Keywords
Laplacian of electron density
matrix metalloproteinase-2
metalloenzymes
nucleophilic addition
QM/MM
reactivity
Abstract
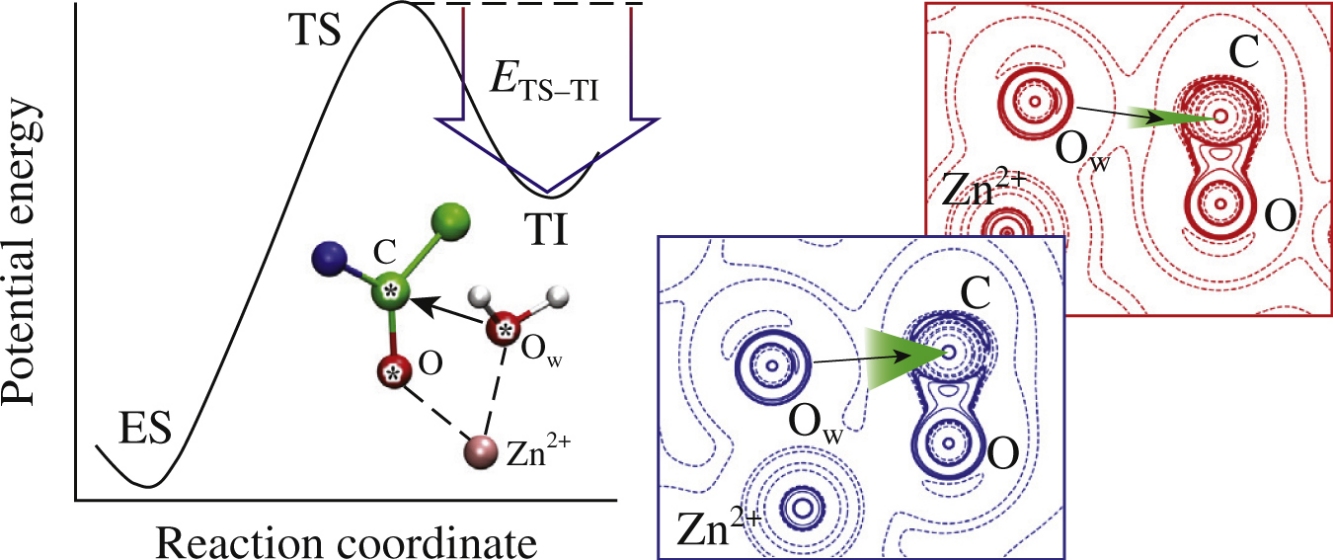
The results of quantum mechanics/molecular mechanics calculations of electron density changes upon oligopeptide hydrolysis by the matrix metalloproteinase-2 are utilized to discriminate between reactive and non-reactive enzyme–substrate complexes. Electron density depletion regions on the carbonyl carbon atom attacked by a catalytic water molecule are found on the 2D maps of electron density Laplacian in the reactive complexes. Also, the computed Fukui function quantitatively describes reactivity of the nucleophilic and electrophilic sites.
References
1.

Radzicka A., Wolfenden R.
Journal of the American Chemical Society,
1996
2.
10.1016/j.mencom.2020.09.010_bib0010
Handbook of Proteolytic Enzymes,
2013
3.

Klein T., Eckhard U., Dufour A., Solis N., Overall C.M.
Chemical Reviews,
2017
4.

Vasilevskaya T., Khrenova M.G., Nemukhin A.V., Thiel W.
Journal of Computational Chemistry,
2015
5.
Vasilevskaya T., Khrenova M.G., Nemukhin A.V., Thiel W.
Mendeleev Communications,
2016
6.

Vasilevskaya T., Khrenova M.G., Nemukhin A.V., Thiel W.
Journal of Computational Chemistry,
2016
7.

Kanaan N., Roca M., Tuñón I., Martí S., Moliner V.
Physical Chemistry Chemical Physics,
2010
8.

Martí S., Roca M., Andrés J., Moliner V., Silla E., Tuñón I., Bertrán J.
Chemical Society Reviews,
2004
9.

Khrenova M.G., Kulakova A.M., Nemukhin A.V.
Organic and Biomolecular Chemistry,
2020
10.

Carroll M.T., Cheeseman J.R., Osman R., Weinstein H.
The Journal of Physical Chemistry,
1989
11.

Shi Z., Boyd R.J.
Journal of the American Chemical Society,
1993
12.

Tsirelson V.G., Shishkina A.V., Stash A.I., Parsons S.
Acta Crystallographica Section B Structural Science,
2009
13.
Khrenova M.G., Tsirelson V.G.
Mendeleev Communications,
2019
14.

Bartashevich E., Matveychuk Y., Tsirelson V.
Molecules,
2019
15.

Varadwaj P.R., Varadwaj A., Jin B.
Physical Chemistry Chemical Physics,
2014
16.

Parr R.G., Yang W.
Journal of the American Chemical Society,
1984
17.
A. A. Granovsky, Fairfly, http://classic.chem.msu.su/gran/firefly/index.html.
18.

Schmidt M.W., Baldridge K.K., Boatz J.A., Elbert S.T., Gordon M.S., Jensen J.H., Koseki S., Matsunaga N., Nguyen K.A., Su S., Windus T.L., Dupuis M., Montgomery J.A.
Journal of Computational Chemistry,
1993
19.

Lu T., Chen F.
Journal of Computational Chemistry,
2011
20.

Humphrey W., Dalke A., Schulten K.
Journal of Molecular Graphics,
1996