Keywords
p-aminosalicylic acid
acute toxicity
analgesic activity
aromatic nucleophilic substitution
organofluorine compounds
polyfluoroarenes
salicylamide
tuberculostatic activity
Abstract
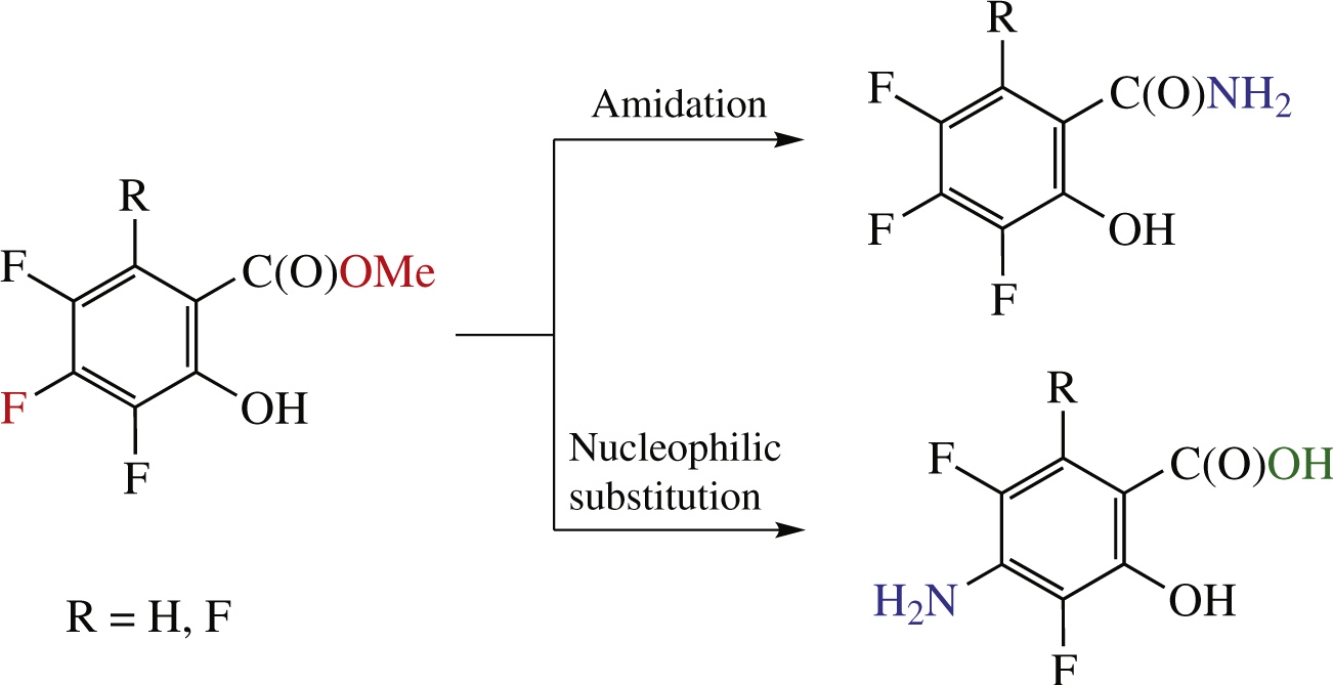
Polyfluorinated analogues of salicylamide and p-aminosalicylic acid have been synthesized based on methyl polyfluorosalicylates. Polyfluorosalicylamides were obtained by the reaction with aqueous ammonia, while 4-aminopolyfluorosalicylic acids were prepared in two steps via regio-oriented nucleophilic replacement of para-positioned fluorine atom with azido group followed by its reduction. 3,4,5-Trifluorosalicylamide showed pronounced analgesic in vivo activity in hot plate test while 4-amino-3,5-difluorosalicylic acid revealed high tuberculostatic activity.
References
1.

Ekinci D., Şentürk M., Küfrevioğlu Ö.İ.
Expert Opinion on Therapeutic Patents,
2011
2.
10.1016/j.mencom.2020.09.028_bib0010
Swan
J.S. Afr. Vet. Assoc.,
1999
3.

Darias V., Bravo L., Abdallah S.S., Mateo C.C., Expósito-orta M.A., Lissavetsky J., Manzanares J.
Archiv der Pharmazie,
1992
4.

Elliott H., Meredith P., Sumner D., Reid J.
British Journal of Clinical Pharmacology,
1984
5.

Evangelista S.
Journal of International Medical Research,
1999
6.

Picchio M., Elisei W., Brandimarte G., Di Mario F., Malfertheiner P., Scarpignato C., Tursi A.
Journal of Clinical Gastroenterology,
2016
7.

