Keywords
1,3-oxathiolanes
bis-1,4-O,S-nucleophiles
cyclopropenium salts
ferrocene
oxathiepines
prop-2-enethioates
Abstract
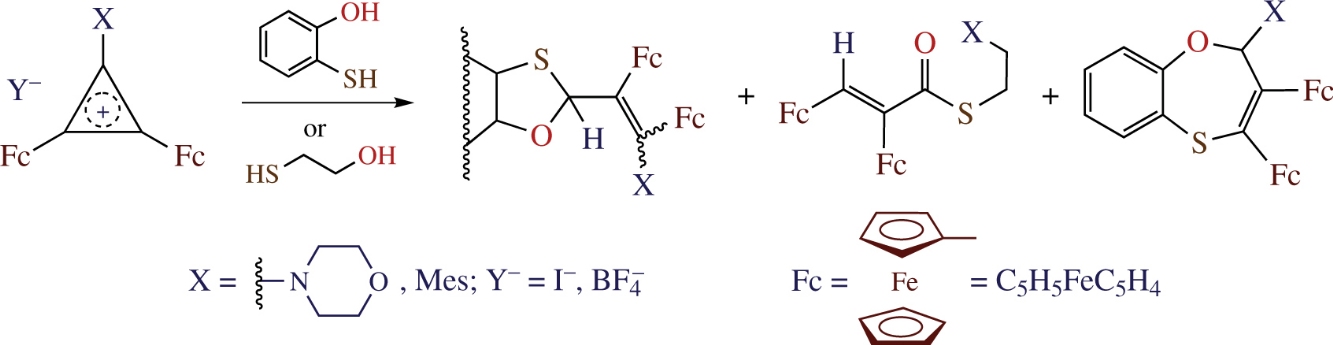
Reactions of 3-R-1,2-diferrocenylcyclopropenium ions (R is 4-morpholino or SMe) with 2-mercaptophenol or 2-mercaptoethanol proceed via a ring opening and result in three types of products, namely, diferrocenyl-substituted 1,3-oxathiolanes, 1,4-oxathiepines and alkyl prop-2-enethioates.
References
1.

Klimova E.I., Berestneva T.K., Ortega S.H., Iturbide D.M., Marquez A.G., García M.M.
Journal of Organometallic Chemistry,
2005
2.

Komatsu K., Kitagawa T.
Chemical Reviews,
2003
3.

4.

Klimova E.I., Klimova T., Álamo M.F., Iturbide D.M., García M.M.
Journal of Heterocyclic Chemistry,
2009
5.

Sánchez García J.J., Flores-Alamo M., Martínez Klimov M.E., Klimova E.I.
Pure and Applied Chemistry,
2016
6.

Klimova E.I., Vázquez López E.A., Alamo M.F., Ortiz-Frade L.A., Hernández-Sánchez G., Sotelo Domínguez V.H., García M.M.
Journal of Heterocyclic Chemistry,
2012
7.
![Synthesis, Electrochemistry and Antitumor Activity of 1’H, 3’H(Me)-spiro-[(aza)benzimidazoline-2’, 3-(1,2-diferrocenylcyclopropenes)], 2-(1,2-Diferrocenylvinyl)benz- and Azabenzimidazoles](/storage/images/resized/v9EPb1eSTDKmfxNHbxZm7U5tRbyjR8FaZCPnRb7F_small_thumb.webp)
García J.J., Ortiz-Frade L., Martínez-Klimova E., Ramos J.C., Flores-Alamo M., Apan T.R., Klimova E.I.
Open Journal of Synthesis Theory and Applications,
2014
8.
![Diferrocenyl(areno)oxazoles, spiro(arenooxazole)cyclopropenes, quinolines and areno[1,4-]oxazines: Synthesis, characterization and study of their antitumor activity](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Sánchez García J.J., Flores-Álamo M., Martínez-Klimova E., Ramírez Apan T., Klimova E.I.
Journal of Organometallic Chemistry,
2018
9.

Wilson G.E., Huang M.G., Schloman W.W.
Journal of Organic Chemistry,
1968
10.

van Staveren D.R., Metzler-Nolte N.
Chemical Reviews,
2004
11.

Shvekhgeimer M.A.
Russian Chemical Reviews,
1996
12.

Sisko J., Kassick A.J., Mellinger M., Filan J.J., Allen A., Olsen M.A.
Journal of Organic Chemistry,
2000
13.

Yavari I., Ghazanfarpour-Darjani M., Hossaini Z., Sabbaghan M., Hosseini N.
Synlett,
2008
14.

Singh M., Shukla G., Nagaraju A., Srivastava A., Verma G., Raghuvanshi K.
Synthesis,
2014
15.

Ozturk T., Ertas E., Mert O.
Chemical Reviews,
2007
16.

Sánchez García J.J., Flores-Alamo M., Ortiz-Frade L., Klimova E.I.
Journal of Organometallic Chemistry,
2017
17.

Jesberger M., Davis T.P., Barner L.
Synthesis,
2003
18.
CrysAlis PRO, CrysAlis RED, Agilent Technologies, Yarnton, Oxfordshire, UK, 2013.
19.

Clark R.C., Reid J.S.
Acta Crystallographica Section A Foundations of Crystallography,
1995
20.
10.1016/j.mencom.2020.09.008_sbref0095a
Sheldrick
Acta Crystallogr.,
2015
21.
10.1016/j.mencom.2020.09.008_sbref0095b
Sheldrick
Acta Crystallogr.,
2015
22.

Farrugia L.J.
Journal of Applied Crystallography,
2012
23.

Macrae C.F., Edgington P.R., McCabe P., Pidcock E., Shields G.P., Taylor R., Towler M., van de Streek J.
Journal of Applied Crystallography,
2006