Keywords
N-methyl-N-nitrosourea
3a,4,7,7a-tetrahydro-1H-indene
cyclopropanation
diazomethane
palladium catalysis
Abstract
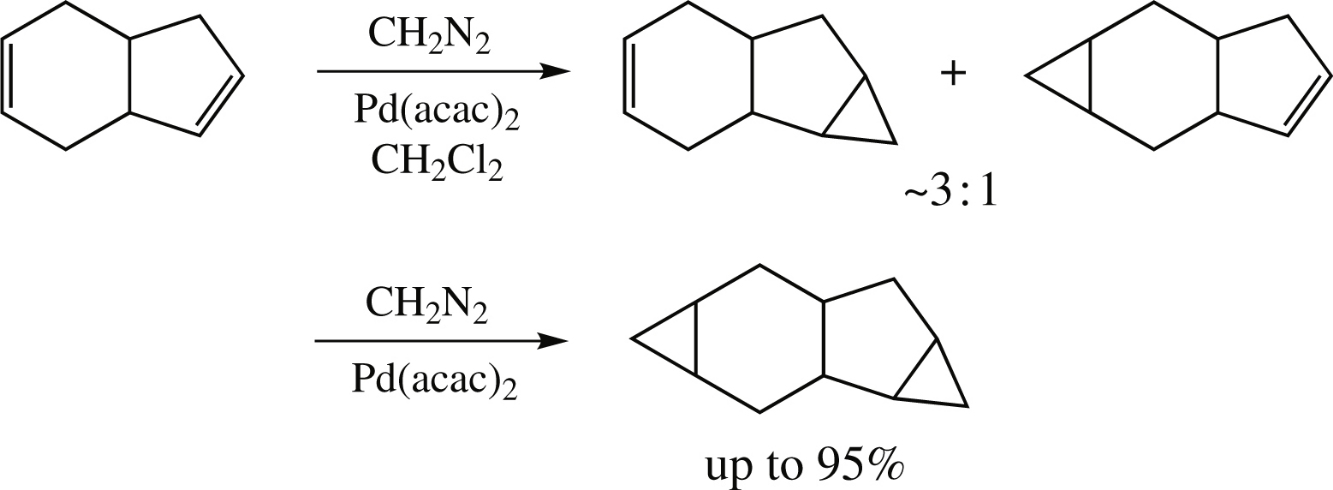
The Pd-catalyzed cyclopropanation of 3a,4,7,7a-tetrahydro-1H-indene with diazomethane unexpectedly affords monoand dicyclopropanation products in good yields, the cyclopentene double bond being approximately three times more reactive than the cyclohexene one. In contrast, similar independent competitive cyclopropanation of a cyclopentene–cyclohexene mixture has shown that cyclohexene exhibits an abnormally low reactivity differing by about two orders of magnitude.
References
1.
10.1016/j.mencom.2020.09.020_bib0005
Burke
Active Pharmaceutical Ingredients in Synthesis: Catalytic Processes in Research and Development,
2018
2.

Fan Y., Gao X., Yue J.
Science China Chemistry,
2016
3.

Keglevich P., Keglevich A., Hazai L., Kalaus G., Szantay C.
Current Organic Chemistry,
2014
4.

Novakov I.A., Babushkin A.S., Yablokov A.S., Nawrozkij M.B., Vostrikova O.V., Shejkin D.S., Mkrtchyan A.S., Balakin K.V.
Russian Chemical Bulletin,
2018
5.
10.1016/j.mencom.2020.09.020_bib0025
Kulinkovich
Cyclopropanes in Organic Synthesis,
2015
6.

Qin Y., Tang P.
Synthesis,
2012
7.

Tomilov Y.V., Menchikov L.G., Novikov R.A., Ivanova O.A., Trushkov I.V.
Russian Chemical Reviews,
2018
8.

Tomilov Y.V., Dokitchev V.A., Dzhemilev U.M., Nefedov O.M.
Russian Chemical Reviews,
1993
9.

Zhang Y., Wang J.
European Journal of Organic Chemistry,
2011
10.
Nefedov O.M., Tomilov Y.V., Kostitsyn A.B., Dzhemilev U.M., Dokitchev V.A.
Mendeleev Communications,
1992
11.
![Catalytic cyclopropanation of spiro[2.4]hepta-4,6-diene with diazomethane](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Shulishov E.V., Pantyukh O.A., Menchikov L.G., Tomilov Y.V.
Tetrahedron Letters,
2019
12.
![Unusual Side Transformation of Spiro[2,4]hepta‐4,6‐dienes into Fulvene Derivatives During Pd‐Catalyzed Cyclopropanation with Diazomethane](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Shulishov E.V., Pantyukh O.A., Menchikov L.G., Tomilov Y.V.
ChemistrySelect,
2020
13.
![Highly Strained Cage Compounds from a Fourfold Bridged Tricyclo[4.2.0.0 2,5 ]octa‐3,7‐diene](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Gleiter R., Pflästerer G., Irngartinger H.
Chemische Berichte,
1993
14.

Ghosh S., Tuhina K., Bhowmik D.R., Venkateswaran R.V.
Tetrahedron,
2007
15.

Szántay C., Hajós G., Incze M., Dörnyei G., Kovács P., Egyed O.
Heterocycles,
2013
16.

Tomilov Y.V., Bordakov V.G., Dolgii I.E., Nefedov O.M.
Russian Chemical Bulletin,
1984
17.

Dzhemilev U.M., Dokichev V.A., Sultanov S.Z., Khursan S.L., Nefedov O.M., Tomilov Y.V., Kostitsyn A.B.
Russian Chemical Bulletin,
1992
18.

Lin Y., Linc C., Liou K., Cheng S., Chang M.
Journal of the Chinese Chemical Society,
1986
19.
10.1016/j.mencom.2020.09.020_bib0095
Molchanov
J. Org. Chem. USSR (Engl. Transl.),
1991
20.
10.1016/j.mencom.2020.09.020_bib0100
Molchanov
ChemInform,
1992
21.

Bulletin of the Korean Chemical Society,
2007
22.
10.1016/j.mencom.2020.09.020_bib0110
Arndt
Org. Synth. Coll.,
1943
23.
10.1016/j.mencom.2020.09.020_bib0115
Duarte
2019
24.

Zhang M., Liu D., Fan G., Wang R., Lu X., Gu Y., Shi Q.
Heterocyclic Communications,
2016
25.

Wang A., Song H., An H., Huang Q., Luo X., Dong J.
Chemistry and Biodiversity,
2015