Purser S., Moore P.R., Swallow S., Gouverneur V.
Chemical Society Reviews,
2008
8.

Hagmann W.K.
Journal of Medicinal Chemistry,
2008
9.

O’Hagan D.
Journal of Fluorine Chemistry,
2010
10.

Wang J., Sánchez-Roselló M., Aceña J.L., del Pozo C., Sorochinsky A.E., Fustero S., Soloshonok V.A., Liu H.
Chemical Reviews,
2013
11.
10.1016/j.mencom.2020.09.028_bib0055
Fluorine in Medicinal Chemistry and Chemical Biology,
2009
12.

Zafrani Y., Yeffet D., Sod-Moriah G., Berliner A., Amir D., Marciano D., Gershonov E., Saphier S.
Journal of Medicinal Chemistry,
2017
13.
Daněk O.
Collection of Czechoslovak Chemical Communications,
2013
14.
R. Pfirmann, T. Papenfuhs and K. Forstinger, Patent EP 624567, 1994.
15.
U. Bickers, L. Willms and C. Rosinger, Patent US 20050037922, 2005.
16.
M. H. Javaid, S. Gomez, X.-L. F. Cockcroft, K.A. Menear and N. M. B. Martin, Patent WO 2007144639, 2007.
17.

Menear K.A., Adcock C., Alonso F.C., Blackburn K., Copsey L., Drzewiecki J., Fundo A., Le Gall A., Gomez S., Javaid H., Lence C.F., Martin N.M., Mydlowski C., Smith G.C.
Bioorganic and Medicinal Chemistry Letters,
2008
18.
M. Dobler, E. Lacey, N. Le-Van and T. Schuez, Patent GB 2357971, 2001.
19.
J. M. Kauffman and F. Kobarfard, Patent US 6482982, 2002.
20.
10.1016/j.mencom.2020.09.028_bib0100
Esfahanizadeh
Iran. J. Pharm. Res.,
2014
21.

Bazyl' I.T., Kisil' S.P., Burgart Y.V., Saloutin V.I., Chupakhin O.N.
Journal of Fluorine Chemistry,
1999
22.

Shchegol’kov E.V., Shchur I.V., Burgart Y.V., Saloutin V.I., Solodnikov S.Y., Krasnykh O.P., Kravchenko M.A.
Bioorganic and Medicinal Chemistry Letters,
2016
23.

Shchegol'kov E.V., Shchur I.V., Burgart Y.V., Saloutin V.I., Trefilova A.N., Ljushina G.A., Solodnikov S.Y., Markova L.N., Maslova V.V., Krasnykh O.P., Borisevich S.S., Khursan S.L.
Bioorganic and Medicinal Chemistry,
2017
24.

Keana J.F., Cai S.X.
Journal of Organic Chemistry,
1990
25.

Shchur I.V., Shchegolkov E.V., Burgart Y.V., Triandafilova G.A., Maslova V.V., Solodnikov S.Y., Krasnykh O.P., Borisevich S.S., Khursan S.L., Saloutin V.I.
ChemistrySelect,
2019
26.
J. Feutrill, C. Leriche and D. Middlemiss, Patent WO 2013037705A2, 2013.
27.

Newton R., Bowler K.A., Burns E.M., Chapman P.J., Fairweather E.E., Fritzl S.J., Goldberg K.M., Hamilton N.M., Holt S.V., Hopkins G.V., Jones S.D., Jordan A.M., Lyons A.J., Nikki March H., McDonald N.Q., et. al.
European Journal of Medicinal Chemistry,
2016
28.

Zhang J., Gao Y., Kang X., Zhu Z., Wang Z., Xi Z., Yi L.
Organic and Biomolecular Chemistry,
2017
29.
10.1016/j.mencom.2020.09.028_bib0145
Drug Discovery and Evaluation: Pharmacological Assays,
2008
30.
Rukovodstvo po provedeniyu doklinicheskikh issledovanii lekarstvennykh sredstv (Guidelines for Preclinical Studies of Drugs), part 1, ed. A. N. Mironov, Grif&Ko, Moscow, 2012.(in Russian).
31.

Gein V.L., Popov A.V., Kolla V.É., Popova N.A., Potemkin K.D.
Pharmaceutical Chemistry Journal,
1993
32.

Zheng J., Rubin E.J., Bifani P., Mathys V., Lim V., Au M., Jang J., Nam J., Dick T., Walker J.R., Pethe K., Camacho L.R.
Journal of Biological Chemistry,
2013
33.

Collins L., Franzblau S.G.
Antimicrobial Agents and Chemotherapy,
1997
34.

Smith D.S., Lindholm-Levy P., Huitt G.A., Heifets L.B., Cook J.L.
Clinical Infectious Diseases,
2000