Keywords
dichlorine heptoxide
explosives
furazans
perchloryl fluoride
perchlorylamines
Abstract
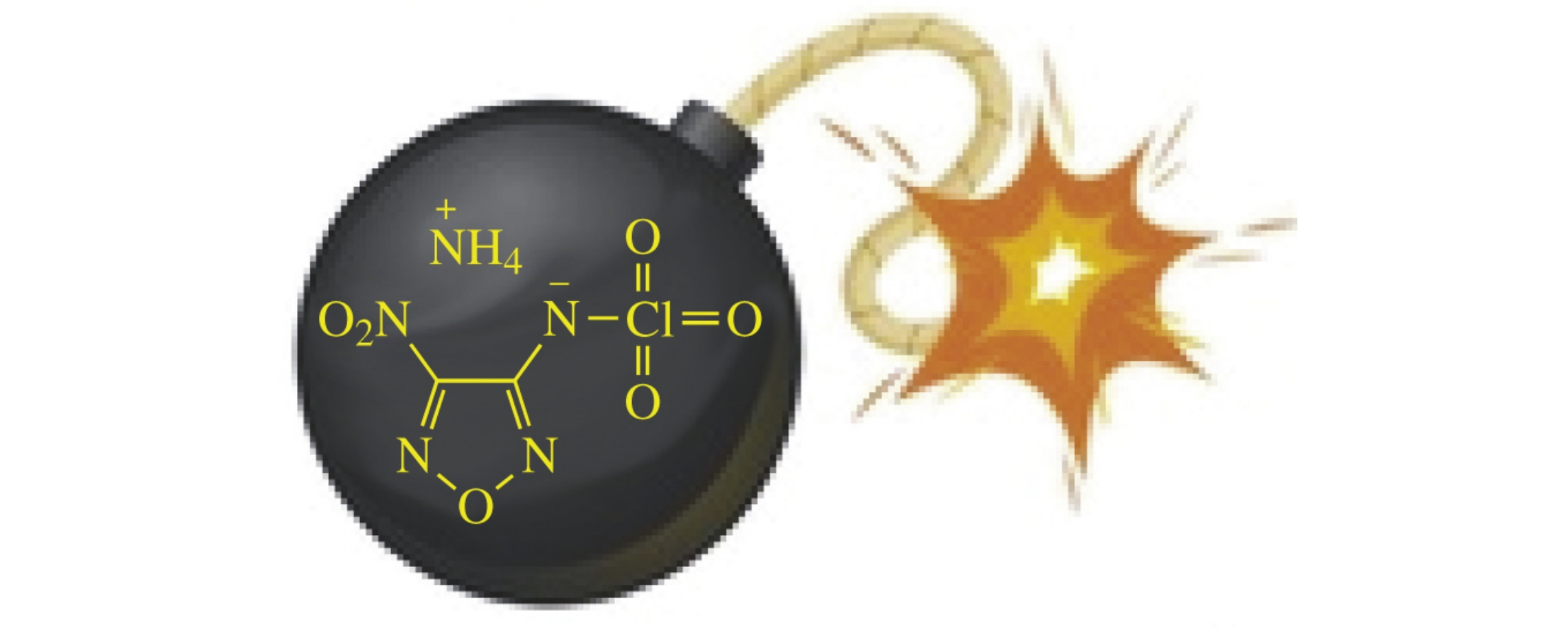
The first perchlorylamino heterocycles, perchlorylaminofurazans, were obtained by the reaction of N-lithiated and N-silylated aminofurazans with FClO3 or Cl2O7. Energetic salts comprising nitrogen-rich 1,2,4-triazolbased cations and 3-nitro-4-(perchlorylamino)furazan anion whose predicted explosive performance was close or superior to that of HMX were synthesized and characterized. Although the explosive output of perchlorylaminofurazan salts is better compared to relative nitraminofurazan ones, they would more readily degrade (explode!) upon impact, friction or heating.
References
1.

Lobanova A.A., Il'yasov S.G., Sakovich G.V.
Russian Chemical Reviews,
2010
2.

Fridman A.L., Ivshin V.P., Novikov S.S.
Russian Chemical Reviews,
1969
3.

Sysolyatin S.V., Lobanova A.A., Chernikova Y.T., Sakovich G.V.
Russian Chemical Reviews,
2005
4.

Sysolyatin S.V., Sakovich G.V., Surmachev V.N.
Russian Chemical Reviews,
2007
5.
10.1016/j.mencom.2020.07.028_sbref0010d
Willer
J. Mex. Chem. Soc.,
2009
6.

Gafarov A.N., Shakirova G.T.
Russian Chemical Bulletin,
2009
7.

Il'yasov S.G., Sakovich G.V., Lobanova A.A.
Propellants, Explosives, Pyrotechnics,
2013
8.
![Oxaazatetracyclo[5.5.0.03, 10.05, 8]Dodecanes – a Promising Foundation for the Design of Thermally Stable, High-Density Energetic Compounds](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Paromov A.E., Sysolyatin S.V.
Chemistry of Heterocyclic Compounds,
2017
9.
10.1016/j.mencom.2020.07.028_sbref0015a
Agrawal
Organic Chemistry of Explosives,
2007
10.

