Keywords
angular H-bond distribution
computer simulations
heat capacity change
hydrophobic and hydrophilic hydration
tetramethylurea
urea
Abstract
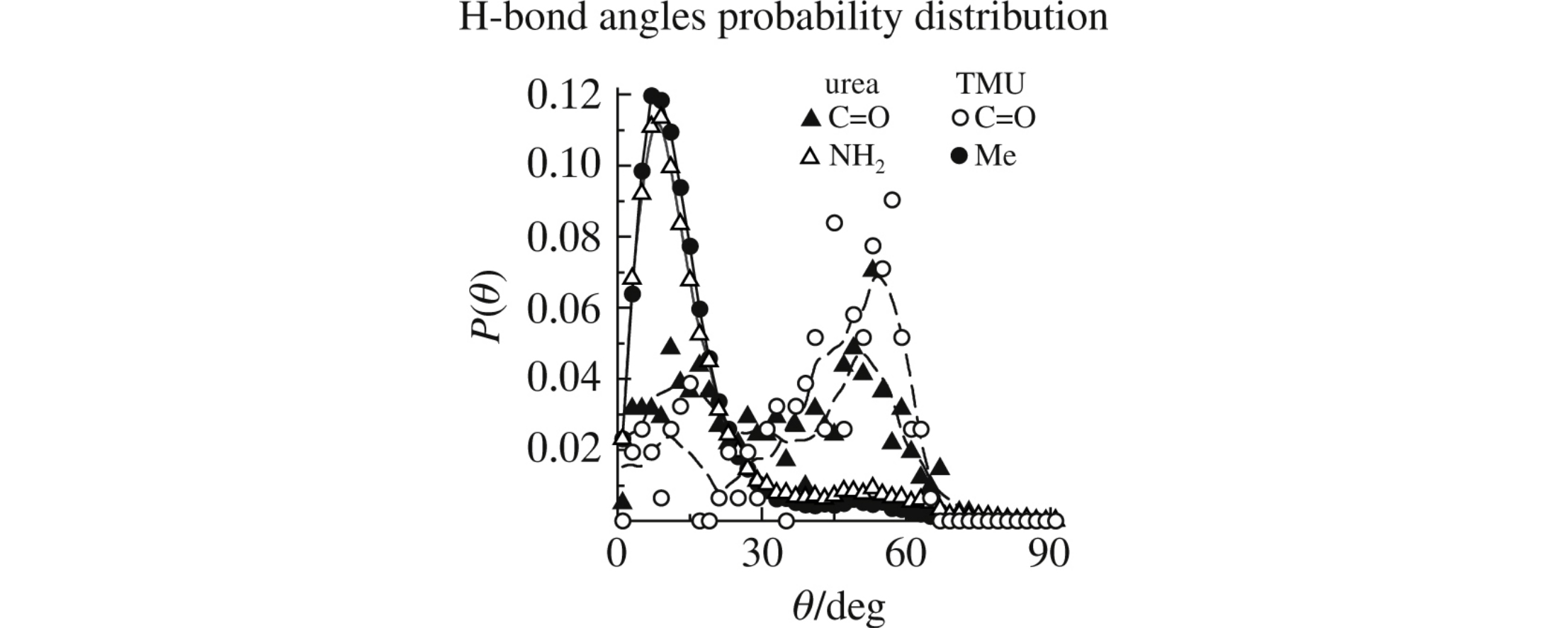
Molecular dynamic simulation of the hydrophilic urea and hydrophobic tetramethylurea aqueous solutions using the random network model of liquid water reveals that apolar Me groups induce an increase in the population of water molecules with linear and shorter H-bonds in their first hydration shell, whereas the carbonyl oxygen atom causes an opposite effect with elevation in the population of high angle/distance water molecules pairs. This behavior of water is the major reason for opposite changes in the heat capacity of hydration for apolar and polar species.
References
1.
K. A. Sharp, Encyclopedia of Life Sciences, 2001, DOI:10.1038/npg. els.0003116.
2.

Pratt L.R.
Annual Review of Physical Chemistry,
2002
3.

Chandler D.
Nature,
2005
4.

Hydrophobic hydration from small to large lengthscales: Understanding and manipulating the crossover
Rajamani S., Truskett T.M., Garde S.
Proceedings of the National Academy of Sciences of the United States of America,
2005
5.
10.1016/j.mencom.2020.07.040_bib0025
Kustov
Gidrofobnye effekty. Strukturnye, termodinamicheskie, prikladnye aspekty. Dostizheniya poslednikh let (Hydrophobic Effects. Structural, Thermodynamic, Applied Aspects. Achievements of the Last Years),
2014
6.

Kustov A.V.
Russian Journal of Inorganic Chemistry,
2011
7.

Kustov A.V., Smirnova N.L.
Journal of Physical Chemistry B,
2011
8.

Swope W.C., Andersen H.C.
The Journal of Physical Chemistry,
1984
9.

Gill S.J., Dec S.F., Olofsson G., Wadsoe I.
The Journal of Physical Chemistry,
1985
10.
10.1016/j.mencom.2020.07.040_bib0050
Kessler
Solvophobic Effects,
1994
11.

Kadtsyn E.D., Anikeenko A.V., Medvedev N.N.
Journal of Molecular Liquids,
2019
12.

Madan B., Sharp K.
The Journal of Physical Chemistry,
1996
13.

Vanzi F., Madan B., Sharp K.
Journal of the American Chemical Society,
1998
14.

Gallagher K.R., Sharp K.A.
Journal of the American Chemical Society,
2003
15.

Henn A.R., Kauzmann W.
The Journal of Physical Chemistry,
1989
16.

Smith W., Yong C.W., Rodger P.M.
Molecular Simulation,
2002
17.

Berendsen H.J., Grigera J.R., Straatsma T.P.
The Journal of Physical Chemistry,
1987
18.

Duffy E.M., Severance D.L., Jorgensen W.L.
Israel Journal of Chemistry,
1993
19.

Weerasinghe S., Smith P.E.
Journal of Physical Chemistry B,
2003
20.

Belletato P., Carlos Gomide Freitas L., Arêas E.P., Se´rgio Santos P.
Physical Chemistry Chemical Physics,
1999
21.

Kustov A.V., Smirnova N.L.
Journal of Chemical & Engineering Data,
2010
22.

Kustov A.V., Smirnova N.L., Antonova O.A.
Journal of Chemical Thermodynamics,
2019
23.

Tovchigrechko A., Rodnikova M., Barthel J.
Journal of Molecular Liquids,
1999