Keywords
C–H functionalization
carbenoids
diazo compounds
homogeneous catalysis
nitrogen heterocycles
organofluorine compounds
purines
Abstract
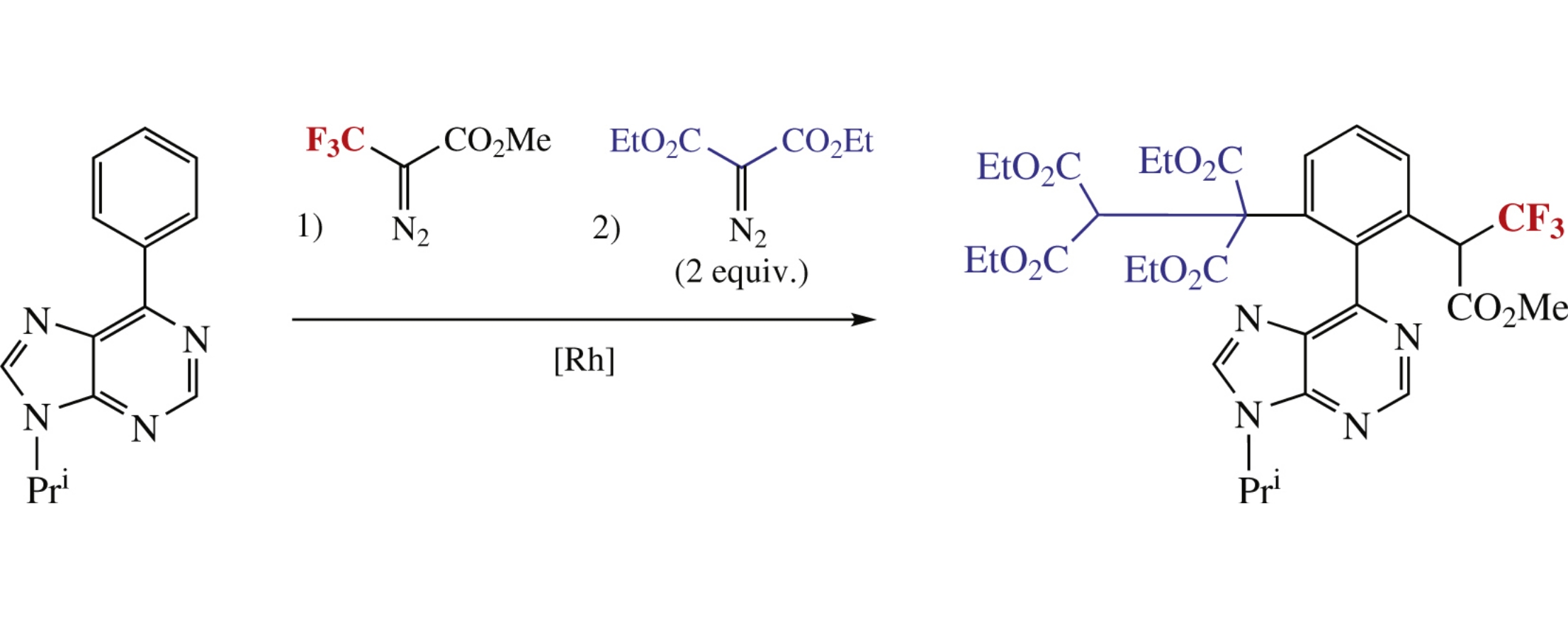
The consecutive alkylation of 9-isopropyl-6-phenyl-9H-purine with methyl 2-diazo-3,3,3-trifluoropropionate and diethyl diazomalonate proceeds at the phenyl substituent firstly via ortho-C–H activation under chelation-assistance of the purine core, followed by classical electrophilic metal carbenoid insertion to the C−H bond of a malonate moiety.
References
1.

Ford A., Miel H., Ring A., Slattery C.N., Maguire A.R., McKervey M.A.
Chemical Reviews,
2015
2.

Ye T., McKervey M.A.
Chemical Reviews,
1994
3.

Zhang L., Zhao J., Mou Q., Teng D., Meng X., Sun B.
Advanced Synthesis and Catalysis,
2020
4.

Ciszewski Ł.W., Durka J., Gryko D.
Organic Letters,
2019
5.
Kuvshinova S.S., Smol’yakov A.F., Vorobyeva D.V., Osipov S.N., Loginov D.A.
Mendeleev Communications,
2018
6.

Wang L., Li Z., Qu X., Peng W.M., Hu S.Q., Wang F.
Russian Journal of General Chemistry,
2018
7.

Yan S., Ling P., Shi B.
Advanced Synthesis and Catalysis,
2017
8.

Iagafarova I.E., Vorobyeva D.V., Loginov D.A., Peregudov A.S., Osipov S.N.
European Journal of Organic Chemistry,
2017
9.

Vorobyeva D.V., Vinogradov M.M., Nelyubina Y.V., Loginov D.A., Peregudov A.S., Osipov S.N.
Organic and Biomolecular Chemistry,
2018
10.

Sambiagio C., Schönbauer D., Blieck R., Dao-Huy T., Pototschnig G., Schaaf P., Wiesinger T., Zia M.F., Wencel-Delord J., Besset T., Maes B.U., Schnürch M.
Chemical Society Reviews,
2018
11.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
12.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
13.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
14.

Pomerantz M., Levanon M., Xiaomin G., Dias H.V.
Tetrahedron,
1997
15.

Wulfman D.S., Peace B.W., McDaniel R.S.
Tetrahedron,
1976
16.

Yoshimoto R., Usuki Y., Satoh T.
Chemistry - An Asian Journal,
2020
17.

Hazra S., Hirano K., Miura M.
Asian Journal of Organic Chemistry,
2019
18.

Yoshimoto R., Usuki Y., Satoh T.
Chemistry Letters,
2019
19.

Golubev A.S., Pasternak P.V., Shidlovskii A.F., Savelèva L.N., Averkiev B.B., Nesterov V.N., Antipin M.Y., Peregudov A.S., Chkanikov N.D.
Journal of Fluorine Chemistry,
2002
20.

Liu Y., Zhou C., Xiong M., Jiang J., Wang J.
Organic Letters,
2018
21.

Iagafarova I.E., Vorobyeva D.V., Peregudov A.S., Osipov S.N.
European Journal of Organic Chemistry,
2015
22.

Tsyshchuk I.E., Vorobyeva D.V., Peregudov A.S., Osipov S.N.
European Journal of Organic Chemistry,
2014
23.

Davies H.M., Manning J.R.
Nature,
2008
24.

Davies H.M., Hedley S.J.
Chemical Society Reviews,
2007