Tang J., Chen D., Zhang G., Yang H., Cheng G.
Synlett,
2019
11.

Badgujar D.M., Talawar M.B., Zarko V.E., Mahulikar P.P.
Combustion, Explosion and Shock Waves,
2019
12.

Zlotin S.G., Dalinger I.L., Makhova N.N., Tartakovsky V.A.
Russian Chemical Reviews,
2020
13.

Engelbrecht A., Atzwanger H.
Journal of Inorganic and Nuclear Chemistry,
1956
14.

Mandell H.C., Barth-Wehrenalp G.
Journal of Inorganic and Nuclear Chemistry,
1959
15.

Golovina N.I., Klitskaya G.A., Atovmyan L.O.
Journal of Structural Chemistry,
1968
16.
10.1016/j.mencom.2020.07.028_sbref0025c
Rosolovskii
Zh. Neorg. Khim.,
1968
17.

Gardner D.M., Helitzer R., Mackley C.J.
Journal of Organic Chemistry,
1964
18.

Pritzkow H.
Zeitschrift fur Naturforschung - Section B Journal of Chemical Sciences,
1976
19.
10.1016/j.mencom.2020.07.028_sbref0035b
Titova
Zh. Neorg. Khim.,
1995
20.

Baumgarten D., Jander J., Hiltl E., Meussdoerffer J.
Zeitschrift fur Anorganische und Allgemeine Chemie,
1974
21.

Beard C.D., Baum K.
Journal of the American Chemical Society,
1974
22.

Hennrichs W., Jander J.
Zeitschrift fur Anorganische und Allgemeine Chemie,
1983
23.

Hennrichs W., Jander J.
Zeitschrift fur Anorganische und Allgemeine Chemie,
1985
24.

Hennrichs W., Jander J.
Zeitschrift fur Anorganische und Allgemeine Chemie,
1987
25.

Titova K.V., Kolmakova E.I., Rosolovskii V.Y.
Russian Chemical Bulletin,
1979
26.

Loginova E.N., Titova K.V., Rosolovskii V.Y.
Russian Chemical Bulletin,
1979
27.
10.1016/j.mencom.2020.07.028_bib0050
Ilyushin
Hanneng Cailiao,
1999
28.

Yin P., Zhang Q., Shreeve J.M.
Accounts of Chemical Research,
2015
29.

Ostrovskii V.A., Popova E.A., Trifonov R.E.
Advances in Heterocyclic Chemistry,
2017
30.

Grigoriev Y.V., Voitekhovich S.V., Karavai V.P., Ivashkevich O.A.
Chemistry of Heterocyclic Compounds,
2017
31.

Kizhnyaev V.N., Golobokova T.V., Pokatilov F.A., Vereshchagin L.I., Estrin Y.I.
Chemistry of Heterocyclic Compounds,
2017
32.

Fershtat L.L., Makhova N.N.
ChemPlusChem,
2019
33.

Suponitsky K.Y., Lyssenko K.A., Antipin M.Y., Aleksandrova N.S., Sheremetev A.B., Novikova T.S.
Russian Chemical Bulletin,
2009
34.

Sheremetev A.B., Aleksandrova N.S., Palysaeva N.V., Struchkova M.I., Tartakovsky V.A., Suponitsky K.Y.
Chemistry - A European Journal,
2013
35.

Sheremetev A.B., Ivanova E.A., Spiridonova N.P., Melnikova S.F., Tselinsky I.V., Suponitsky K.Y., Antipin M.Y.
Journal of Heterocyclic Chemistry,
2005
36.
10.1016/j.mencom.2020.07.028_bib0070
1975
37.

H�hne K., Jander J., Knuth K., Schlegel D.
Zeitschrift fur Anorganische und Allgemeine Chemie,
1971
38.

Kharitonov Y.Y., Karelin A.I., Rosolovoskii V.Y.
Journal of Molecular Structure,
1973
39.

Dmitriev D.E., Strelenko Y.À., Sheremetev A.B.
Russian Chemical Bulletin,
2002
40.

Klapötke T., Schmid P., Stierstorfer J.
Crystals,
2015
41.

Apasov �.T., Sheremet'ev A.B., Dzhetigenov B.A., Kalinin A.V., Tartakovskii V.A.
Russian Chemical Bulletin,
1992
42.

Dmitriev D.E., Strelenko Y.A., Sheremetev A.B.
Russian Chemical Bulletin,
2013
43.

Kustova L.V., Kirpichev E.P., Rubtsov Y.I., Avdonin V.V., Korepin A.G., Eremenko L.T.
Russian Chemical Bulletin,
1981
44.

Kizin A.N., Dvorkin P.L., Ryzhova G.L., Lebedev Y.A.
Russian Chemical Bulletin,
1986
45.

Sagadeev E.V., Gimadeev A.A., Barabanov V.P.
Theoretical Foundations of Chemical Engineering,
2009
46.

Holmes J.L., Aubry C.
Journal of Physical Chemistry A,
2012
47.

Smirnov A.S., Smirnov S.P., Pivina T.S., Lempert D.B., Maslova L.K.
Russian Chemical Bulletin,
2016
48.

Tsyshevsky R., Pagoria P., Smirnov A.S., Kuklja M.M.
Journal of Physical Chemistry C,
2017