Keywords
Abstract
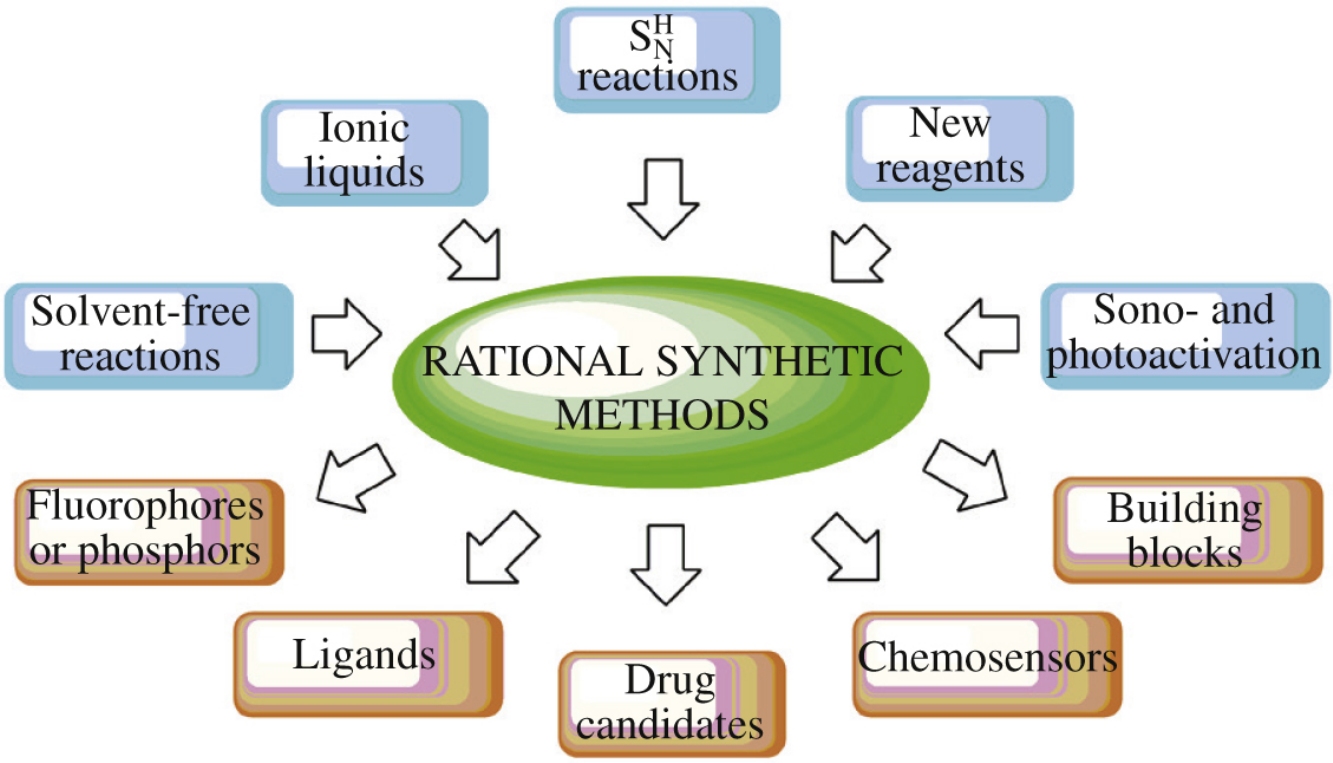
In this focus article, the recent syntheses of various (hetero)aromatic/macrocyclic molecules/scaffolds such as (thia)calixarenes, iptycenes, [poly](aza)arenes, 2,2′bipyridines, 1,2,4triazines, (ox)azoles and aziri(di)nes are reviewed. The employed rational synthetic schemes and green chemistry principles such as solventfree reactions, reactions in ionic liquids, arynemediated reactions, domino and multicomponent reactions, sono, photoand mechanoactivated processes, and direct C–H functionalization are described. The obtained compounds exhibited promising photophysical and coordination properties, intriguing supramolecular properties as well as biological activities. The possible application of these compounds as fluorophores, luminophores, organic phosphors, chemosensors, ligands and drug candidates is discussed.
References









![Zeolite supported Brønsted-acid ionic liquids: an eco approach for synthesis of spiro[indole-pyrido[3,2-e]thiazine] in water under ultrasonication](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)








![Inclusion of Organic Cations by Calix[n]arenes](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
![Association and orientation of C70 on complexation with calix[5]arene](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)


![Cyclooligomeric phenol-aldehyde condensation products. 2. Stereoselective synthesis and DNMR study of two 1,8,15,22-tetraphenyl[14]metacyclophan-3,5,10,12,17,19,24,26-octols](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)


![para-Bridged symmetrical pillar[5]arenes: their Lewis acid catalyzed synthesis and host-guest property.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)

![Synthesis and host-guest properties of pillar[6]arenes](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
![The template effect of solvents on high yield synthesis, co-cyclization of pillar[6]arenes and interconversion between pillar[5]- and pillar[6]arenes](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
![Conversion from Pillar[5]arene to Pillar[6–15]arenes by Ring Expansion and Encapsulation of C60 by Pillar[n]arenes with Nanosize Cavities](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
![Calixarenes. 13. The conformational properties of calix[4]arenes, calix[6]arenes, calix[8]arenes, and oxacalixarenes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
![Counter-ion and solvent effects on the acidity of calix[4]arene and para-tert-butylcalix[6]arene](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
![Solvent-free synthesis of pillar[6]arenes](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
![Synthesis and structural exploration of disulfide bridged [2n] pillararene-like molecules](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)





![p-tert-BUTYLCALIX[8]ARENE](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)









![Synthesis, characterization, and gas transport properties of novel iptycene-based poly[bis(benzimidazobenzisoquinolinones)]](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)



















































![(Benzo[h])Quinolinyl-Substituted Monoazatriphenylenes: Synthesis and Photophysical Properties](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)


















![Syntheses of the benzo[a]naphthacenequinone pigments G-2N and G-2A](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)





![Guest Penetration Deep within the Cavity of Calix[4]arene Hosts: The Tight Binding of Nitric Oxide to Distal (Cofacial) Aromatic Groups](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)

![Sensing and Fixation of NO2/N2O4 by Calix[4]Arenes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)











![Brønsted acidic ionic liquid-catalyzed tandem reaction: an efficient approach towards regioselective synthesis of pyrano[3,2-c]coumarins under solvent-free conditions bearing lower E-factors](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)















![Benzyne-mediated rearrangement of 3-(2-pyridyl)-1,2,4-triazines into 10-(1H-1,2,3-triazol-1-yl)pyrido[1,2-a]indoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)








![Aryne-mediated transformations of 5-perfluorophenyl-substituted 3-(pyridin-2-yl)-1,2,4-triazines in the design of novel 10-(1H-1,2,3-triazol-1-yl)pyrido[1,2-a]indole fluorophores](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)