Keywords
Castagnoli–Cushman reaction
cyclic anhydrides
enolization
imines
resonance stabilization
δ-lactams
Abstract
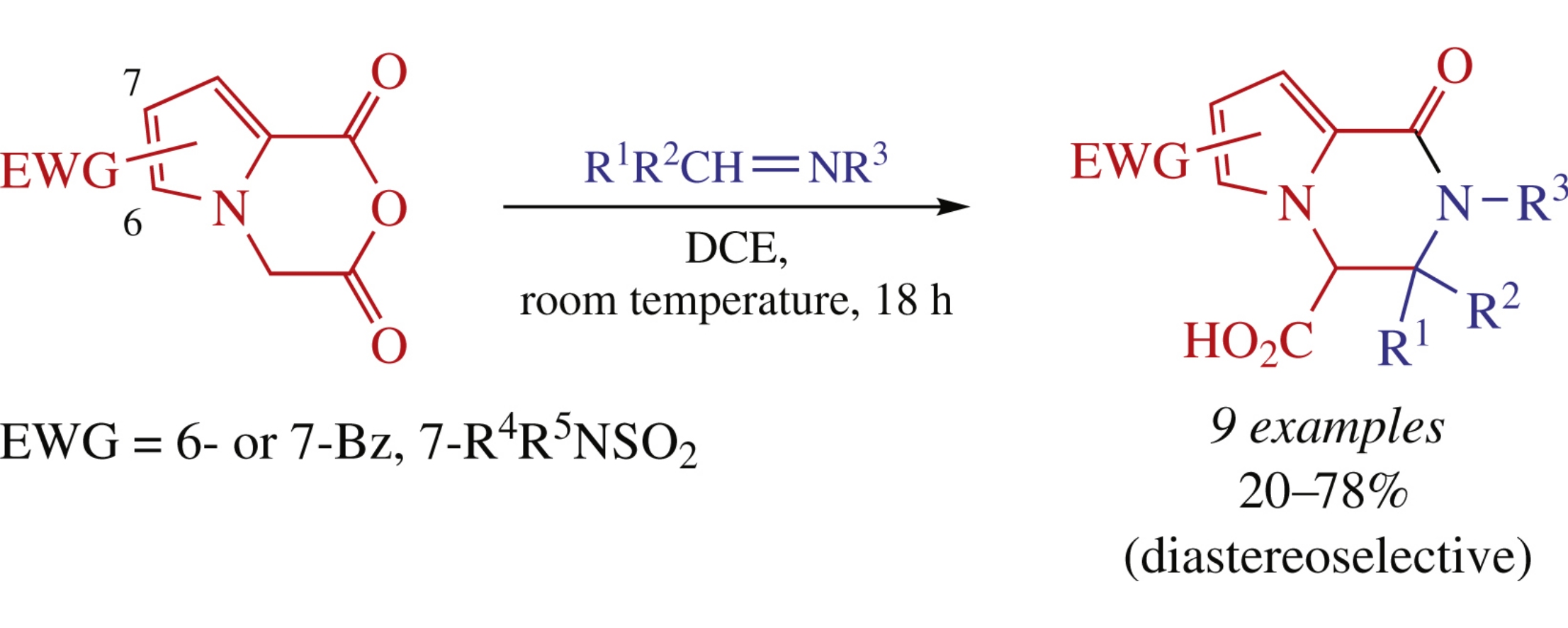
Four anhydrides of 1-(carboxymethyl)pyrrole-2-carboxylic acids bearing electron-withdrawing substituents at positions 6 or 7 of the bicyclic system have been investigated in the Castagnoli–Cushman reaction with imines. 6-Benzoyl- and 7-sulfamoyl-substituted anhydrides demonstrated lower reactivity while 7-benzoyl derivative displayed broader substrate scope. These findings have been rationalized from the mechanistic perspective.
References
1.

Castagnoli N.
Journal of Organic Chemistry,
1969
2.

Cushman M., Castagnoli N.
Journal of Organic Chemistry,
1973
3.

Beng T.K., Langevin S., Farah A.O., Goodsell J., Wyatt K.
New Journal of Chemistry,
2019
4.

Adamovskyi M.I., Ryabukhin S.V., Sibgatulin D.A., Rusanov E., Grygorenko O.O.
Organic Letters,
2016
5.

Krasavin M., Bakulina O., Dar’in D.
Synlett,
2017
6.

Bakulina O., Chizhova M., Dar'in D., Krasavin M.
European Journal of Organic Chemistry,
2018
7.

Krasavin M., Dar’in D.
Tetrahedron Letters,
2016
8.

González-López M., Shaw J.T.
Chemical Reviews,
2009
9.

Chizhova M., Khoroshilova O., Dar’in D., Krasavin M.
Journal of Organic Chemistry,
2018
10.

Ryabukhin S.V., Panov D.M., Granat D.S., Ostapchuk E.N., Kryvoruchko D.V., Grygorenko O.O.
ACS Combinatorial Science,
2014
11.

Zhang S., Han L., Li C., Wang J., Wang W., Yuan Z., Gao X.
Tetrahedron,
2012
12.

Schmuck C., Geiger L.
Journal of the American Chemical Society,
2004