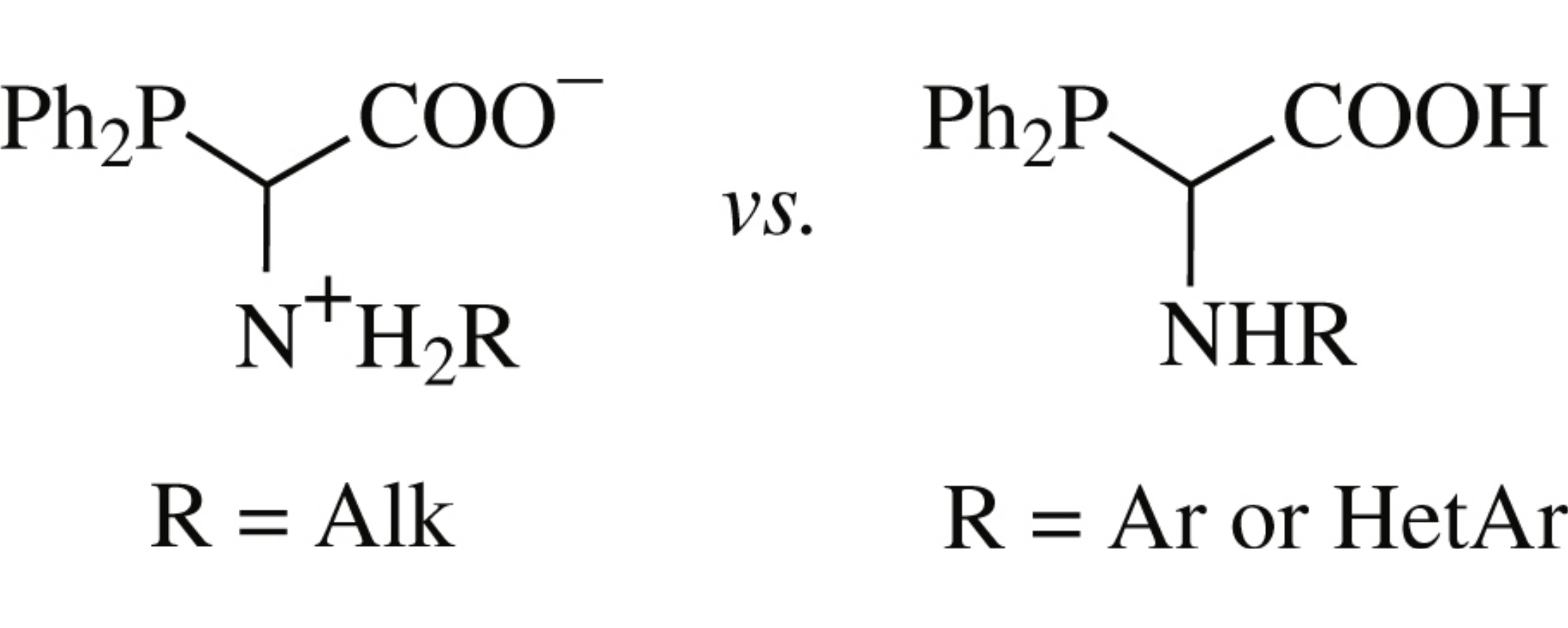Keywords
amino acids
diphenylphosphinoglycines
quantum-chemical calculations
thermodynamic stability
unionized form
X-ray analysis
zwitterionic form
α-diphenylphosphino-α-amino acids
Abstract
The relative thermodynamic stability of unionized and zwitterionic forms of α-phosphino-α-amino acids is governed by the substituent R at the nitrogen atom, namely, (het)aryl substituents favour the formation of the unionized form RHNCH(PPh2)COOH, while in the case of alkyl analogues the zwitterions RH2N+CH(PPh2)COO− predominate. The experimentally observed trends have been supported by quantum-chemical calculations. The synthesis and X-ray crystal structure analysis of a new unionized α-phosphino-α-amino acid [α-diphenylphosphino-N-(2-methoxycarbonylphenyl) glycine] are reported.
References
1.

Kotha S., Goyal D., Chavan A.S.
Journal of Organic Chemistry,
2013
2.

