Keywords
acetals
catalysis
Cyclization
furo[2,3-c][1,2]dioxoles
hydrogen peroxide
iodination
iodine
peroxides
phosphomolybdic acid
Abstract
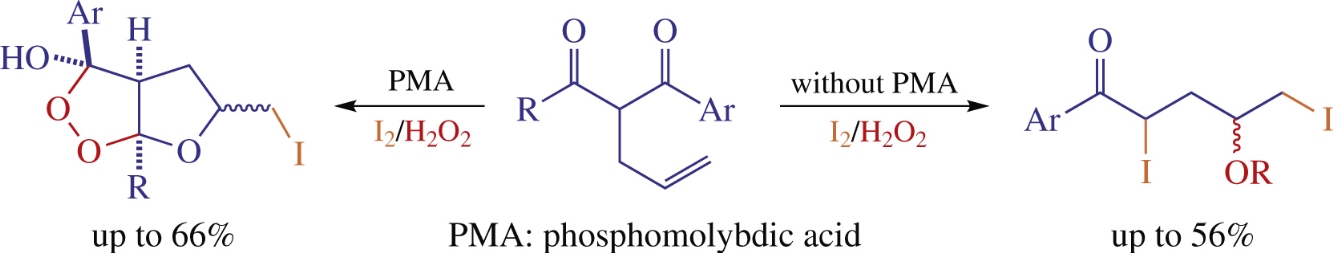
Treatment of 1-aryl-2-allylalkane-1,3-diones with I2/H2O2 system in the presence of catalytic amount of phosphomolybdic acid affords furo[2,3-c][1,2]dioxole derivatives. With other acidic catalysts such as BF3 · Et2O, SnCl4, H2SO4 or TsOH, mixtures with linear 4-acyloxy-2,5-diiodoalkan-1-ones are formed.
References
1.
10.1016/j.mencom.2020.09.018_sbref0005a
Odian
Principles of Polymerization,
2004
2.
10.1016/j.mencom.2020.09.018_sbref0005b
Denisov
Handbook of Free Radical Initiators,
2003
3.

Mott B.T., Tripathi A., Siegler M.A., Moore C.D., Sullivan D.J., Posner G.H.
Journal of Medicinal Chemistry,
2013
4.

Singh C., Kanchan R., Chaudhary S., Puri S.K.
Journal of Medicinal Chemistry,
2012
5.

Rubush D.M., Morges M.A., Rose B.J., Thamm D.H., Rovis T.
Journal of the American Chemical Society,
2012
6.

Cvijetić I.N., Žižak Ž.P., Stanojković T.P., Juranić Z.D., Terzić N., Opsenica I.M., Opsenica D.M., Juranić I.O., Drakulić B.J.
European Journal of Medicinal Chemistry,
2010
7.

Terent’ev A., Krylov I., Borisov D., Nikishin G.
Synthesis,
2007
8.

Terent'ev A.O., Zdvizhkov A.T., Kulakova A.N., Novikov R.A., Arzumanyan A.V., Nikishin G.I.
RSC Advances,
2014
9.

Žmitek K., Zupan M., Iskra J.
Organic and Biomolecular Chemistry,
2007
10.

Žmitek K., Zupan M., Stavber S., Iskra J.
Journal of Organic Chemistry,
2007
11.
![Iodine-catalyzed one-pot synthesis and antimalarial activity evaluation of symmetrically and asymmetrically substituted 3,6-diphenyl[1,2,4,5]tetraoxanes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kumar N., Khan S.I., Sharma M., Atheaya H., Rawat D.S.
Bioorganic and Medicinal Chemistry Letters,
2009
12.
10.1016/j.mencom.2020.09.018_bib0035
Terent’ev
Synthesis,
2009
13.

Jereb M., Zupan M., Stavber S.
Green Chemistry,
2005
14.

Iskra J., Stavber S., Zupan M.
Synthesis,
2004
15.

Kim M.M., Ruck R.T., Zhao D., Huffman M.A.
Tetrahedron Letters,
2008
16.
10.1016/j.mencom.2020.09.018_sbref0045c
Pavlinac
Synthesis,
2006
17.

Jereb M., Zupan M., Stavber S.
Chemical Communications,
2004
18.

Barluenga J., Marco-Arias M., González-Bobes F., Ballesteros A., González J.M.
Chemical Communications,
2004
19.

