Keywords
cocrystals
ferrocene
ferrocenyl
halogen bonding
hydrogen bonding
intermolecular interactions
polyiodide
supramolecular
ternary crystals
Abstract
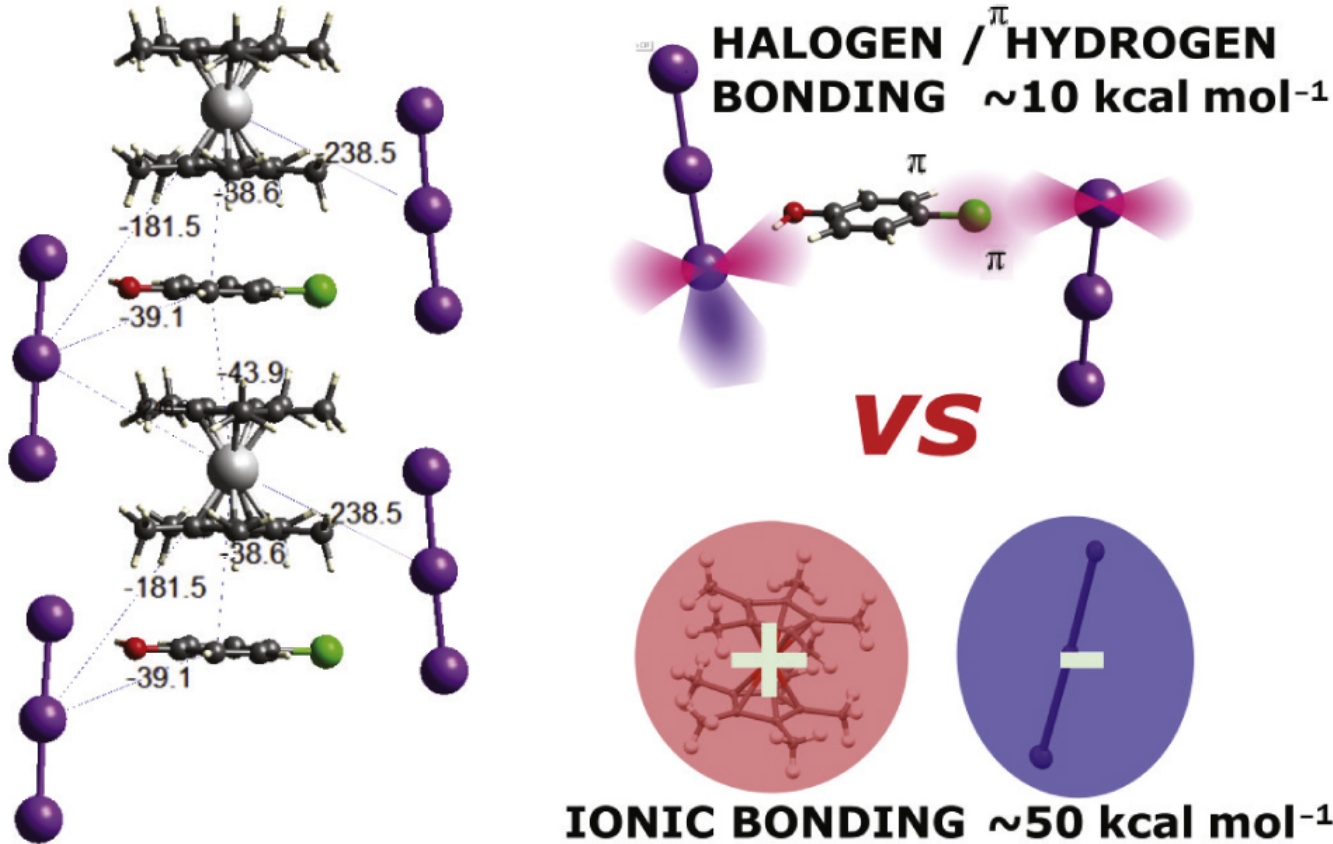
In the cocrystalline salts [(Me5C5)2Fe]+I3−/(4-XC6H4OH) (X = Cl, I) the directionality of \( \mathrm{X \cdots I\text{-}I_2} \) halogen bonds is a significant packing factor notwithstanding their relatively low energies (∼10 kcal mol−1), as compared to the fivefold stronger ionic bonding between [(Me5C5)2Fe+] and [I3]− (∼50 kcal mol−1). This adds significant details to the structural landscape of [(Me5C5)2Fe+]I3 and offers an illustrative example of the stronger structure-defining effect of halogen bonding over the hydrogen one.
References
1.

Aakeröy C.B., Fasulo M.E., Desper J.
Molecular Pharmaceutics,
2007
2.

Childs S.L., Stahly G.P., Park A.
Molecular Pharmaceutics,
2007
3.

Aitipamula S., Banerjee R., Bansal A.K., Biradha K., Cheney M.L., Choudhury A.R., Desiraju G.R., Dikundwar A.G., Dubey R., Duggirala N., Ghogale P.P., Ghosh S., Goswami P.K., Goud N.R., Jetti R.R., et. al.
Crystal Growth and Design,
2012
4.

Mukherjee A.
Crystal Growth and Design,
2015
5.

Ganguly P., Desiraju G.R.
CrystEngComm,
2010
6.
7.

Horn C.J., Blake A.J., Champness N.R., Garau A., Lippolis V., Wilson C., Schröder M.
Chemical Communications,
2003
8.

Aragoni M.C., Arca M., Caltagirone C., Castellano C., Demartin F., Garau A., Isaia F., Lippolis V., Montis R., Pintus A.
CrystEngComm,
2012
9.
![Über die Decamethylferroceniumpolyiodide [(Me5C5)2Fe]Ix mit x = 3, 5 und 6,5. Darstellung und strukturelle Charakterisierung eines Triiodids (DMFc)I3, eines Pentaiodids (DMFc)I5 und eines Hexacosaiodids (DMFc)4I26](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Tebbe K., Buchem R.
Zeitschrift fur Anorganische und Allgemeine Chemie,
1998
10.

Zeitschrift fur Kristallographie - Crystalline Materials,
1995
11.

Sharada D., Saraswatula V.G., Saha B.K.
Crystal Growth and Design,
2018
12.

Tothadi S., Desiraju G.R.
Chemical Communications,
2013
13.

Gamekkanda J.C., Sinha A.S., Desper J., Đaković M., Aakeröy C.B.
New Journal of Chemistry,
2018
14.

Desiraju G.R., Parthasarathy R.
Journal of the American Chemical Society,
1989
15.

Torubaev Y.V., Skabitskiy I.V., Rusina P., Pasynskii A.A., Rai D.K., Singh A.
CrystEngComm,
2018
16.

Torubaev Y.V., Skabitsky I.V.
CrystEngComm,
2019
17.

Bolla G., Nangia A.
Chemical Communications,
2015
18.

Topić F., Rissanen K.
Journal of the American Chemical Society,
2016
19.
![Crystal Solvates of Energetic 2,4,6,8,10,12-Hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane Molecule with [bmim]-Based Ionic Liquids](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Fedyanin I.V., Lyssenko K.A., Fershtat L.L., Muravyev N.V., Makhova N.N.
Crystal Growth and Design,
2019
20.

Nelyubina Y.V., Lyssenko K.A.
Chemistry - A European Journal,
2015