Keywords
gold compounds
organogold compounds
organoselenium compounds
selenophenes
X-ray
Abstract
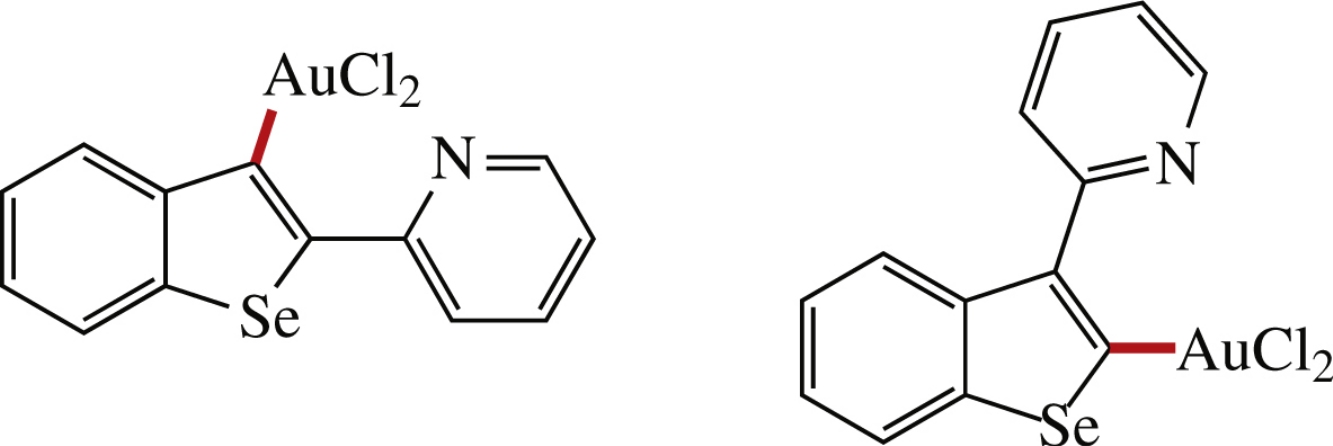
2(3)-Dichloroauryl-3(2)-(pyridin-2-yl)benzo[b]selenophenes bearing covalent bond with gold(iii) were synthesized by the treatment of (pyridin-2-yl)benzoselenophenes with sodium tetrachloroaurate. Molecular structures were unambiguously confirmed by X-ray analysis.
References
1.

Bertrand B., Williams M.R., Bochmann M.
Chemistry - A European Journal,
2018
2.

Bertrand B., Casini A.
Dalton Transactions,
2014
3.

Wenzel M.N., Bonsignore R., Thomas S.R., Bourissou D., Barone G., Casini A.
Chemistry - A European Journal,
2019
4.

González J.J., Ortega E., Rothemund M., Gold M., Vicente C., de Haro C., Bautista D., Schobert R., Ruiz J.
Inorganic Chemistry,
2019
5.

Wang Y., Lackner A.D., Toste F.D.
Accounts of Chemical Research,
2013
6.

Gorin D.J., Sherry B.D., Toste F.D.
Chemical Reviews,
2008
7.

Jiménez-Núñez E., Echavarren A.M.
Chemical Reviews,
2008
8.

Li Z., Brouwer C., He C.
Chemical Reviews,
2008
9.
Yakushkin S.S., Nuzhdin A.L., Artiukha E.A., Plyusnin P.E., Bukhtiyarova G.A., Martyanov O.N.
Mendeleev Communications,
2018
10.

Kaur R., Bariwal J., Voskressensky L.G., Van der Eycken E.V.
Chemistry of Heterocyclic Compounds,
2018
11.

Shu T., Wang J., Su L., Zhang X.
Critical Reviews in Analytical Chemistry,
2018
12.

Pujadas M., Rodríguez L.
Coordination Chemistry Reviews,
2020
13.
Semenova A.A., Tarasov A.B., Goodilin E.A.
Mendeleev Communications,
2019
14.

Do T.G., Hupf E., Lork E., Kögel J.F., Mohr F., Brown A., Toyoda R., Sakamoto R., Nishihara H., Mebs S., Beckmann J.
European Journal of Inorganic Chemistry,
2019
15.
![2,6-Diphenylbenzo[1,2-b:4,5-b‘]dichalcogenophenes: A New Class of High-Performance Semiconductors for Organic Field-Effect Transistors](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Takimiya K., Kunugi Y., Konda Y., Niihara N., Otsubo T.
Journal of the American Chemical Society,
2004
16.
Arsenyan P., Vasiljeva J., Belyakov S.
Mendeleev Communications,
2014
17.

Yang L., Gu W., Lv L., Chen Y., Yang Y., Ye P., Wu J., Hong L., Peng A., Huang H.
Angewandte Chemie - International Edition,
2018
18.

Li P., Schon T.B., Seferos D.S.
Angewandte Chemie - International Edition,
2015
19.
Chulanova E.A., Semenov N.A., Pushkarevsky N.A., Gritsan N.P., Zibarev A.V.
Mendeleev Communications,
2018
20.

Data P., Motyka R., Lapkowski M., Suwinski J., Jursenas S., Kreiza G., Miasojedovas A., Monkman A.P.
Electrochimica Acta,
2015
21.

Chen B., Nie H., Hu R., Qin A., Zhao Z., Tang B.
Science China Chemistry,
2016
22.

Onk I., Hizalan G., Cevher S.C., Hacioglu S.O., Toppare L., Cirpan A.
Journal of Macromolecular Science - Pure and Applied Chemistry,
2017
23.

Petrenko A., Bezvikonnyi O., Volyniuk D., Danyliv Y., Simokaitiene J., Belyakov S., Grazulevicius J.V., Arsenyan P.
New Journal of Chemistry,
2020
24.

Arsenyan P., Petrenko A., Leitonas K., Volyniuk D., Simokaitiene J., Klinavičius T., Skuodis E., Lee J., Gražulevičius J.V.
Inorganic Chemistry,
2019
25.

Petrenko A., Leitonas K., Volyniuk D., Baryshnikov G.V., Belyakov S., Minaev B.F., Ågren H., Durgaryan H., Gražulevičius J.V., Arsenyan P.
Dalton Transactions,
2020
26.

Seki T., Kurenuma S., Ito H.
Chemistry - A European Journal,
2013
27.

Maity A., Sulicz A.N., Deligonul N., Zeller M., Hunter A.D., Gray T.G.
Chemical Science,
2015
28.

Heckler J.E., Zeller M., Hunter A.D., Gray T.G.
Angewandte Chemie - International Edition,
2012
29.

Janzen D.E., Doherty S.R., VanDerveer D.G., Hinkle L.M., Benefield D.A., Vashi H.M., Grant G.J.
Journal of Organometallic Chemistry,
2014