Noisier A.F., Brimble M.A.
Chemical Reviews,
2014
3.

Mondal S., Chowdhury S.
Advanced Synthesis and Catalysis,
2018
4.

Lewis J.C.
Current Opinion in Chemical Biology,
2015
5.

Heravi M.M., Fathi Vavsari V.
Advances in Heterocyclic Chemistry,
2015
6.

Roy S.G., De P.
Journal of Applied Polymer Science,
2014
7.

Zarezin D.P., Nenajdenko V.G.
Russian Chemical Reviews,
2019
8.

Marsh R.E., Donohue J.
Advances in Protein Chemistry,
1967
9.

Peulecke N., Kindermann M.K., Köckerling M., Heinicke J.
Polyhedron,
2012
10.

Kwon I., Yang B.
Industrial & Engineering Chemistry Research,
2017
11.

Harkiss A.H., Sutherland A.
Organic and Biomolecular Chemistry,
2016
12.

Avan I., Hall C.D., Katritzky A.R.
Chemical Society Reviews,
2014
13.

Yu Y., Cui C., Wang J., Lu Y.
Science China Chemistry,
2016
14.
10.1016/j.mencom.2020.07.038_sbref0020e
Hicks
Unnatural Amino Acids: Methods and Protocols,
2012
15.

Young D., Maza J., Jacobs T., Uthappa D.
Synlett,
2016
16.

Dutta A., DuBois D.L., Roberts J.A., Shaw W.J.
Proceedings of the National Academy of Sciences of the United States of America,
2014
17.

Oughli A.A., Ruff A., Boralugodage N.P., Rodríguez-Maciá P., Plumeré N., Lubitz W., Shaw W.J., Schuhmann W., Rüdiger O.
Nature Communications,
2018
18.

Galan B.R., Reback M.L., Jain A., Appel A.M., Shaw W.J.
European Journal of Inorganic Chemistry,
2013
19.

Priyadarshani N., Ginovska B., Bays J.T., Linehan J.C., Shaw W.J.
Dalton Transactions,
2015
20.

Peulecke N., Yakhvarov D.G., Heinicke J.W.
European Journal of Inorganic Chemistry,
2018
21.

Heinicke J., Peulecke N., Jones P.G.
Chemical Communications,
2005
22.

Lach J., Peulecke N., Kindermann M.K., Palm G.J., Köckerling M., Heinicke J.W.
Tetrahedron,
2015
23.

Lach J., Guo C., Kindermann M.K., Jones P.G., Heinicke J.
European Journal of Organic Chemistry,
2010
24.
J. Lach, PhD Thesis, Greifswald, 2009.
25.
10.1016/j.mencom.2020.07.038_sbref0050a
Fomina
Uch. Zap. Kazan. Univ., Ser. Estestv. Nauki,
2012
26.

Lach J., Peulecke N., Jones P.G., Dix I., Heinicke J.W.
Polyhedron,
2016
27.
Soficheva O.S., Bekmukhamedov G.E., Dobrynin A.B., Heinicke J.W., Sinyashin O.G., Yakhvarov D.G.
Mendeleev Communications,
2019
28.

Soficheva O.S., Kislitsyn Y.A., Nesterova A.A., Dobrynin A.B., Yakhvarov D.G.
Russian Journal of Electrochemistry,
2020
29.
APEX2, version 2.1, SAINTPlus, Data Reduction and Correction Program, version 7.31A, Bruker Advanced X-ray Solutions, Bruker AXS, Madison, WI, USA, 2006.
30.
G. M. Sheldrick, SADABS, Program for Empirical X-ray Absorption Correction, Bruker-Nonius, 1990-2004.
31.
10.1016/j.mencom.2020.07.038_bib0075
Sheldrick
Acta Crystallogr.,
2015
32.

Farrugia L.J.
Journal of Applied Crystallography,
1999
33.
10.1016/j.mencom.2020.07.038_bib0085
Spek
Acta Crystallogr.,
2009
34.

Macrae C.F., Bruno I.J., Chisholm J.A., Edgington P.R., McCabe P., Pidcock E., Rodriguez-Monge L., Taylor R., van de Streek J., Wood P.A.
Journal of Applied Crystallography,
2008