Keywords
aldimines
C-amidoalkylation
chloral
cyclopentadiene
Diels–Alder cycloaddition
dienophiles
sulfonylimines
Abstract
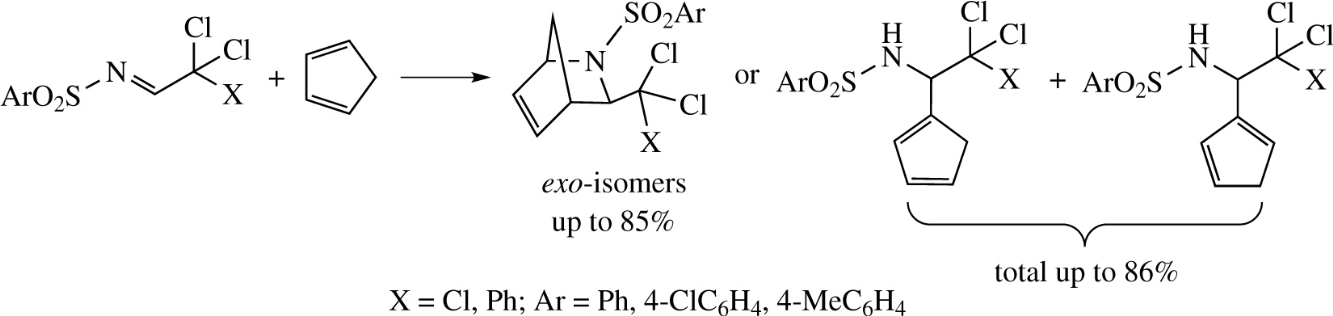
The reaction of chloral or dichloro(phenyl)acetaldehyde N-arylsulfonylimines with cyclopentadiene, depending on the reaction conditions, affords either Diels–Alder adducts, 2-arylsulfonyl-2-azabicyclo[2.2.1]hept-5-enes, or unexpectedly new amidoalkylated derivatives of cyclopentadiene, N-[(cyclopentadienyl)(polychloromethyl)methyl]arenesulfonamides.
References
1.

Levkovskaya G.G., Drozdova T.I., Rozentsveig I.B., Mirskova A.N.
Russian Chemical Reviews,
1999
2.
10.1016/j.mencom.2020.09.022_sbref0005b
De Kimpe
The Chemistry of α-Haloaldehydes and α-Haloimines,
1988
3.

Kimpe N.D., Schamp N.
Organic Preparations and Procedures International,
1979
4.

De Kimpe N., Verhé R., Buyck L.D., Schamp N.
Organic Preparations and Procedures International,
1980
5.

Mangelinckx S., Giubellina N., De Kimpe N.
Chemical Reviews,
2004
6.
Kondrashov E.V., Rozentsveig I.B., Levkovskaya G.G., Mirskova A.N.
Mendeleev Communications,
2003
7.
10.1016/j.mencom.2020.09.022_sbref0010b
Rozentsveig
Russ. J. Org. Chem.,
2000
8.
10.1016/j.mencom.2020.09.022_sbref0010c
Levkovskaya
Russ. J. Org. Chem.,
2000
9.
10.1016/j.mencom.2020.09.022_sbref0010d
Levkovskaya
Russ. J. Org. Chem.,
2000
10.

Rozentsveig I.B., Shainyan B.A., Kondrashov E.V., Rudyakova E.V., Rozentsveig G.N., Chernyshev K.A., Levkovskaya G.G.
Russian Journal of Organic Chemistry,
2008
11.

Kenis S., D'hooghe M., Verniest G., Nguyen V.D., Thi T.A., Nguyen T.V., Kimpe N.D.
Organic and Biomolecular Chemistry,
2011
12.

Rozentsveig I.B., Rozentsveig G.N., Serykh V.Y., Chernyshev K.A., Levkovskaya G.G.
European Journal of Organic Chemistry,
2011
13.