Jereb M., Iskra J., Zupan M., Stavber S.
Letters in Organic Chemistry,
2005
20.

Trossarello J., Egunjobi A., Morgan W., Ahamed R., Aiken K.
Current Organic Synthesis,
2013
21.

Gaikwad D.D., Dake S.A., Kulkarni R.S., Jadhav W.N., Kakde S.B., Pawar R.P.
Synthetic Communications,
2007
22.

Ferreira V., Ferreira S., Kaiser C.
Synlett,
2008
23.

Froehr T., Sindlinger C.P., Kloeckner U., Finkbeiner P., Nachtsheim B.J.
Organic Letters,
2011
24.

Uyanik M., Okamoto H., Yasui T., Ishihara K.
Science,
2010
25.

Uyanik M., Suzuki D., Yasui T., Ishihara K.
Angewandte Chemie - International Edition,
2011
26.

Jiang H., Huang H., Cao H., Qi C.
Organic Letters,
2010
27.
![Mild metal-free sequential dual oxidative amination of C(sp3)-H bonds: efficient synthesis of imidazo[1,5-a]pyridines.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Yan Y., Zhang Y., Zha Z., Wang Z.
Organic Letters,
2013
28.

Jefford C.W., Li Y., Jaber A., Boukouvalas J.
Synthetic Communications,
1990
29.

Ramirez A., Woerpel K.A.
Organic Letters,
2005
30.

Li Y., Hao H., Wu Y.
Organic Letters,
2009
31.

Hao H., Li Y., Han W., Wu Y.
Organic Letters,
2011
32.

Li Y., Hao H., Zhang Q., Wu Y.
Organic Letters,
2009
33.

Iskra J., Bonnet-Delpon D., Bégué J.
Tetrahedron Letters,
2003
34.

Žmitek K., Stavber S., Zupan M., Bonnet-Delpon D., Charneau S., Grellier P., Iskra J.
Bioorganic and Medicinal Chemistry,
2006
35.

Lu X., Liu Y., Sun B., Cindric B., Deng L.
Journal of the American Chemical Society,
2008
36.

Ghorai P., Dussault P.H.
Organic Letters,
2008
37.

Ghorai P., Dussault P.H.
Organic Letters,
2008
38.

Hamann H., Hecht M., Bunge A., Gogol M., Liebscher J.
Tetrahedron Letters,
2011
39.

Das B., Krishnaiah M., Veeranjaneyulu B., Ravikanth B.
Tetrahedron Letters,
2007
40.

Das B., Veeranjaneyulu B., Krishnaiah M., Balasubramanyam P.
Journal of Molecular Catalysis A Chemical,
2008
41.

Bunge A., Hamann H., Liebscher J.
Tetrahedron Letters,
2009
42.

Azarifar D., Khosravi K., Soleimanei F.
Molecules,
2010
43.

Murahashi S., Zhang D.
Chemical Society Reviews,
2008
44.

Murahashi S., Naota T., Kuwabara T., Saito T., Kumobayashi H., Akutagawa S.
Journal of the American Chemical Society,
1990
45.

Murahashi S., Naota T., Yonemura K.
Journal of the American Chemical Society,
1988
46.

Li Z., Li C.
Journal of the American Chemical Society,
2005
47.

Punniyamurthy T., Rout L.
Coordination Chemistry Reviews,
2008
48.

Saussine L., Brazi E., Robine A., Mimoun H., Fischer J., Weiss R.
Journal of the American Chemical Society,
1985
49.
![A Mechanistic Probe for Oxygen Activation by Metal Complexes and Hydroperoxides and Its Application to Alkane Functionalization by [FeIIICl2tris(2-pyridinylmethyl)amine]+ BF4-](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Arends I.W., Ingold K.U., Wayner D.D.
Journal of the American Chemical Society,
1995
50.

Minisci F., Fontana F., Araneo S., Recupero F.
Journal of the Chemical Society Chemical Communications,
1994
51.

Minisci F., Fontana F., Araneo S., Recupero F., Banfi S., Quici S.
Journal of the American Chemical Society,
1995
52.

Zdvizhkov A.T., Terent’ev A.O., Radulov P.S., Novikov R.A., Tafeenko V.A., Chernyshev V.V., Ilovaisky A.I., Levitsky D.O., Fleury F., Nikishin G.I.
Tetrahedron Letters,
2016