Keywords
1,2,4-triazines
2,2′-bipyridines
acetylenes
aza-Diels–Alder reaction
nucleophilic substitution of hydrogen
Abstract
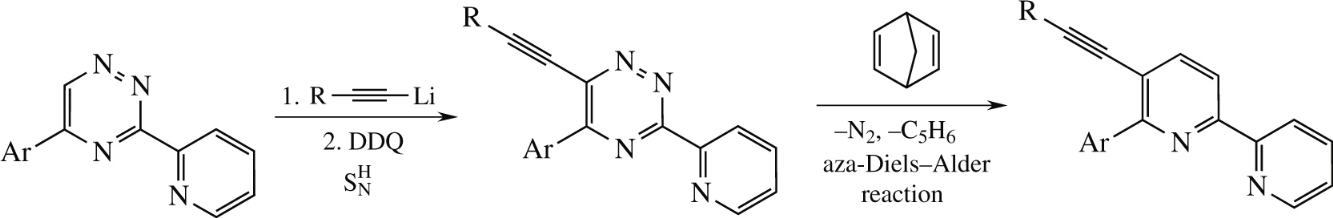
5-Alkynyl-6-aryl-2,2′-bipyridines were conveniently prepared in two steps comprising oxidative SN H ethynylation of 5-aryl-3-(2-pyridyl)-1,2,4-triazines at position 6. At the second step, the 1,2,4-triazine moiety was transformed into the pyridine one employing aza-Diels–Alder reaction with 2,5-norbornadiene.
References
1.

Birckner E., Grummt U.-., Göller A.H., Pautzsch T., Egbe D.A., Al-Higari M., Klemm E.
Journal of Physical Chemistry A,
2001
2.

Harriman A., Hissler M., Ziessel R.
Physical Chemistry Chemical Physics,
1999
3.

Karnahl M., Krieck S., Görls H., Tschierlei S., Schmitt M., Popp J., Chartrand D., Hanan G.S., Groarke R., Vos J.G., Rau S.
European Journal of Inorganic Chemistry,
2009
4.

Vicente J., Gil-Rubio J., Barquero N., Jones P.G., Bautista D.
Organometallics,
2008
5.

Goze C., Kozlov D.V., Tyson D.S., Ziessel R., Castellano F.N.
New Journal of Chemistry,
2003
6.

Shiotsuka M., Inui Y., Sekioka Y., Yamamoto Y., Onaka S.
Journal of Organometallic Chemistry,
2007
7.
B. H. Lange and T. W. Schwartz, Patent WO 2003055477 A1, 2003.
8.

Camerel F., Ulrich G., Ziessel R.
Organic Letters,
2004
9.

Wang C., Ikhlef D., Kahlal S., Saillard J., Astruc D.
Coordination Chemistry Reviews,
2016
10.
Knyazeva I.R., Matveeva V.I., Khrizanforova V.V., Syakaev V.V., Budnikova Y.H., Habicher W.D., Burilov A.R.
Mendeleev Communications,
2019
11.
Callegario Zacchi C.H., Maior Federighi S.S., Ramos Gadelha F., Terra Martins F., Brondi Alves R., de Fátima Â.
Mendeleev Communications,
2018
12.
Druzina A.A., Shmalko A.V., Andreichuk E.P., Zhidkova O.B., Kosenko I.D., Semioshkin A., Sivaev I.B., Mandal S., Shen Z., Bregadze V.I.
Mendeleev Communications,
2019
13.

Conjugated Microporous Polymer as Heterogeneous Ligand for Highly Selective Oxidative Heck Reaction.
Zhou Y., Wang Y., Ning L., Ding Z., Wang W., Ding C., Li R., Chen J., Lu X., Ding Y., Zhan Z.
Journal of the American Chemical Society,
2017
14.
J. Chen and C. Lu, Patent CN 109678896 A, 2019.
15.

Kopchuk D.S., Taniya O.S., Khasanov A.F., Krinochkin A.P., Kovalev I.S., Pospelova T.A., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2019
16.

Prokhorov A.M., Slepukhin P.A., Kozhevnikov D.N.
Journal of Organometallic Chemistry,
2008
17.

Prokhorov A.M., Mąkosza M., Chupakhin O.N.
Tetrahedron Letters,
2009
18.

Charushin V.N., Chupakhin O.N.
Russian Chemical Bulletin,
2019
19.

Chupakhin O.N., Charushin V.N.
Tetrahedron Letters,
2016
20.

Charushin V.N., Chupakhin O.N.
Topics in Heterocyclic Chemistry,
2013
21.
![Synthesis and Pharmacological Evaluation of Phenylethynyl[1,2,4]methyltriazines as Analogues of 3-Methyl-6-(phenylethynyl)pyridine](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Carroll F.I., Kotturi S.V., Navarro H.A., Mascarella S.W., Gilmour B.P., Smith F.L., Gabra B.H., Dewey W.L.
Journal of Medicinal Chemistry,
2007
22.
Khasanov A.F., Kopchuk D.S., Kovalev I.S., Taniya O.S., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2015
23.

Pfu¨ller O.C., Sauer J.
Tetrahedron Letters,
1998
24.

Stanforth S.P., Tarbit B., Watson M.D.
Tetrahedron Letters,
2003
25.

Kozhevnikov V.N., Kozhevnikov D.N., Nikitina T.V., Rusinov V.L., Chupakhin O.N., Zabel M., König B.
Journal of Organic Chemistry,
2003
26.

Kozhevnikov V.N., Shabunina O.V., Kopchuk D.S., Ustinova M.M., König B., Kozhevnikov D.N.
Tetrahedron,
2008
27.

Kozhevnikov D.N., Kozhevnikov V.N., Prokhorov A.M., Ustinova M.M., Rusinov V.L., Chupakhin O.N., Aleksandrov G.G., König B.
Tetrahedron Letters,
2006
28.

Savchuk M.I., Khasanov A.F., Kopchuk D.S., Krinochkin A.P., Nikonov I.L., Starnovskaya E.S., Shtaitz Y.K., Kovalev I.S., Zyryanov G.V., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2019
29.

Krinochkin A.P., Kopchuk D.S., Kovalev I.S., Santra S., Zyryanov G.V., Majee A., Rusinov V.L., Chupakhin O.N.
ChemistrySelect,
2020
30.

Kopchuk D.S., Nikonov I.L., Khasanov A.F., Giri K., Santra S., Kovalev I.S., Nosova E.V., Gundala S., Venkatapuram P., Zyryanov G.V., Majee A., Chupakhin O.N.
Organic and Biomolecular Chemistry,
2018
31.

Khasanov A.F., Kopchuk D.S., Kovalev I.S., Krinochkin A.P., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
AIP Conference Proceedings,
2019