013
18.

Adamovich S.N., Mirskov R.G., Mirskova A.N.
Russian Chemical Bulletin,
2015
19.

Li L., Zhao P., Hu J., Liu J., Liu Y., Wang Z., Xia Y., Dai Y., Chen L.
European Journal of Medicinal Chemistry,
2015
20.

Yen G., Chen Y., Sun F., Chiang Y., Lu S., Weng C.
European Journal of Pharmaceutical Sciences,
2011
21.

Chen D., Zhang B., Liu X., Li X., Yang X., Zhou L.
Bioorganic and Medicinal Chemistry Letters,
2018
22.

Saavedra L.M., Ruiz D., Romanelli G.P., Duchowicz P.R.
Ecotoxicology and Environmental Safety,
2015
23.
10.1016/j.mencom.2020.09.029_bib0115
Shu
Eur. Chem. Bull.,
2013
24.
Stefanovic O., Radojevic I., Comic L.
Hemijska Industrija,
2014
25.

Burla M.C., Caliandro R., Camalli M., Carrozzini B., Cascarano G.L., Giacovazzo C., Mallamo M., Mazzone A., Polidori G., Spagna R.
Journal of Applied Crystallography,
2012
26.
CrysAlisPro, Version 1.171.36.32, Agilent Technologies, 2013.
27.
M07-A5. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 6th edn., NCCLS, Wayne, PA, 2000.
28.
M38-P. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Conidium Forming Filamentous Fungi, NCCLS, Wayne, PA, 1998.
29.

Alves M.J., Ferreira I.C., Froufe H.J., Abreu R.M., Martins A., Pintado M.
Journal of Applied Microbiology,
2013