Keywords
B. subtilis
E. coli
3,5-dicyanopyridines
antimicrobial activity
bacteria
halogen bonding
Abstract
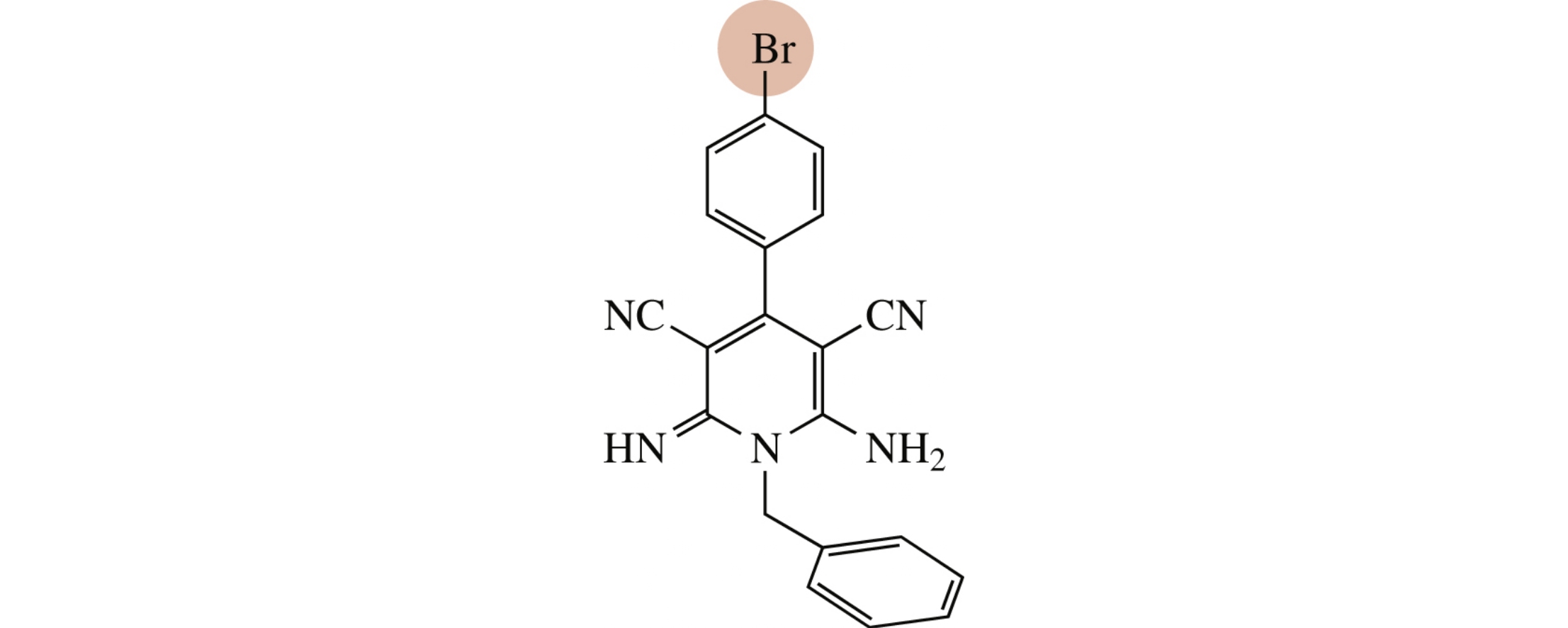
New 2-amino-4-aryl-3-cyanopyridines were obtained by the three-component condensation of arylidenemalononitriles, malononitrile and (di)amine system or acetoacetanilide. Antibacterial activity of all synthesized compounds against the Gram-negative bacteria E. coli and the Gram-positive B. subtilis was tested, the only representative showed a substantial antimicrobial effect.
References
1.

Cabrele C., Reiser O.
Journal of Organic Chemistry,
2016
2.

Taylor A.P., Robinson R.P., Fobian Y.M., Blakemore D.C., Jones L.H., Fadeyi O.
Organic and Biomolecular Chemistry,
2016
3.
Mamedov I.G., Khrustalev V.N., Dorovatovskii P.V., Naghiev F.N., Maharramov A.M.
Mendeleev Communications,
2019
4.
Elinson M.N., Ryzhkov F.V., Vereshchagin A.N., Zaimovskaya T.A., Korolev V.A., Egorov M.P.
Mendeleev Communications,
2016
5.
Chapyshev S.V., Kuzina S.I., Mikhailov A.I.
Mendeleev Communications,
2007
6.

Goda F.E., Abdel-Aziz A.A., Attef O.A.
Bioorganic and Medicinal Chemistry,
2004
7.
![Synthesis and antibacterial activity evaluation of 2,6-bis(6-substituted-1,2,4-triazolo[3,4-b][1,3,4]thiadiazol-3-yl)pyridine derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Xiao H., Li P., Guo D., Hu J., Chai Y., He W.
Medicinal Chemistry Research,
2013
8.

Abadi A.H., Abouel-Ella D.A., Lehmann J., Tinsley H.N., Gary B.D., Piazza G.A., Abdel-Fattah M.A.
European Journal of Medicinal Chemistry,
2010
9.

Eissa A.A., Farag N.A., Soliman G.A.
Bioorganic and Medicinal Chemistry,
2009
10.

Rupert K.C., Henry J.R., Dodd J.H., Wadsworth S.A., Cavender D.E., Olini G.C., Fahmy B., Siekierka J.J.
Bioorganic and Medicinal Chemistry Letters,
2003
11.
WHO News Release: High Levels of Antibiotic Resistance Round Worldwide, New Data Shows, January 2018.
12.

van Duin D., Doi Y.
Virulence,
2016
13.
J. O’Neill, Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations, The Review on Antimicrobial Resistance, https://amr-review.org/, 2014.
14.
10.1016/j.mencom.2020.07.031_bib0070
Naghiyev
Bulg. Chem. Commun.,
2018
15.

Cocco M.T., Congiu C., Lilliu V., Onnis V.
European Journal of Medicinal Chemistry,
2005
16.

Samadi A., de la Fuente Revenga M., Pérez C., Iriepa I., Moraleda I., Rodríguez-Franco M.I., Marco-Contelles J.
European Journal of Medicinal Chemistry,
2013
17.

Samadi A., Marco-Contelles J., Soriano E., Álvarez-Pérez M., Chioua M., Romero A., González-Lafuente L., Gandía L., Roda J.M., López M.G., Villarroya M., García A.G., Ríos C.D.
Bioorganic and Medicinal Chemistry,
2010
18.
10.1016/j.mencom.2020.07.031_bib0090
Mahmoud
J. Chem.,
2018
19.

Sarkar S., Das D.K., Khan A.T.
RSC Advances,
2014
20.

Wilcken R., Zimmermann M.O., Lange A., Joerger A.C., Boeckler F.M.
Journal of Medicinal Chemistry,
2013
21.

Mendez L., Henriquez G., Sirimulla S., Narayan M.
Molecules,
2017