Serykh V.Y., Rozentsveig I.B., Rozentsveig G.N., Chernyshev K.A.
Chemistry of Heterocyclic Compounds,
2012
14.
10.1016/j.mencom.2020.09.022_sbref0015d
Serykh
ARKIVOC,
2018
15.
Gorbunova E.V., Buev E.M., Moshkin V.S., Sosnovskikh V.Y.
Mendeleev Communications,
2019
16.

Pelipko V.V., Baichurin R.I., Makarenko S.V.
Russian Chemical Bulletin,
2019
17.

Callebaut G., Mangelinckx S., Kiss L., Sillanpää R., Fülöp F., De Kimpe N.
Organic and Biomolecular Chemistry,
2012
18.

Soloshonok V., Aceña J., Sorochinsky A.
Synthesis,
2012
19.

Milcent T., Hao J., Kawada K., Soloshonok V.A., Ongeri S., Crousse B.
European Journal of Organic Chemistry,
2014
20.

Weinreb S.M., Staib R.R.
Tetrahedron,
1982
21.

Krow G.R., Pyun C., Rodebaugh R., Marakowski J.
Tetrahedron,
1974
22.
10.1016/j.mencom.2020.09.022_sbref0030b
Mirskova
Zh. Org. Khim.,
1983
23.

Imagawa T., Sisido K., Kawanisi M.
Bulletin of the Chemical Society of Japan,
1973
24.

Raasch M.S.
Journal of Organic Chemistry,
1975
25.

Krow G., Rodebaugh R., Marakowski J., Ramey K.C.
Tetrahedron Letters,
1973
26.

Dunne A.M., Mix S., Blechert S.
Tetrahedron Letters,
2003
27.
Malpass J.R., Belkacemi D., Griffith G.A., Robertson M.D.
Arkivoc,
2002
28.
![Rearrangement of 2-azanorbornenes to tetrahydrocyclopenta[c]pyridines under the action of activated alkynes – A short pathway for construction of the altemicidin core](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Nasirova D.K., Malkova A.V., Polyanskii K.B., Yankina K.Y., Amoyaw P.N., Kolesnik I.A., Kletskov A.V., Godovikov I.A., Nikitina E.V., Zubkov F.I.
Tetrahedron Letters,
2017
29.
![Synthesis of 6-Substituted 7-Bomoazabicyclo[2.2.1]heptanesvia Nucleophilic Addition to 3-Bromo-1-azoniatricyclo[2.2.1.0]-heptane Bromide](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Gayet A., Andersson P.
Advanced Synthesis and Catalysis,
2005
30.
![Absolute Configuration of 2-Substituted 2-Azabicyclo[2.2.1]hept-5-enes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Pombo-Villar E., Boelsterli J., Cid M.M., France J., Fuchs B., Walkinshaw M., Weber H.
Helvetica Chimica Acta,
1993
31.

Baxter E.W., Labaree D., Chao S., Mariano P.S.
Journal of Organic Chemistry,
1989
32.

Baxter E.W., Labaree D., Ammon H.L., Mariano P.S.
Journal of the American Chemical Society,
1990
33.

Bowser A.M., Madalengoitia J.S.
Tetrahedron Letters,
2005
34.

Rossegger E., Oláh L., Fischer R., Kaschnitz P., Varga O., Kállay M., Scheipl G., Stelzer F., Wiesbrock F.
Polymer Chemistry,
2012
35.
![Two-Step Regioselective Synthesis of 3-(Sulfonylamino)imidazo[1,2-a]pyrimidines from 2-Aminopyrimidines andN-(2,2-Dichloro-2-phenylethylidene)arensulfonamides](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Rozentsveig I.B., Serykh V.Y., Chernysheva G.N., Kondrashov E.V., Fedotova A.I., Ushakov I.A., Tretyakov E.V., Romanenko G.V.
European Journal of Organic Chemistry,
2014
36.

Krivdin L.B.
Progress in Nuclear Magnetic Resonance Spectroscopy,
2018
37.

Yonezawa T., Morishima I., Fukuta K., Ohmori Y.
Journal of Molecular Spectroscopy,
1969